Abstract
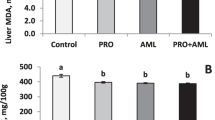
To investigate the effects of niacin on rumen fermentation, rumen epithelial antioxidant activity, and rumen epithelial cell apoptosis on high concentrate (HC) diets, nine male Hu sheep were randomly fed one of three diets: low concentrate diet (LC; concentrate: forage (C:F) = 20:80, high concentrate diet (HC; C:F = 80:20), and HCN diet (HC diet + niacin at 800 mg/kg diet air-dry matter). Compared with the LC group, the HC group had a lower rumen pH, higher volatile fatty acids and lactic acid in the rumen, reduced activity of antioxidant enzymes and total antioxidant capacity, and increased malondialdehyde content in the rumen epithelium (P < 0.05). Rumen epithelial papilla morphology was decreased, and apoptosis-related indicators and serum inflammatory cytokines were increased in the HC group over the LC group (P < 0.05). Compared with the HC diet, the HCN diet increased rumen pH, rumen epithelium antioxidant capacity, and rumen epithelial papilla morphology, decreased rumen lactate content, serum inflammatory cytokines, and apoptosis-related indicators (P < 0.05). Therefore, adding 800 mg/kg niacin helped protect against rumen epithelial damage by avoiding drastic changes in the rumen environment and improved rumen epithelial antioxidant capacity to inhibit rumen epithelial cell apoptosis in sheep on a HC diet.


Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are not publicly available due to all the results in the form of means and statistics are presented in this paper, but are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Allen MS, Bradford BJ, Oba M (2009) Board invited review: the hepatic oxidation theory of the control of feed intake and its application to ruminants. J Anim Sci 87:3317–3334. https://doi.org/10.2527/jas.2009-1779
Arroyo JM, Hosseini A, Zhou Z, Alharthi A, Trevisi E, Osorio JS, Loor JJ (2017) Reticulo-rumen mass, epithelium gene expression, and systemic biomarkers of metabolism and inflammation in Holstein dairy cows fed a high-energy diet. J Dairy Sci 100:9352–9360. https://doi.org/10.3168/jds.2017-12866
Bergmann A (2007) Autophagy and cell death: no longer at odds. Cell 131:1032–1034. https://doi.org/10.1016/j.cell.2007.11.027
Bühler S, Frahm J, Liermann W, Tienken R, Kersten S, Meyer U, Huber K, Dänicke S (2018) Effects of energy supply and nicotinic acid supplementation on phagocytosis and ROS production of blood immune cells of periparturient primi- and pluriparous dairy cows. Res Vet Sci 116:62–71. https://doi.org/10.1016/j.rvsc.2017.09.012
Chen J, Cui X, Zacharek A, Ding GL, Shehadah A, Jiang Q, Lu M, Chopp M (2009) Niaspan treatment increases tumor necrosis factor-alpha-converting enzyme and promotes arteriogenesis after stroke. J Cereb Blood Flow Metab 29:911–920. https://doi.org/10.1038/jcbfm.2009.11
Dai H, Ma N, Chang G, Aabdin ZU, Shen X (2020) Long-term high-concentrate diet feeding induces apoptosis of rumen epithelial cells and inflammation of rumen epithelium in dairy cows. Anim Biotechnol 3:1–8. https://doi.org/10.1080/10495398.2020.1806073
Dong H, Wang S, Jia Y, Ni Y, Zhang Y, Zhuang S, Shen X, Zhao R (2013) Long-term effects of subacute ruminal acidosis (SARA) on milk quality and hepatic gene expression in lactating goats fed a high-concentrate diet. PLoS One 8(12):e82850. https://doi.org/10.1371/journal.pone.0082850
Freiberg RA, Spencer DM, Choate KA, Peng PD, Schreiber SL, Crabtree GR, Khavari PA (1996) Specific triggering of the Fas signal transduction pathway in normal human keratinocytes. J Biol Chem 271:31666–31669. https://doi.org/10.1074/jbc.271.49.31666
Ganji SH, Kashyap ML, Kamanna VS (2015) Niacin inhibits fat accumulation, oxidative stress, and inflammatory cytokine IL-8 in cultured hepatocytes: impact on non-alcoholic fatty liver disease. Metabolism 64:982–990. https://doi.org/10.1016/j.metabol.2015.05.002
Goad DW, Goad CL, Nagaraja TG (1998) Ruminal microbial and fermentative changes associated with experimentally induced subacute acidosis in steers. J Anim Sci 76:234–241. https://doi.org/10.2527/1998.761234x
Gozho GN, Plaizier JC, Krause DO, Kennedy AD, Wittenberg KM (2005) Subacute ruminal acidosis induces ruminal lipopolysaccharide endotoxin release and triggers an inflammatory response. J Dairy Sci 88:1399–1403. https://doi.org/10.3168/jds.S0022-0302(05)72807-1
Gui H, Shen Z (2016) Concentrate diet modulation of ruminal genes involved in cell proliferation and apoptosis is related to combined effects of short-chain fatty acid and pH in rumen of goats. J Dairy Sci 99:6627–6638. https://doi.org/10.3168/jds.2015-10446
Günther C, Neumann H, Neurath MF, Becker C (2013) Apoptosis, necrosis and necroptosis: cell death regulation in the intestinal epithelium. Gut 62:1062–1071. https://doi.org/10.1136/gutjnl-2011-301364
Hodge S, Novembre FJ, Whetter L, Gelbard HA, Dewhurst S (1998) Induction of fas ligand expression by an acutely lethal simian immunodeficiency virus, SIVsmmPBj14. Virology 252:354–363. https://doi.org/10.1006/viro.1998.9477
Hu HL (2008) Study on the nutritional and physiological mechanism of subacute ruminal acidosis in dairy goats [dissertation]. Hohhot (IM): Inner Mongolia Agricultural University
Hua JL, Guo L, Fu JW, Yu FY (2018) Effect of ratios of peanut straw and corn silage on rumen fermentation characteristics of Hu sheep. J Northeast Agric Univ 49:29–35
Ilkhani F, Hosseini B, Saedisomeolia A (2016) Niacin and oxidative stress: a mini-review. J Nutri Med Diet Care 2:014
John LJ, Fromm M, Schulzke J (2011) Epithelial barriers in intestinal inflammation. Antioxid Redox Sign 15:1255–1270. https://doi.org/10.1089/ars.2011.3892
Kennedy DO (2016) B vitamins and the brain: mechanisms, dose and efficacy—a review. Nutrients 8:68. https://doi.org/10.3390/nu8020068
Khafipour E, Krause DO, Plaizier JC (2009) A grain-based subacute ruminal acidosis challenge causes translocation of lipopolysaccharide and triggers inflammation. J Dairy Sci 92:1060–1070. https://doi.org/10.3168/jds.2008-1389
Li W, Gelsinger S, Edwards A, Riehle C, Koch D (2019) Transcriptome analysis of rumen epithelium and meta-transcriptome analysis of rumen epimural microbial community in young calves with feed induced acidosis. Sci Rep-UK 9:4744
Li Y, Shang H, Zhao X, Qu M, Peng T, Guo B, Hu Y, Song X (2021) Radix Puerarin extract (Puerarin) could improve meat quality of heat-stressed beef cattle through changing muscle antioxidant ability and fiber characteristics. Front Vet Sci 7:615086. https://doi.org/10.3389/fvets.2020.615086
Luo D, Gao Y, Lu Y, Qu M, Xiong X, Xu L, Zhao X, Pan K, Ouyang K (2017) Niacin alters the ruminal microbial composition of cattle under high-concentrate condition. Anim Nutr 3:92–97
Luo D, Gao Y, Lu Y, Zhang Q, Qu M, Xiong X, Xu L, Zhao X, Pan K, Ouyang K (2019a) Niacin supplementation improves growth performance and nutrient utilisation in Chinese Jinjiang cattle. Ital J Anim Sci 18:57–62. https://doi.org/10.1080/1828051X.2018.1480426
Luo D, Peng Z, Yang L, Qu M, Xiong X, Xu L, Zhao X, Pan K, Ouyang K (2019b) Niacin protects against butyrate-induced apoptosis in rumen epithelial cells. Oxidative Med Cell Longev 3:1–8. https://doi.org/10.1155/2019/2179738
Mao SY, Huo WJ, Zhu WY (2015) Microbiome-metabolome analysis reveals unhealthy alterations in the composition and metabolism of ruminal microbiota with increasing dietary grain in a goat model. Environ Microbiol 18:525–541. https://doi.org/10.1111/1462-2920.12724
Martínez-Alfaro M, Alcaraz-Contreras Y, Cárabez-Trejo A, Leo-Amador GE (2011) Oxidative stress effects of thinner inhalation. J Occup Environ Med 15:87–92. https://doi.org/10.4103/0019-5278.93195
Mccann JC, Luan S, Cardoso FC, Derakhshani H, Khafipour E, Loor JJ (2016) Induction of subacute ruminal acidosis affects the ruminal microbiome and epithelium. Front Microbiol 7:701. https://doi.org/10.3389/fmicb.2016.00701
Nagaraja TG, Titgemeyer EC (2007) Ruminal acidosis in beef cattle: the current microbiological and nutritional outlook. J Dairy Sci 90:E17–E38. https://doi.org/10.3168/jds.2006-478
Oba M, Allen MS (2000) Effects of brown midrib 3 mutation in corn silage on productivity of dairy cows fed two concentrations of dietary neutral detergent fiber: 3. Digestibility and microbial efficiency. J Dairy Sci 83:1350–1358. https://doi.org/10.3168/jds.S0022-0302(00)75001-6
Plaizier JC, Khafipour E, Li S, Gozho GN, Krause D (2012) Subacute ruminal acidosis (SARA), endotoxins and health consequences. Anim Feed Sci Tech 172:9–21. https://doi.org/10.1016/j.anifeedsci.2011.12.004
Raj L, Ide T, Gurkar AU et al (2011) Selective killing of cancer cells by a small molecule targeting the stress response to ROS. Nature 475:231–234. https://doi.org/10.1038/nature10789
Sun YY, Cheng M, Xu M, Song LW, Gao M, Hu HL (2018) The effects of subacute ruminal acidosis on rumen epithelium barrier function in dairy goats. Small Ruminant Res 1-7. https://doi.org/10.1016/j.smallrumres.2018.09.017
Suo XJ, Zhang N, Yang QP, Tao H, Xiong Q, Li XF, Chen MX (2019) Effects of peanut vine instead of alfalfa in granulated total mixed ration on growth performance, rumen fermentation and serum biochemical indexes of Hubei black-head goats. Chin J Anim Nutr 31:865–873
Tang K, Sham H, Hui E, Kirkland JB (2008) Niacin deficiency causes oxidative stress in rat bone marrow cells but not through decreased NADPH or glutathione status. J Nutr Biochem 19:746–753. https://doi.org/10.1016/j.jnutbio.2007.10.003
Terpilowska S, Siwicki AK (2019) Cell cycle and transmembrane mitochondrial potential analysis after treatment with chromium(iii), iron(iii), molybdenum(iii) or nickel(ii) and their mixtures. Toxicol Res-UK 8:188–195. https://doi.org/10.1039/c8tx00233a
Thakur VS, Gupta K, Gupta S (2012) Green tea polyphenols increase p53 transcriptional activity and acetylation by suppressing class I histone deacetylases. Int J Oncol 41:353–361. https://doi.org/10.3892/ijo.2012.1449
Walker PR, Saas P, Dietrich P (1998) Tumor expression of Fas ligand (CD95L) and the consequences. Curr Opin in Immunol 10:564–572. https://doi.org/10.1016/S0952-7915(98)80225-2
Wang J, Zhang H, Zhang T, Zhang R, Liu R, Chen Y (2015) Molecular mechanism on cadmium-induced activity changes of catalase and superoxide dismutase. Int J Biol Macromol 77:59–67. https://doi.org/10.1016/j.ijbiomac.2015.02.037
Wang Y, Tian Z, Shi H, Zhong J, Ren G, Li Y, Liu Y, Wu Q, Wang J, Zhang Z, Ding K, Yu Z, Zhao Z (2017) Nutritional characteristics and muscle fiber histological characters of muscle from Hu sheep. Chin J Anim Nutr 29:2867–2874
Yang Y, Qu MR, Ouyang KH, Zhao XH, Yi ZH, Song XZ (2013) Effects of nicotinic acid on lactate and volatile fatty acid concentrations, and related enzyme activities in rumen of Jinjiang cattle. Chin J Anim Nutr 25:1610–16166
Zhang L, Yue HY, Wu SG, Xu L, Zhang HJ, Yan HJ, Cao YL, Gong YS, Qi GH (2010) Transport stress in broilers. II. Superoxide production, adenosine phosphate concentrations, and mRNA levels of avian uncoupling protein, avian adenine nucleotide translocator, and avian peroxisome proliferator-activated receptor-γ coactivator-1α in skeletal muscles. Poultry Sci 89:393–400. https://doi.org/10.3382/ps.2009-00281
Zhang H, Liu Y, Lao M, Ma Z, Yi X (2011) Puerarin protects Alzheimer's disease neuronal cybrids from oxidant-stress induced apoptosis by inhibiting pro-death signaling pathways. Exp Gerontol 46:30–37. https://doi.org/10.1016/j.exger.2010.09.013
Zhang Q, Qu MR, Ouyang KH, Xiong XW, Pan K, Wen Q (2014) Effects of niacotinic acid on lactic acid fermentation of Streptococcus bovis by an in vitro method. Chin J Anim Nutr 26:2623–2629
Funding
This work was funded by the National Natural Science Foundation of China (31560648), Jiangxi Provincial Cattle and Sheep Industry Technology & System (JXARS-13), China Agriculture Research System of MOF and MARA (CARS-37).
Author information
Authors and Affiliations
Contributions
Z.G., K.Ouyang. designed the overall study. Z.G., Y.L., C.X., D.L., Q.Q., K.P., X.X., M.Q. performed experiments. Z.G., Y.L. wrote the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Animal Care and Use Committee of the College of Animal Science and Technology of Jiangxi Agricultural University (JXAULL-2020-27).
Consent to participate
Authors have permission to participate.
Consent for publication
Authors have permission for publication.
Competing interests
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, Z., Li, Y., Xu, C. et al. Niacin mitigates rumen epithelial damage in vivo by inhibiting rumen epithelial cell apoptosis on a high concentrate diet. Vet Res Commun 46, 699–709 (2022). https://doi.org/10.1007/s11259-022-09885-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-022-09885-9