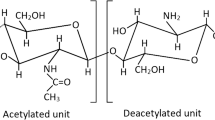
Abstract
In this study, the efficiency of chitin and chitosan toward the removal of ethylbenzene from aqueous solutions was investigated. Batch adsorption experiments of ethylbenzene-contaminated waters (5–200 mg/L) were carried out to evaluate the removal performance. Ethylbenzene uptake was determined from the changes in concentration, as the residual concentration was measured by gas chromatography with mass spectroscopy. The results indicated that the adsorption of ethylbenzene by chitin and chitosan were in agreement with the Langmuir isotherm, for two parameters model, and Redlich–Peterson isotherm, for three parameters model. A maximum removal percentage of 65% of ethylbenzene can be achieved using chitosan as adsorbent material. The adsorption capacity of ethylbenzene followed the order chitosan > chitin. The pseudo-second order rate model described best the adsorption kinetics of ethylbenzene for the two selected adsorbents. The kinetic studies also revealed that the pore diffusion is not the only rate controlling step in the removal of ethylbenzene. Overall, the study demonstrated that chitosan is a potential adsorbent for the removal of ethylbenzene at concentrations as high as 200 mg/L.



Similar content being viewed by others
Abbreviations
- a :
-
Langmuir constant or the maximum adsorbent-phase concentration of adsorbate when surface sites are saturated with adsorbate (milligrammes adsorbate/grammes adsorbent)
- a R :
-
Redlich–Peterson isotherm constant (litres per milligram) to the beta root
- b :
-
Langmuir constant or constant that is related to the free energy of adsorption
- B 1 :
-
Temkin isotherm constant
- B D :
-
Dubinin–Radushkevich isotherm constant
- b T :
-
Temkin isotherm constant
- C o :
-
Initial adsorbate concentration (milligrammes per litre)
- C e :
-
Residual adsorbate concentration at equilibrium (milligrammes per litre)
- E :
-
Mean free energy of adsorption (kilojoules per gramme)
- h :
-
Initial sorption rate (milligrammes per gramme·hour)
- I :
-
Intercept of intra-particle diffusion equation (milligrammes per gramme)
- k o :
-
Constant of Bangham’s equation (grammes)
- k id :
-
Intra-particle diffusion rate constant (milligrams per (grams per square root of hour))
- K :
-
Freundlich constant (milligrams per gram) ((milligrams per litre) to the nth root)
- K f :
-
Overall pseudo-first order rate constant (per hour)
- K R :
-
Redlich–Peterson isotherm constant (litres per gramme)
- K T :
-
Temkin isotherm constant (litres per milligramme)
- K s :
-
Overall pseudo-second order rate constant (grammes per milligramme·hour))
- m :
-
Amount of adsorbent used per litre of solution (grammes per litre)
- M :
-
Amount of solid adsorbent material used (grammes)
- n :
-
Freundlich constant that indicate the intensity of the adsorption
- q d :
-
Dubinin-Radushkevich isotherm constant (milligrammes per gramme)
- R :
-
Universal gas constant 8.314 J/mol K
- R L :
-
Dimensionless separation factor
- R 2 :
-
Correlation coefficient
- T :
-
Absolute temperature (kelvin)
- t :
-
Time (hours)
- V :
-
Volume of adsorbate solution (millilitres)
- X :
-
Amount of adsorbate adsorbed at time (milligrammes)
- β :
-
Exponent in Redlich–Peterson equation
- α :
-
Constant of Bangham’s equation
References
Boving, T. B., & Zhang, W. (2004). Removal of aqueous-phase polynuclear aromatic hydrocarbons using aspen wood fibers. Chemosphere, 54, 839–881.
Cheremisinoff, P. N. (1992). A guide, to underground storage tanks, evaluation, site assessment & remediation. Englewood Cliffs, NJ: Prentice-Hall.
Crisafully, R., Milhome, M. A. L., Cavalcante, R. M., Silveira, E. R., De Keukeleire, D., & Nascimento, R. F. (2008). Removal of some polycyclic aromatic hydrocarbons from petrochemical wastewater using low-cost adsorbents of natural origin. Bioresource Technology, 99, 4515–4519.
Daifullah, A. A. M., & Girgis, B. S. (2003). Impact of surface characteristics of activated carbon on adsorption of BTEX. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 214(1), 181–193.
Garcia, N. M., & Nollet, L. M. L. (2006). BTEX. In L. M. L. Nollet (Ed.), Chromatographic analysis of the environment (3rd ed., pp. 513–554). London: Taylor & Francis Group.
Gitipour, S., BOWERS, M. T., HUFF, W., & BODOCSI, A. (1997). The efficiency of modified bentonite clays for removal of aromatic organics from oily liquid wastes. Spill Science & Technology Bulletin, 4(3), 155–164.
Grupta, V. K., Jain, C. K., Ali, I., Chandra, S., & Agarwal, S. (2002). Removal of lindane and malathion from wastewater using bagasse fly ash—a sugar industry waste. Water Research, 36, 2483–2490.
Janks, J. S., & Cadena, F. (1991). Identification and Properties of Modified Zeolites for the Removal of Benzene, Toluene, and Xylene From Aqueous Solutions. Paper presented at the SPE Annual Technical Conference and Exhibition, Dallas, Texas.
Kumar Dutta, P., Dutta, J., & Tripathi, V. S. (2004). Chitin and chitosan: Chemistry, properties and applications. Journal of Scientific & Industrial Research, 63, 20–31.
Lin, S. H., & Huang, C. Y. (1999). Adsorption of BTEX from aqueous solution by macroreticular resins. Journal of Hazardous Materials, 70(1–2), 21–37.
Lu, C., Su, F., & Hu, S. (2008). Surface modification of carbon nanotubes for enhancing BTEX adsorption from aqueous solutions. Applied Surface Science, 254(21), 7035–7041.
Mackay, A. A., & Gschwend, P. M. (2000). Sorption of monoaromatic hydrocarbons to wood. Environmental Science Technology, 34, 839–845.
Mall, I. D., Srivastava, V. C., Agarwal, N. K., & Mishra, I. M. (2005). Adsorptive removal of malachite green dye from aqueous solution by bagasse fly ash and activated carbon-kinetic study and equilibrium isotherm analyses. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 264, 17–28.
Mckay, G. (Ed.). (1995). Use of adsorbents for the removal of pollutants from wastewater. Boca Raton, FL, USA: CRC Press.
Ravi Kumar, M. N. V. (2000). A review of chitin and chitosan applications. Reactive and Functional Polymers, 46(1), 1–27.
Ravi Kumar, M. N. V., Muzzarelli, R. A. A., Muzzarelli, C., Sashiwa, H., & Domb, A. J. (2004). Chitosan chemistry and pharmaceutical perspectives. Chemical Reviews, 104, 6017–6084.
Sharmasarkar, S. M., Jaynes, W. F., & Vance, G. F. (2000). BTEX Sorption by montmorillonite organo-clays: TMPA, Adam, HDTMA. Water, Air, & Soil Pollution, 119(1–4), 257–273.
Su, F., Lu, C., & Hu, S. (2010). Adsorption of benzene, toluene, ethylbenzene and p-xylene by NaOCl-oxidized carbon nanotubes. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 353(1), 83–91.
Sun, Q. Y., & Yang, L. Z. (2003). The adsorption of basic dyes from aqueous solution on modified peat-resin particle. Water Research, 37, 1535–1544.
Swamy, M. M., Mall, I. D., Prasad, B., & Mishra, I. M. (1997). Removal of phenol by adsorption on coal fly ash and activated carbon. Pollution Research, 16(3), 170–175.
WHO (1996). Ethylbenzene in drinking-water. WHO guidelines for drinking-water quality, Report WHO/SDE/WSH/03.04/26 (2nd ed., Vol. 2). Geneva, Switzerland: World Health Organization.
Zheng, S., Yang, Z., Jo Hyun, D., & Park, Y. H. (2004). Removal of chlorophenols from groundwater by chitosan sorption. Water Research, 38, 2315–2322.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohamed, M., Ouki, S.K. Kinetic and Removal Mechanisms of Ethylbenzene from Contaminated Solutions by Chitin and Chitosan. Water Air Soil Pollut 220, 131–140 (2011). https://doi.org/10.1007/s11270-010-0740-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-010-0740-2