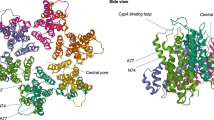
Abstract
The HIV-1 pre-integration phase and the subsequent integration of viral genome to the host of nuclear chromosomes are not well analyzed so far. Many studies are discussing the question of pre- and post-nuclear viral entry which is to support the assumption that HIV-1 integrase (IN) is maintained in the volume of intact conical structure’s capsids through HIV entry. The aim of the current study is to identify the prevalence of capsid’s (CA) signatures among drug-naïve and antiretroviral (ARV)-treated patients in a cohort of 827 HIV-1 B-subtype-infected individuals, and subsequently the relationship between IN and CA amino acid’s changes was evaluated. These analyses suggest a conceivable co-evolution of IN–CA sequences, especially in relation to steps of nuclear viral entry. The frequency of mutations was calculated, and statistically has been compared between treatment-naïve and ARV-treated patients. The binomial correlation coefficient was used to assess covariation among CA and IN mutations; then, the average linkage hierarchical agglomerative clustering was performed. The results show a detailed conservation of HIV-1 CA protein both in drug-naïve and in ARV-treated patients. Moreover, the specific CA substitutions are significantly associated with different IN signatures at the amino acid level and the topology of the dendrogram has revealed the existence of two strong sub-clusters associated with hypothetical different mutational pathways. The in vitro and in vivo studies are necessary to exclude the hypothetical statistical false positive results and in order to confirm that some CA amino acid signatures are going to establish specific and precise implication in the HIV life cycle.



Similar content being viewed by others
References
T. Schaller, K.E. Ocwieja, J. Rasaiyaah, A.J. Price, T.L. Brady, S.L. Roth, S. Hue, A.J. Fletcher, K. Lee, V.N. KewalRamani, M. Noursadeghi, R.G. Jenner, L.C. James, F.D. Bushman, G.J. Towers, HIV-1 capsid-cyclophilin interactions determine nuclear import pathway, integration targeting and replication efficiency. PLoS Pathog. 7, e1002439 (2011)
A. Fassati, Multiple roles of the capsid protein in the early steps of HIV-1 infection. Virus Res. 170, 15–24 (2012)
N.J. Arhel, S. Souquere-Besse, S. Munier, P. Souque, S. Guadagnini, S. Rutherford, M.C. Prevost, T.D. Allen, P. Charneau, HIV-1 DNA Flap formation promotes uncoating of the pre-integration complex at the nuclear pore. EMBO J. 26, 3025–3037 (2007)
N. Arhel, Revisiting HIV-1 uncoating. Retrovirology. 7, 96 (2010)
K. Lee, Z. Ambrose, T.D. Martin, I. Oztop, A. Mulky, J.G. Julias, N. Vandegraaff, J.G. Baumann, R. Wang, W. Yuen, T. Takemura, K. Shelton, I. Taniuchi, Y. Li, J. Sodroski, D.R. Littman, J.M. Coffin, S.H. Hughes, D. Unutmaz, A. Engelman, V.N. Kewalramani, Flexible use of nuclear import pathways by HIV-1. Cell Host Microbe. 7, 221–233 (2010)
L. Krishnan, K.A. Matreyek, I. Oztop, K. Lee, C.H. Tipper, X. Li, M.J. Dar, V.N. Kewalramani, A. Engelman, The requirement for cellular transportin 3 (TNPO3 or TRN-SR2) during infection maps to human immunodeficiency virus type 1 capsid and not integrase. J. Virol. 84, 397–406 (2010)
K.A. Matreyek, A. Engelman, The requirement for nucleoporin NUP153 during human immunodeficiency virus type 1 infection is determined by the viral capsid. J. Virol. 85, 7818–7827 (2011)
M. Lelek, F. Di Nunzio, R. Henriques, P. Charneau, N. Arhel, C. Zimmer, Superresolution imaging of HIV in infected cells with FlAsH-PALM. Proc. Natl. Acad. Sci. USA 109, 8564–8569 (2012)
L. Zhou, E. Sokolskaja, C. Jolly, W. James, S.A. Cowley, A. Fassati, Transportin 3 promotes a nuclear maturation step required for efficient HIV-1 integration. PLoS Pathog. 7, e1002194 (2011)
K.D. Jayappa, Z. Ao, X. Yao, The HIV-1 passage from cytoplasm to nucleus: the process involving a complex exchange between the components of HIV-1 and cellular machinery to access nucleus and successful integration. Int. J. Biochem. Mol. Biol. 3, 70–85 (2012)
S. Dimonte, M. Babakir-Mina, S. Aquaro, C.F. Perno, Specific VpU codon changes were significantly associated with gp120 V3 tropic signatures in HIV-1 B-subtype. Virol. Sin. 27, 360–368 (2012)
S. Dimonte, V. Svicher, R. Salpini, F. Ceccherini-Silberstein, C.F. Perno, M. Babakir-Mina, HIV-2 A-subtype gp125C2-V3-C3 mutations and their association with CCR5 and CXCR4 tropism. Arch. Virol. 156, 1943–1951 (2011)
K. Tamura, D. Peterson, N. Peterson, G. Stecher, M. Nei, S. Kumar, MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739 (2011)
Y. Benjamini, Y. Hochberg, Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B. 57, 289–300 (1995)
S. Dimonte, M. Babakir-Mina, F. Mercurio, D. Di Pinto, F. Ceccherini-Silberstein, V. Svicher, C.F. Perno, Selected amino acid changes in HIV-1 subtype-C gp41 are associated with specific gp120(V3) signatures in the regulation of co-receptor usage. Virus Res. 168, 73–83 (2012)
S. Dimonte, F. Mercurio, V. Svicher, R. D’Arrigo, C.F. Perno, F. Ceccherini-Silberstein, Selected amino acid mutations in HIV-1 B subtype gp41 are associated with specific gp120V3 signatures in the regulation of co-receptor usage. Retrovirology 8, 33 (2011)
S. Dimonte, M. Babakir-Mina, S. Aquaro, C.F. Perno, Natural polymorphisms of HIV-1 subtype-C integrase coding region in a large group of ARV-naive infected individuals. Infection 41, 1097–1102 (2013)
F. Ceccherini-Silberstein, I. Malet, R. D’Arrigo, A. Antinori, A.G. Marcelin, C.F. Perno, Characterization and structural analysis of HIV-1 integrase conservation. AIDS Rev. 11, 17–29 (2009)
J. Nouhin, T. Donchai, K.T. Hoang, S. Ken, J. Kamkorn, T. Tran, A. Ayouba, M. Peeters, M.L. Chaix, T.X. Lien, E. Nerrienet, N. Ngo-Giang-Huong, Natural polymorphisms of HIV-1 CRF01_AE integrase coding region in ARV-naive individuals in Cambodia, Thailand and Vietnam: an ANRS AC12 working group study. Infect. Genet. Evol. 11, 38–43 (2011)
F. Ceccherini-Silberstein, I. Malet, L. Fabeni, S. Dimonte, V. Svicher, R. D’Arrigo, A. Artese, G. Costa, S. Bono, S. Alcaro, A. d’Arminio, Monforte, C. Katlama, V. Calvez, A. Antinori, A.G. Marcelin, and C.F. Perno, Specific HIV-1 integrase polymorphisms change their prevalence in untreated versus antiretroviral-treated HIV-1-infected patients, all naive to integrase inhibitors. J. Antimicrob. Chemother. 65, 2305–2318 (2010)
A. Iamarino, F.L. de Melo, C.T. Braconi, P.M. Zanotto, BF integrase genes of HIV-1 circulating in Sao Paulo, Brazil, with a recurrent recombination region. PLoS One 7, e34324 (2012)
G. Zhao, J.R. Perilla, E.L. Yufenyuy, X. Meng, B. Chen, J. Ning, J. Ahn, A.M. Gronenborn, K. Schulten, C. Aiken, P. Zhang, Mature HIV-1 capsid structure by cryo-electron microscopy and all-atom molecular dynamics. Nature 497, 643–646 (2013)
C. Berthet-Colominas, S. Monaco, A. Novelli, G. Sibai, F. Mallet, S. Cusack, Head-to-tail dimers and interdomain flexibility revealed by the crystal structure of HIV-1 capsid protein (p24) complexed with a monoclonal antibody Fab. EMBO J. 18, 1124–1136 (1999)
P.A. Gallay, R.G. Ptak, M.D. Bobardt, J.M. Dumont, G. Vuagniaux, B. Rosenwirth, Correlation of naturally occurring HIV-1 resistance to DEB025 with capsid amino acid polymorphisms. Viruses 5, 981–997 (2013)
H. Gatanaga, D. Das, Y. Suzuki, D.D. Yeh, K.A. Hussain, A.K. Ghosh, H. Mitsuya, Altered HIV-1 Gag protein interactions with cyclophilin A (CypA) on the acquisition of H219Q and H219P substitutions in the CypA binding loop. J. Biol. Chem. 281, 1241–1250 (2006)
M. Veillette, K. Bichel, P. Pawlica, S.M. Freund, M.B. Plourde, Q.T. Pham, C. Reyes-Moreno, L.C. James, L. Berthoux, The V86 M mutation in HIV-1 capsid confers resistance to TRIM5alpha by abrogation of cyclophilin A-dependent restriction and enhancement of viral nuclear import. Retrovirology 10, 25 (2013)
B. Li, A.D. Gladden, M. Altfeld, J.M. Kaldor, D.A. Cooper, A.D. Kelleher, T.M. Allen, Rapid reversion of sequence polymorphisms dominates early human immunodeficiency virus type 1 evolution. J. Virol. 81, 193–201 (2007)
S. Matsuoka, E. Dam, D. Lecossier, F. Clavel, A.J. Hance, Modulation of HIV-1 infectivity and cyclophilin A-dependence by Gag sequence and target cell type. Retrovirology. 6, 21 (2009)
B.K. Ganser-Pornillos, U.K. von Schwedler, K.M. Stray, C. Aiken, W.I. Sundquist, Assembly properties of the human immunodeficiency virus type 1 CA protein. J. Virol. 78, 2545–2552 (2004)
B.N. Kelly, S. Kyere, I. Kinde, C. Tang, B.R. Howard, H. Robinson, W.I. Sundquist, M.F. Summers, C.P. Hill, Structure of the antiviral assembly inhibitor CAP-1 complex with the HIV-1 CA protein. J. Mol. Biol. 373, 355–366 (2007)
O. Pornillos, B.K. Ganser-Pornillos, B.N. Kelly, Y. Hua, F.G. Whitby, C.D. Stout, W.I. Sundquist, C.P. Hill, M. Yeager, X-ray structures of the hexameric building block of the HIV capsid. Cell 137, 1282–1292 (2009)
Y.F. Chang, S.M. Wang, K.J. Huang, C.T. Wang, Mutations in capsid major homology region affect assembly and membrane affinity of HIV-1 Gag. J. Mol. Biol. 370, 585–597 (2007)
P. Coric, S. Turcaud, F. Souquet, L. Briant, B. Gay, J. Royer, N. Chazal, S. Bouaziz, Synthesis and biological evaluation of a new derivative of bevirimat that targets the Gag CA-SP1 cleavage site. Eur. J. Med. Chem. 62, 453–465 (2013)
W.S. Blair, J. Cao, J. Fok-Seang, P. Griffin, J. Isaacson, R.L. Jackson, E. Murray, A.K. Patick, Q. Peng, M. Perros, C. Pickford, H. Wu, S.L. Butler, New small-molecule inhibitor class targeting human immunodeficiency virus type 1 virion maturation. Antimicrob. Agents Chemother. 53, 5080–5087 (2009)
A.N. Galkin, E.Y. Gagarina, N.S. Lukyanova, T.V. Danilova, Full-length genomic sequencing and analysis of four HIV type 1 subtype B isolates circulating in the territory of Russia. AIDS Res. Hum. Retroviruses 22, 1192–1197 (2006)
Y.E. Wang, B. Li, J.M. Carlson, H. Streeck, A.D. Gladden, R. Goodman, A. Schneidewind, K.A. Power, I. Toth, N. Frahm, G. Alter, C. Brander, M. Carrington, B.D. Walker, M. Altfeld, D. Heckerman, T.M. Allen, Protective HLA class I alleles that restrict acute-phase CD8 + T-cell responses are associated with viral escape mutations located in highly conserved regions of human immunodeficiency virus type 1. J. Virol. 83, 1845–1855 (2009)
R. Yang, J. Shi, I.J. Byeon, J. Ahn, J.H. Sheehan, J. Meiler, A.M. Gronenborn, C. Aiken, Second-site suppressors of HIV-1 capsid mutations: restoration of intracellular activities without correction of intrinsic capsid stability defects. Retrovirology 9, 30 (2012)
T.A. Wilkinson, K. Januszyk, M.L. Phillips, S.S. Tekeste, M. Zhang, J.T. Miller, S.F. Le Grice, R.T. Clubb, S.A. Chow, Identifying and characterizing a functional HIV-1 reverse transcriptase-binding site on integrase. J. Biol. Chem. 284, 7931–7939 (2009)
S.A. Knoepfel, B. Berkhout, On the role of four small hairpins in the HIV-1 RNA genome. RNA Biol. 10(4), 540–552 (2013)
V. D’Souza, M.F. Summers, How retroviruses select their genomes. Nat. Rev. Microbiol. 3, 643–655 (2005)
A. Borodavka, R. Tuma, P.G. Stockley, Evidence that viral RNAs have evolved for efficient, two-stage packaging. Proc. Natl. Acad. Sci. USA 109, 15769–15774 (2012)
D.H. Bunka, S.W. Lane, C.L. Lane, E.C. Dykeman, R.J. Ford, A.M. Barker, R. Twarock, S.E. Phillips, P.G. Stockley, Degenerate RNA packaging signals in the genome of Satellite Tobacco Necrosis Virus: implications for the assembly of a T=1 capsid. J. Mol. Biol. 413, 51–65 (2011)
J.B. Bancroft, G.J. Hills, R. Markham, A study of the self-assembly process in a small spherical virus. Formation of organized structures from protein subunits in vitro. Virology 31, 354–379 (1967)
Acknowledgments
Many thanks to Stefania Carmen Spinazzola and Bahra Rzgar Abdullah for their excellent technical assistance and Dana Hameed Mahmood and Prof. Domenico Sturino for punctuation and grammatical revision.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dimonte, S., Babakir-Mina, M. & Aquaro, S. HIV-1 B-subtype capsid protein: a characterization of amino acid’s conservation and its significant association with integrase signatures. Virus Genes 48, 429–437 (2014). https://doi.org/10.1007/s11262-014-1039-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-014-1039-y