Abstract
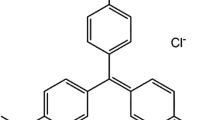
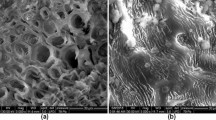
The adsorption capacity of pine tree leaves for removal of methylene blue (MB) from aqueous solution was investigated in a batch system. The effects of the process variables, such as solution pH, contact time, initial dye concentration, amount of adsorbent, agitation speed, salt concentration, and system temperature on the adsorption process were studied. The extent of methylene blue dye adsorption increased with increase in initial dye concentration, contact time, agitation speed, temperature, and solution pH but decreased with increased in amount of adsorbent and salt concentration. Equilibrium data were best described by both Langmuir isotherm and Freundlich adsorption isotherm. The maximum monolayer adsorption capacity of pine tree leaves biomass was 126.58 mg/g at 30 °C. The value of separation factor, R L , from Langmuir equation and Freundlich constant, n, both give an indication of favorable adsorption. The intrapartical diffusion model, liquid film diffusion model, double exponential model, pseudo-first and second order model were used to describe the kinetic and mechanism of adsorption process. A single stage bath adsorber design for the MB adsorption onto pine tree leaves has been presented based on the Langmuir isotherm model equation. Thermodynamic parameters such as standard Gibbs free energy (ΔG 0), standard enthalpy (ΔH 0), and standard entropy (ΔS 0) were calculated.














Similar content being viewed by others
Abbreviations
- A :
-
Activation energy of adsorption (kilojoules per mole)
- C f :
-
Final MB concentration parts per million (milligrams per liter)
- C 0 :
-
Initial MB concentration parts per million (milligrams per liter)
- C t :
-
MB concentration at time t parts per million (milligrams per liter)
- D :
-
Diffusion coefficient (cubic centimeter per second)
- ΔG 0 :
-
Gibbs free energy change (kilojoules per mole)
- ΔH 0 :
-
Enthalpy change (kilojoules per mole)
- ΔS 0 :
-
Entropy change (Joules per kilogram mole)
- k 1 :
-
Pseudo-first order rate constant (per minute)
- k 2 :
-
Pseudo-second order rate constant (milligrams per gram per minute)
- K f :
-
Freundlich adsorption constant (milligrams per gram)
- M :
-
Mass of adsorbent per unit volume (grams per liter)
- m :
-
Amount of adsorbent added (grams)
- n :
-
Freundlich constant
- q :
-
Amount of adsorbate per gram of adsorbent (milligrams per gram)
- q e :
-
Amount of adsorbate per gram of adsorbent at equilibrium (milligrams per gram)
- q t :
-
Amount of adsorbate per gram of adsorbent at any time t
- q m :
-
Equilibrium adsorption capacity using model
- q max :
-
Maximum adsorption capacity (milligrams per gram)
- R 2 :
-
Linear correlation coefficient
- R L :
-
Separation factor
- r 0 :
-
Radius of adsorbent particle (centimeter)
- t :
-
Time (minute)
- T :
-
Temperature (Kelvin)
- V :
-
Volume of the solution (milliliter)
References
Abd EI-Latif, M. M., Ibrahim, A. M., & EI-Kady, M. F. (2010). Adsorption equilibrium, kinetics and thermodynamics of methylene blue from aqueous solutions using biopolymer oak sawdust composite. Journal of American Science, 6, 267–283.
Argun, M. E., Dursun, S., Karatas, M., & Guru, M. (2008). Activation of pine cone using fenton oxidation for Cd (II) and Pb (II) removal. Bioresource Technology, 99, 8691–8698.
Arias, F., & Sen, T. K. (2009). Removal of zinc metal ion (Zn2+) from its aqueous solution by kaolin clay mineral: a kinetic and equilibrium study. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 348, 100–108.
Ayyappan, R., Sophia, A. C., Swaminathan, K., & Sandhya, S. (2005). Removal of Pb (II) from aqueous solution using carbon derived from agricultural wastes. Process Biochemistry, 40, 1293–1299.
Bestani, B., Benderdouche, N., Benstaali, B., Belhakem, M., & Addou, A. (2008). Methylene blue and iodine adsorption onto an activated desert plant. Bioresource Technology, 99, 8441–8444.
Cui, L., Liu, C., & Wu, G. (2008). Performance and mechanism of methylene blue biosorption on orange peel. Environmental Technology, 29, 1021–1030.
Das, N., Vimala, R., & Karthika, P. (2008). Biosorption of heavy metals—an overview. Indian Journal of Biotechnology, 7, 159–169.
Dawood, S., & Sen, T. K. (2012). Removal of anionic dye Congo red from aqueous solution by raw pine and acid-treated pine cone powder as adsorbent: equilibrium, thermodynamic, kinetics, mechanism and process design. Water Research, 46(6), 1933–1946.
Dogan, M., Alkan, M., Türkyilmaz, A. I., & Özdemir, Y. (2004). Kinetics and mechanism of removal of methylene blue by adsorption onto perlite. Journal of Hazardous Materials, 109, 141–148.
Garg, V., Gupta, R., Bala Yadav, A., & Kumar, R. (2003). Dye removal from aqueous solution by adsorption on treated sawdust. Bioresource Technology, 89, 121–124.
Garg, V., Amita, M., Kumar, R., & Gupta, R. (2004). Basic dye (methylene blue) removal from simulated wastewater by adsorption using Indian Rosewood sawdust: a timber industry waste. Dyes and Pigments, 63, 243–250.
Gupta, V., Suhas, Ali, I., & Saini, V. (2004). Removal of rhodamine B, fast green, and methylene blue from wastewater using red mud, an aluminum industry waste. Industrial and Engineering Chemistry Research, 43, 1740–1747.
Haimour, N., & Sayed, S. (1997). Dirsat Nat Eng Sci 24(2), 215–224.
Han, R., Wang, Y., Han, P., Shi, J., Yang, J., & Lu, Y. (2006). Removal of methylene blue from aqueous solution by chaff in batch mode. Journal of Hazardous Materials, 137, 550–557.
Izanloo, H., & Nasseri, S. (2005). Cadmium removal from aqueous solutions by ground pine cone. Iranian Journal of Environmental Health Science & Engineering, 2(1), 33–42.
Jaman, H., Chakraborty, D., & Saha, P. (2009). A study of the thermodynamics and kinetics of copper adsorption using chemically modified rice husk. CLEAN–Soil, Air, Water, 37, 704–711.
Janos, P., Buchtova, H., & Ryznarova, M. (2003). Sorption of dyes from aqueous solutions onto fly ash. Water Research, 37, 4938–4944.
Karagöz, S., Tay, T., Ucar, S., & Erdem, M. (2008). Activated carbons from waste biomass by sulfuric acid activation and their use on methylene blue adsorption. Bioresource Technology, 99, 6214–6222.
Kumar, S., Ramalingam, S., Senthamarai, C., Niranjanaa, M., Vijayalakshmi, P., & Sivanesan, S. (2010). Adsorption of dye from aqueous solution by cashew nut shell: studies on equilibrium isotherm, kinetics and thermodynamics of interactions. Desalination, 261, 52–60.
Mahmoode, N. M., Hagali, B., Ararm, M., & Lan, C. (2011). Adsorption of textile dyes on pine cone from collared wastewater: kinetics, equilibrium and thermodynamic studies. Desalination, 268, 117–125.
Mckay, G., & Allen, S. J. Jr. (1983). Single resistance mass transfer model for the adsorption of dyes on peat. J Separation Sci Technol 4(3), 1–7
Meena, A. K., Mishra, G., Rai, P., Rajagopal, C., & Nagar, P. (2005). Removal of heavy metal ions from aqueous solutions using carbon aerogel as an adsorbent. Journal of Hazardous Materials, 122, 161–170.
Nandi, B., Goswami, A., & Purkait, M. (2009). Removal of cationic dyes from aqueous solutions by kaolin: kinetic and equilibrium studies. Applied Clay Science, 42, 583–590.
Ofomaja, A., Naidoo, E., & Modise, S. (2009). Removal of copper (II) from aqueous solution by pine and base modified pine cone powder as biosorbent. Journal of Hazardous Materials, 168, 909–917.
Ofomaja, A., Naidoo, E., & Modise, S. (2010). Biosorption of copper (II) and lead (II) onto potassium hydroxide treated pine cone powder. Journal of Environmental Management, 91, 1674–1685.
Oladoja, N., Aboluwoye, C., Oladimeji, Y., Ashogbon, A., & Otemuyiwa, I. (2008). Studies on castor seed shell as a sorbent in basic dye contaminated wastewater remediation. Desalination, 227, 190–203.
Pehlivan, E., & Arslan, G. (2007). Removal of metal ions using lignite in aqueous solution–low cost biosorbents. Fuel Processing Technology, 88, 99–106.
Quintelas, C., Rocha, Z., Silva, B., Fonseca, B., Figueiredo, H., & Tavares, T. (2009). Removal of Cd (II), Cr (VI), Fe (III) and Ni (II) from aqueous solutions by an E. coli biofilm supported on kaolin. Chemical Engineering Journal, 149, 319–324.
Robinson, T., Chandran, B., & Nigam, P. (2002). Removal of dyes from an artificial textile dye effluent by two agricultural waste residues, corncob and barley husk. Environment International, 28, 29–33.
Santhi, T., Manonmani, S., Smitha, T., Sugirtha, D., & Mahalakshmi, K. (2009). Uptake of cationic dyes from aqueous solution by bioadsorption onto granular Cucumis sativa. Journal of Applied Sciences in Environmental Sanitation, 4, 29–35.
Sari, A., Tuzen, M., Citak, D., & Soylak, M. (2007). Equilibrium, kinetic and thermodynamic studies of adsorption of Pb (II) from aqueous solution onto Turkish kaolinite clay. Journal of Hazardous Materials, 149, 283–291.
Saueprasearsit, P., ta Nuanjaraen, M., & Chinlapa, M. (2010). Biosorption of lead (Pb2+) by luffa cylindrical fiber. Environmental Research Journal, 4, 157–166.
Sciban, M., Klasnja, M., & Skrbic, B. (2008). Adsorption of copper ions from water by modified agricultural by-products. Desalination, 229, 170–180.
Sen, T. K., Afroze, S., & Ang, H. (2011). Equilibrium, kinetics and mechanism of removal of methylene blue from aqueous solution by adsorption onto pine cone biomass of Pinus radiata. Water, Air, & Soil Pollution, 218(1–4), 499–515.
Shahryari, Z., Goharrizi, A. S., & Azadi, M. (2010). Experimental study of methylene blue adsorption from aqueous solutions onto carbon nano tubes. International Journal of Water Resources and Environmental Engineering, 2, 16–28.
Sharma, Y. C. (2009). Optimization of parameters for adsorption of methylene blue on a low-cost activated carbon. Journal of Chemical & Engineering Data, 55, 435–439.
Sharma, P., Kaur, R., Baskar, C., & Chung, W. J. (2010). Removal of methylene blue from aqueous waste using rice husk and rice husk ash. Desalination, 259, 249–257.
Sivaraj, R., Namasivayam, C., & Kadirvelu, K. (2001). Orange peel as an adsorbent in the removal of acid violet 17 (acid dye) from aqueous solutions. Waste Management, 21, 105–110.
Tarawou, T., & Horsfall, M. (2007). Adsorption of methylene blue dye on pure and carbonized water weeds. Bioremediation Journal, 11, 77–84.
Tsai, W. T., Hsien, K. J., & Hsu, H. C. (2009). Adsorption of organic compounds from aqueous solution onto the synthesized zeolite. Journal of Hazardous Materials, 166, 635–641.
Ucun, H., Bayhan, Y. K., Kaya, Y., Cakici, A., & Faruk Algur, O. (2002). Biosorption of chromium (VI) from aqueous solution by cone biomass of Pinus sylvestris. Bioresource Technology, 85, 155–158.
Ucun H., Bayhan, Y. K., Kaya, Y., Cakici, A., & Algur, O. F. (2003). Biosorption of lead (II) from aqueous solution by cone biomass of Pinus sylvestris. Desalination 154, 233–238.
Vadivelan, V., & Kumar, K. V. (2005). Equilibrium, kinetics, mechanism, and process design for the sorption of methylene blue onto rice husk. Journal of Colloid and Interface Science, 286, 90–100.
Visa, M., Bogatu, C., & Duta, A. (2010). Simultaneous adsorption of dyes and heavy metals from multicomponent solutions using fly ash. Applied Surface Science, 256, 5486–5491.
Wang, X. S., Zhou, Y., Jiang, Y., & Sun, C. (2008). The removal of basic dyes from aqueous solutions using agricultural by-products. Journal of Hazardous Materials, 157, 374–385.
Weber, W. J., & Morris, J. C. (1963). Kinetics of adsorption on carbon from solution. Journal of Sanitary Engineering Division, American Society of Civil Engineers, 89, 31–60.
Yu, Z., Qi, T., Qu, J., Wang, L., & Chu, J. (2009). Removal of Ca (II) and Mg (II) from potassium chromate solution on Amberlite IRC 748 synthetic resin by ion exchange. Journal of Hazardous Materials, 167, 406–412.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yagub, M.T., Sen, T.K. & Ang, H.M. Equilibrium, Kinetics, and Thermodynamics of Methylene Blue Adsorption by Pine Tree Leaves. Water Air Soil Pollut 223, 5267–5282 (2012). https://doi.org/10.1007/s11270-012-1277-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-012-1277-3