Abstract
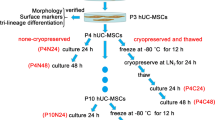
Antimicrobial peptides (AMPs) are naturally produced by all living organisms at a constitutive rate. They represent the first line of active defence systems against invading microorganisms, helping in innate immunity. Besides their therapeutic applications, great attention has also been given to the mesenchymal stem cells (MSCs) due to their antimicrobial activities. The study aimed to observe the mRNA expression profile of few antimicrobial peptides (AMPs) in canine MSCs during standard in vitro culture. MSCs were isolated from canine umbilical cord tissue, propagated and characterized by morphology, surface markers and tri-lineage differentiation capability. The mRNA expression of eleven commonly known antimicrobial peptides was checked by Reverse Transcriptase PCR. It has been found for the first time that canine MSCs naturally express the mRNAs of AMPs like C-X-C motif chemokine ligand 8 (CXCL8), Elafin (PI3), Hepcidin (HAMP), Lipocalin 2 (LCN2) and Secretory leukocyte protease inhibitor (SLPI). However, their expressions at protein level and, relation with antimicrobial effect of canine MSCs need to be explored.

Similar content being viewed by others
References
Alcayaga-Miranda F, Cuenca J, Khoury M (2017) Antimicrobial activity of mesenchymal stem cells: current status and new perspectives of antimicrobial peptide-based therapies. Front Immunol 8:339. https://doi.org/10.3389/fimmu.2017.00339
Bujňáková D, Čuvalová A, Čížek M, Humenik F, Salzet M, Čížková D (2020) Canine bone marrow mesenchymal stem cell conditioned media affect bacterial growth, biofilm-associated Staphylococcus aureus and AHL-dependent quorum sensing. Microorganisms 8(10):1478. https://doi.org/10.3390/microorganisms8101478
Dahl SL, Woodworth JS, Lerche CJ, Cramer EP, Nielsen PR, Moser C, Thomsen AR, Borregaard N, Cowland JB (2018) Lipocalin-2 functions as inhibitor of innate resistance to Mycobacterium tuberculosis. Front Immunol 9:2717. https://doi.org/10.3389/fimmu.2018.02717
Das K, Madhusoodan AP, Mili B, Kumar A, Saxena AC, Kumar K, Sarkar M, Singh P, Srivastava S, Bag S (2017) Functionalized carbon nanotubes as suitable scaffold materials for proliferation and differentiation of canine mesenchymal stem cells. Int J Nanomedicine 12:3235–3252. https://doi.org/10.2147/IJN.S122945
de Oliveira S, Reyes-Aldasoro CC, Candel S, Renshaw SA, Mulero V, Calado  (2013) Cxcl8 (IL-8) mediates neutrophil recruitment and behavior in the zebrafish inflammatory response. J Immunol 190(8):4349–4359. https://doi.org/10.4049/jimmunol.1203266
Doumas S, Kolokotronis A, Stefanopoulos P (2005) Anti-inflammatory and antimicrobial roles of secretory leukocyte protease inhibitor. Infect Immun 73(3):1271–1274. https://doi.org/10.1128/IAI.73.3.1271-1274.2005
El Gendy F, EL-Hawy M, El Shennawy N (2019) Role of hepcidin in iron metabolism and pathophysiology of different types of anemia. Menoufia Med J 32(3):818. https://doi.org/10.4103/mmj.mmj_20_18
Hancock REW, Nijnik A, Philpott DJ (2012) Modulating immunity as a therapy for bacterial infections. Nat Rev Microbiol 10(4):243–254. https://doi.org/10.1038/nrmicro2745
Harman RM, Yang S, He MK, Van de Walle GR (2017) Antimicrobial peptides secreted by equine mesenchymal stromal cells inhibit the growth of bacteria commonly found in skin wounds. Stem Cell Res Ther 8(1):157. https://doi.org/10.1186/s13287-017-0610-6
Kol A, Foutouhi S, Walker NJ, Kong NT, Weimer BC, Borjesson DL (2014) Gastrointestinal microbes interact with canine adipose-derived mesenchymal stem cells in vitro and enhance immunomodulatory functions. Stem Cells and Development 23(16):1831–1843. https://doi.org/10.1089/scd.2014.0128
Krasnodembskaya A, Song Y, Fang X, Gupta N, Serikov V, Lee JW, Matthay MA (2010) Antibacterial effect of human mesenchymal stem cells is mediated in part from secretion of the antimicrobial peptide LL-37. Stem Cells 28(12):2229–2238. https://doi.org/10.1002/stem.544
Lohajaroensub R, Sawangmake C, Rodkhum C, Tuntivanich N (2022) Expression of antimicrobial peptide genes in the canine amniotic membrane. Vet Sci 9(5):200. https://doi.org/10.3390/vetsci9050200
Meisel R, Brockers S, Heseler K, Degistirici O, Bülle H, Woite C, Stuhlsatz S, Schwippert W, Jäger M, Sorg R, Henschler R, Seissler J, Dilloo D, Däubener W (2011) Human but not murine multipotent mesenchymal stromal cells exhibit broad-spectrum antimicrobial effector function mediated by indoleamine 2,3-dioxygenase. Leukemia 25(4):648–654. https://doi.org/10.1038/leu.2010.310
Shaw L, Wiedow O (2011) Therapeutic potential of human elafin. Biochem Soc Trans 39(5):1450–1454. https://doi.org/10.1042/BST0391450
Funding
The work was funded primarily by the ICAR-Indian Veterinary Research Institute [Grant No. F.7 − 6 /Kolkata/S Bag/2020-21/JD(R)], and in part Department of Science & Technology and Biotechnology, Govt. of West Bengal [Memo No. 262(Sanc.)/ ST/P/S&T/17G-12/2018].
Author information
Authors and Affiliations
Contributions
Camelia Manna: Experimental design, investigation and methodology; Kinsuk Das: Visualization, validation and writing original draft; Durgadas Mandal, Dipak Banerjee, Joydip Mukherjee: Investigation and formal analysis; Indrajit Ganguly: Data curation; Shymal Naskar: Review and editing; Sadhan Bag: Conceptualization, supervision and project administration.
Corresponding author
Ethics declarations
Ethical approval
Umbilical cords were collected after cesarean sections performed at the Veterinary Clinical Complex of West Bengal University of Animal & Fishery Sciences with the approval of Institutional Animal Ethics Committee vide no. 763/GO/Re/SL/03/CPCSEA/17/2021-22.
Consent to participate
All authors read the manuscript and approved their participation.
Consent for publication
All authors approved the publication of the manuscript.
Conflicts to interest
The authors declare that there are no conflicts of interest.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Manna, C., Das, K., Mandal, D. et al. Canine umbilical cord tissue derived mesenchymal stem cells naturally express mRNAs of some antimicrobial peptides. Vet Res Commun 47, 2229–2233 (2023). https://doi.org/10.1007/s11259-023-10098-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10098-x