Abstract
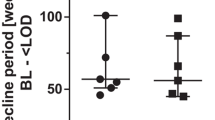
Integrase-strand-transfer inhibitors (INSTIs) are known to rapidly reduce HIV-1 plasma viral load, replication cycles, and new viral integrations, thus potentially limiting viral evolution. Here, we assessed the role of INSTIs on HIV-1 V3 evolution in a cohort of 89 HIV-1-infected individuals starting an INSTI- (N = 41, [dolutegravir: N = 1; elvitegravir: N = 3; raltegravir: N = 37]) or a non-INSTI-based (N = 48) combined antiretroviral therapy (cART), with two plasma RNA V3 genotypic tests available (one before [baseline] and one during cART). V3 sequences were analysed for genetic distance (Tajima-Nei model) and positive selection (dN/dS ratio). Individuals were mainly infected by B subtype (71.9%). Median (interquartile-range, IQR) plasma viral load and CD4 + T cell count at baseline were 4.8 (3.5–5.5) log10 copies/mL and 207 (67–441) cells/mm3, respectively. Genetic distance (median, IQR) between the V3 sequences obtained during cART and those obtained at baseline was 0.04 (0.01–0.07). By considering treatment, genetic distance was significantly lower in INSTI-treated than in non-INSTI-treated individuals (median [IQR]: 0.03[0.01–0.04] vs. 0.05[0.02–0.08], p = 0.026). In line with this, a positive selection (defined as dN/dS ≥ 1) was observed in 36.6% of V3 sequences belonging to the INSTI-treated group and in 56.3% of non-INSTI group (p = 0.05). Multivariable logistic regression confirmed the independent correlation of INSTI-based regimens with a lower probability of both V3 evolution (adjusted odds-ratio: 0.35 [confidence interval (CI) 0.13–0.88], p = 0.027) and positive selection (even if with a trend) (adjusted odds-ratio: 0.46 [CI 0.19–1.11], p = 0.083). Overall, this study suggests a role of INSTI-based regimen in limiting HIV-1 V3 evolution over time. Further studies are required to confirm these findings.

Similar content being viewed by others
References
Waters L, Mandalia S, Randell P, Wildfire A, Gazzard B, Moyle G (2008) The impact of HIV tropism on decreases in CD4 cell count, clinical progression, and subsequent response to a first antiretroviral therapy regimen. Clin Infect Dis 46:1617–1623
Regoes RR, Bonhoeffer S (2005) The HIV coreceptor switch: a population dynamical perspective. Trends Microbiol 13:269–277
Hoffman TL, Doms RW (1999) HIV-1 envelope determinants for cell tropism and chemokine receptor use. Mol Membr Biol 16:57–65
Huang CC, Tang M, Zhang MY, Majeed S, Montabana E, Stanfield RL et al (2005) Structure of a V3-containing HIV-1 gp120 core. Science 310:1025–1028
Vicenzi E, Liò P, Poli G (2013) The puzzling role of CXCR4 in human immunodeficiency virus infection. Theranostics 3:18–25
Koot M, Keet IP, Vos AH, de Goede RE, Roos MT, Coutinho RA, Miedema F, Schellekens PT, Tersmette M (1993) Prognostic value of HIV-1 syncytium-inducing phenotype for rate of CD4 + cell depletion and progression to AIDS. Ann Intern Med 118:681–688
Raymond S, Saliou A, Delobel P, Cazabat M, Pasquier C, Jeanne N, Sauné K, Massip P, Marchou B, Izopet J (2014) Evolution of HIV-1 quasispecies and coreceptor use in cell reservoirs of patients on suppressive antiretroviral therapy. J Antimicrob Chemother 69:2527–2530
Castagna A, Monno L, Carta S, Galli L, Carrara S, Fedele V, Punzi G, Fanti I, Caramello P, Lepri AC, De Luca A, Ceccherini-Silberstein F, Monforte AD (2016) Switch of predicted HIV-1 tropism in treated subjects and its association with disease progression. Medicine 95:e5222
Delobel P, Sandres-Sauné K, Cazabat M, Pasquier C, Marchou B, Massip P, Izopet J (2015) R5 to X4 switch of the predominant HIV-1 population in cellular reservoirs during effective highly active antiretroviral therapy. J Acquir Immune Defic Syndr 38:382–392
Park TE, Mohamed A, Kalabalik J, Sharma R (2015) Review of integrase strand transfer inhibitors for the treatment of human immunodeficiency virus infection. Expert Rev Anti Infect Ther 13:1195–1212
Yingshan H, Mesplède T, Wainberg MA (2017) Investigational HIV integrase inhibitors in phase I and phase II clinical trials. Expert Opin Investig Drugs 26:1207–1213
European AIDS Clinical Society (EACS). EACS Guidelines, Version 9.1. 2018. Available at: http://www.eacsociety.org/files/guidelines_9.0-english.pdf
Department of Health and Human Services (DHHS). Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents (2018) Available at: https://aidsinfo.nih.gov/contentfiles/lvguidelines/adultandadolescentgl.pdf
Haubrich RH, Riddler SA, Ribaudo H, Direnzo G, Klingman KL, Garren KW, Butcher DL, Rooney JF, Havlir DV, Mellors JW (2011) Initial viral decay to assess the relative antiretroviral potency of protease inhibitor-sparing, nonnucleoside reverse transcriptase inhibitor-sparing, and nucleoside reverse transcriptase inhibitor-sparing regimens for first-line therapy of HIV infection. AIDS 25:2269–2278
Lennox JL, DeJesus E, Lazzarin A, Pollard RB, Madruga JV, Berger DS et al (2009) Safety and efficacy of raltegravir based versus efavirenz-based combination therapy in treatment-naïve patients with HIV-1 infection: a multicentre, double-blind randomised controlled trial. Lancet 374:796–806
Sax PE, DeJesus E, Mills A, Zolopa A, Cohen C, Wohl D et al (2012) Co-formulated elvitegravir, cobicistat, emtricitabine, and tenofovir versus co-formulated efavirenz, emtricitabine, and tenofovir for initial treatment of HIV-1 infection: a randomised, double-blind, phase 3 trial, analysis of results after 48 weeks. Lancet 79:2439–2448
DeJesus E, Rockstroh JK, Henry K, Molina JM, Gathe J, Ramanathan S, Wei X, Yale K, Szwarcberg J, White K, Cheng AK, Kearney BP (2012) Co-formulated elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate versus ritonavir boosted atazanavir plus co-formulated emtricitabine and tenofovir disoproxil fumarate for initial treatment of HIV-1 infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet 379:2429–2438
Clotet B, Feinberg J, van Lunzen J, Khuong-Josses MA, Antinori A, Dumitru I, Pokrovskiy V, Fehr J, Ortiz R, Saag M, Harris J, Brennan C, Fujiwara T, Min S (2014) Once-daily dolutegravir versus darunavir plus ritonavir in antiretroviral-naive adults with HIV-1 infection (FLAMINGO): 48 week results from the randomised open-label phase 3b study. Lancet 383:2222–2231
Walmsley SL, Antela A, Clumeck N, Duiculescu D, Eberhard A, Gutiérrez F, Hocqueloux L, Maggiolo F, Sandkovsky U, Granier C, Pappa K, Wynne B, Min S, Nichols G (2013) Dolutegravir plus abacavir lamivudine for the treatment of HIV-1 infection. N Engl J Med 369:1807–1818
Murray JM, Emery S, Kelleher AD, Law M, Chen J, Hazuda DJ, Nguyen BY, Teppler H, Cooper DA (2007) Antiretroviral therapy with the integrase inhibitor raltegravir alters decay kinetics of HIV, significantly reducing the second phase. AIDS 21:2315–2321
Polis MA, Sidorov IA, Yoder C, Jankelevich S, Metcalf J, Mueller BU, Dimitrov MA, Pizzo P, Yarchoan R, Dimitrov DS (2001) Correlation between reduction in plasma HIV-1 RNA concentration 1 week after start of antiretroviral treatment and longer-term efficacy. Lancet 358:1760–1765
Bon I, Calza L, Musumeci G, Longo S, Bertoldi A, D’Urbano V, Gibellini D, Magistrelli E, Viale PL, Re MC (2017) Impact of different antiretroviral strategies on total HIV-DNA level in virologically suppressed HIV-1 infected patients. Curr HIV Res 15:448–455
Malet I, Delelis O, Soulie C, Wirden M, Tchertanov L, Mottaz P, Peytavin G, Katlama C, Mouscadet JF, Calvez V, Marcelin AG (2009) Quasispecies variant dynamics during emergence of resistance to raltegravir in HIV-1-infected patients. J Antimicrob Chemother 63:795–804
Kobayashi M, Yoshinaga T, Seki T, Wakasa-Morimoto C, Brown KW, Ferris R et al (2011) In vitro antiretroviral properties of S/GSK1349572, a next-generation HIV integrase inhibitor. Antimicrob Agents Chemother 55:813–821
Svicher V, D’Arrigo R, Alteri C, Andreoni M, Angarano G, Antinori A et al (2010) Performance of genotypic tropism testing in clinical practice using the enhanced sensitivity version of Trofile as reference assay: results from the OSCAR Study Group. New Microbiol 33:195–206
Tajima F, Nei M (1984) Estimation of evolutionary distance between nucleotide sequences. Mol Biol Evol 1:269–285
Nei M, Gojobori T (1986) Simple methods for estimating the numbers of synonymous and nonsynonymous nucleotide substitutions. Mol Biol Evol 3:418–426
Vandekerckhove LP, Wensing AM, Kaiser R, Brun-Vézinet F, Clotet B, De Luca A et al (2011) European guidelines on the clinical management of HIV-1 tropism testing. Lancet Infect Dis 11:394–407
Svicher V, Alteri C, Artese A, Zhang JM, Costa G, Mercurio F et al (2011) Identification and structural characterization of novel genetic elements in the HIV-1 V3 loop regulating coreceptor usage. Antivir Ther 16:1035–1045
Svicher V, Cento V, Rozera G, Abbate I, Santoro MM, Armenia D et al (2012) The genotypic false positive rate determined by V3 population sequencing can predict the burden of HIV-1 CXCR4-using species detected by pyrosequencing. PLoS ONE 8:e53603
Mesplède T, Moïsi D, Oliveira M, Ibanescu I, Ohnona F, Brenner B, Wainberg MA (2015) Dolutegravir inhibits HIV-1 Env evolution in primary human cells. AIDS 29:659–665
Raffi F, Jaeger H, Quiros-Roldan E, Albrecht H, Belonosova E, Gatell JM, Baril JG, Domingo P, Brennan C, Almond S, Min S (2013) Once-daily dolutegravir versus twice-daily raltegravir in antiretroviral-naive adults with HIV-1 infection (SPRING-2 study): 96 week results from a randomised, double blind, noninferiority trial. Lancet Infect Dis 13:927–935
Cahn P, Pozniak AL, Mingrone H, Shuldyakov A, Brites C, Andrade-Villanueva JF et al (2013) Dolutegravir versus raltegravir in antiretroviral-experienced, integrase-inhibitor-naive adults with HIV: week 48 results from the randomised, double-blind, noninferiority SAILING study. Lancet 382:700–708
Gantner P, Lee GQ, Rey D, Mesplede T, Partisani M, Cheneau C, Beck-Wirth G, Faller JP, Mohseni-Zadeh M, Martinot M, Wainberg MA, Fafi-Kremer S (2018) Dolutegravir reshapes the genetic diversity of HIV-1 reservoirs. J Antimicrob Chemother 73:1045–1053
Acknowledgements
We thank: Massimiliano Bruni and Marzia Romani for data entry; Andrea Biddittu and Alberto Giannetti for data management. Moreover, we wish to thank all the clinicians and virologists throughout Italy who contribute with their work to develop, expand and maintain the ARCA database.
Funding
This work was financially supported by the European Commission Framework 7 Programme (CHAIN, the Collaborative HIV and Anti-HIV Drug Resistance Network, Integrated Project no. 223131), the Italian Ministry of Health (Progetto Ricerca Corrente 2016, line n. 2, project n.2, sub-project n.2d), the Italian Ministry of Education, University and Research (MIUR) (Bandiera InterOmics Protocollo PB05 1°) and an unrestricted grant from AVIRALIA foundation.
Author information
Authors and Affiliations
Contributions
C.A. conceived the presented idea and wrote the manuscript. R.S. developed and performed the statistical analyses. D.A. and M.M.S. contributed to perform and interpret the statistical analyses. A.B., C.G., and I.V. determined the V3 sequences. G.F., C.M.M., C.C., A.C., B.B., M.A., and A.A. provided samples. R.S. and M.M.S. contributed to the writing of the manuscript. M.Z., V.S., F.C.S. and C.F.P. contributed to the interpretation of the results and revised the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests that might be perceived to influence the results and/or discussion reported in this paper. However, Francesca Ceccherini-Silberstein reports personal fees from Gilead Sciences, Bristol-Myers Squibb, Abbvie, Roche Diagnostics, Janssen-Cilag, Abbott Molecular, ViiV Healthcare; grants and personal fees from Merck Sharp & Dohme; grants from Italian Ministry of Education, University and Research (MIUR). Carlo Federico Perno reports grants from Italian Ministry of Instruction, University and Research (MIUR), and from Aviralia Foundation; personal fees from Gilead Sciences, Abbvie, Roche Diagnostics, Janssen-Cilag, Abbott Molecular, and grants and personal fees from Bristol-Myers Squibb, Merck Sharp & Dohme, and ViiV Healthcare. All other authors have nothing to declare.
Informed consent
This study was conducted on data collected for clinical purposes. All data used in the study were previously anonymized, according to the requirements set by Italian Data Protection Code (leg. decree 196/2003) and by the General authorizations issued by the Data Protection Authority. Written informed consent for medical procedures/interventions performed for routine treatment purposes was collected for each patient included in the ARCA database or from other clinical centers involved in the study, in accordance with the ethics standards of the committee on human experimentation and the Helsinki Declaration (1983 revision).
Additional information
Edited by Dr. Wolfram Gerlich.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alteri, C., Scutari, R., Bertoli, A. et al. Integrase strand transfer inhibitor-based regimen is related with a limited HIV-1 V3 loop evolution in clinical practice. Virus Genes 55, 290–297 (2019). https://doi.org/10.1007/s11262-019-01649-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-019-01649-z