Abstract
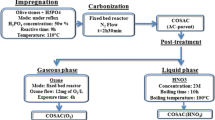
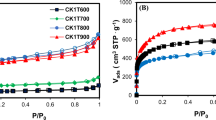
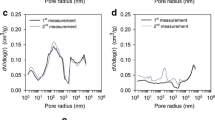
The effectiveness of activated carbon sample (Carbonexp) prepared by KOH chemical activation in hampering oligomerization of multicomponent adsorption was systematically examined. Anoxic (absence of molecular oxygen) and oxic (presence of molecular oxygen) adsorption isotherms of single-solute (2,4-dimethylphenol), binary solute (2-methylphenol/2,4-dimethylphenol), and ternary solute (phenol/2-methylphenol/2,4-dimethylphenol) were studied, using Carbonexp and commercial granular activated carbon F400. Both binary solute adsorption and ternary solute adsorption on Carbonexp indicated no impact of the presence of molecular oxygen on the adsorptive capacity. No significant differences between oxic and anoxic environment were noticed for any multicomponent adsorption systems, which indicated the effectiveness of Carbonexp in hampering the oligomerization of phenolic compounds. On the other hand, in F400, which has lower microporosity and acidic functional groups, significant increases in the adsorptive capacity had been observed when molecular oxygen was present.




Similar content being viewed by others
References
Cooney, D. O., & Xi, Z. P. (1994). Activated carbon catalyzes reactions of phenolics during liquid-phase adsorption. AICHE Journal, 40, 361–364.
Crittenden, J. C., Luft, P., & Hand, D. W. (1985). Prediction of multicomponent adsorption equilibria in background mixtures of unknown composition. Water Research, 19, 1537–1548.
Grant, T. M., & King, C. J. (1990). Mechanism of irreversible adsorption of phenolic-compounds by activated carbons. Industrial and Engineering Chemistry Research, 29, 264–271.
Hamdaoui, O., & Naffrechoux, E. (2007). Modeling of adsorption isotherms of phenol and chlorophenols onto granular activated carbon—Part I. Two-parameter models and equations allowing determination of thermodynamic parameters. Journal of Hazardous Materials, 147, 381–394.
Lu, C. L., Xu, S. P., Gan, Y. X., Liu, S. Q., & Liu, C. H. (2005). Effect of pre-carbonization of petroleum cokes on chemical activation process with KOH. Carbon, 43, 2295–2301.
Lu, Q. L., & Sorial, G. A. (2004a). Adsorption of phenolics on activated carbon—Impact of pore size and molecular oxygen. Chemosphere, 55, 671–679.
Lu, Q. L., & Sorial, G. A. (2004b). The role of adsorbent pore size distribution in multicomponent adsorption on activated carbon. Carbon, 42, 3133–3142.
Lu, Q. L., & Sorial, G. A. (2009). A comparative study of multicomponent adsorption of phenolic compounds on GAC and ACFs. Journal of Hazardous Materials, 167, 89–96.
Mochida, I., Yoon, S. H., & Qiao, W. M. (2006). Catalysts in syntheses of carbon and carbon precursors. Journal of the Brazilian Chemical Society, 17, 1059–1073.
Molina-Sabio, M., & Rodriguez-Reinoso, F. (2004). Role of chemical activation in the development of carbon porosity. Colloids and Surfaces a-Physicochemical and Engineering Aspects, 241, 15–25.
Otowa, T., Nojima, Y., & Miyazaki, T. (1997). Development of KOH activated high surface area carbon and its application to drinking water purification. Carbon, 35, 1315–1319.
Paredes, J. I., Martinez-Alonso, A., Hou, P. X., Kyotani, T., & Tascon, J. M. D. (2006). Imaging the structure and porosity of active carbons by scanning tunneling microscopy. Carbon, 44, 2469–2478.
Pelekani, C., & Snoeyink, V. L. (2000). Competitive adsorption between atrazine and methylene blue on activated carbon: the importance of pore size distribution. Carbon, 38, 1423–1436.
Radke, C. J., & Prausnitz, J. M. (1972). Thermodynamics of multi-solute adsorption from dilute liquid solutions. AICHE Journal, 18, 761.
Singh, B. K., & Rawat, N. S. (1994). Comparative sorption equilibrium studies of toxic phenols on fly-ash and impregnated fly-ash. Journal of Chemical Technology and Biotechnology, 61, 307–317.
Sorial, G. A., Suidan, M. T., Vidic, R. D., & Maloney, S. W. (1993). Competitive adsorption of phenols on Gac.1. Adsorption equilibrium. Journal of Environmental Engineering-Asce, 119, 1026–1043.
Tessmer, C. H., Vidic, R. D., & Uranowski, L. J. (1997). Impact of oxygen-containing surface functional groups on activated carbon adsorption of phenols. Environmental Science and Technology, 31, 1872–1878.
Uranowski, L. J., Tessmer, C. H., & Vidic, R. D. (1998). The effect of surface metal oxides on activated carbon adsorption of phenolics. Water Research, 32, 1841–1851.
Vidic, R. D., Suldan, M. T., & Brenner, R. C. (1993). Oxidative coupling of phenols on activated carbon—Impact on adsorption equilibrium. Environmental Science and Technology, 27, 2079–2085.
Vidic, R. D., Tessmer, C. H., & Uranowski, L. J. (1997). Impact of surface properties of activated carbons on oxidative coupling of phenolic compounds. Carbon, 35, 1349–1359.
Yan, L., & Sorial, G. A. (2011). Chemical activation of bituminous coal for hampering oligomerization of organic contaminants. Journal of Hazardous Materials, 197, 311–319.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, L., Sorial, G.A. Carbon Activation for Hampering Oligomerization of Phenolics in Multicomponent Systems. Water Air Soil Pollut 224, 1588 (2013). https://doi.org/10.1007/s11270-013-1588-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-013-1588-z