Abstract
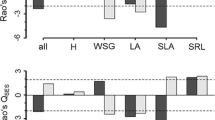
Advances in functional trait ecology allow us to evaluate community structure and community assembly in a more quantitative and generalised manner. Functional traits are known to differ greatly across local canopy openness, but previous studies often ignored such intraspecific variations when plant community structures are evaluated. We examined how considerations of canopy openness affect evaluations of community structure along a secondary successional gradient in a temperate climate zone in Japan. For 59 woody species found in nine forest plots aged from ca. 10 to 100 years old, we measured 12 functional traits with considering local canopy openness to capture their intraspecific variations across light gradients. Functional traits varied extensively both within and across species, and intraspecific variations were often tightly correlated with local canopy openness. Niche differentiation between deciduous and evergreen species in resource use strategy, as indicated by leaf mass per area (LMA), was more evident when taking canopy openness into consideration. Community-weighted means of the canopy-layer communities differed from those of the subordinate-layer communities, suggesting another niche differentiation along the vertical light gradient. Functional richness was often greater in the subordinate layer than in the canopy layer due to the co-occurrence of small individuals of canopy species and shade-tolerant specialists. Our study emphasises the importance of canopy openness when evaluating and understanding niche differentiation and forest community structures.




Similar content being viewed by others
Data availability
The dataset is available in Microsoft Excel format as Table S4.
References
Albert CH, Thuiller W, Yoccoz NG, Soudant A, Boucher F, Saccone P, Lavorel S (2010) Intraspecific functional variability: extent, structure and sources of variation. J Ecol 98:604–613. https://doi.org/10.1111/j.1365-2745.2010.01651.x
Albert CH, Grassein F, Schurr FM, Vieilledent G, Violle C (2011) When and how should intraspecific variability be considered in trait-based plant ecology? Perspect Plant Ecol Evol Syst 13:217–225. https://doi.org/10.1016/j.ppees.2011.04.003
Bond WJ (1989) The tortoise and the hare: ecology of angiosperm dominance and gymnosperm persistence. Biol J Linn Soc 36:227–249
Brodribb TJ, Pittermann J, Coomes DA (2012) Elegance versus speed: examining the competition between conifer and angiosperm trees. Int J Plant Sci 173:673–694. https://doi.org/10.1086/666005
Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12:351–366
Clements FE (1916) Plant succession: an analysis of the development of vegetation. Carnegie Institution of Washington, Washington
Connell JH, Slatyer RO (1977) Mechanisms of succession in natural communities and their role in community stability and organization. The Am Nat 111:1119–1144. https://doi.org/10.1086/283241
Cornwell WK, Schwilk DW, Ackerly DD (2006) A trait-based test for habitat filtering: convex hull volume. Ecology 87:1465–1471
Cramer VA, Hobbs RJ, Standish RJ (2008) What’s new about old fields? Land abandonment and ecosystem assembly. Trends Ecol Evol 23:104–112
Dwyer JM, Hobbs RJ, Mayfield MM (2014) Specific leaf area responses to environmental gradients through space and time. Ecology 95:399–410
Faraway JJ (2014) Linear models with R. CRC Press
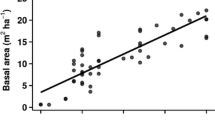
Furuno T, Uenishi Y, Uenishi K (1986) Information of nine studied forest stands that differed in forest ages in Wakayama, Japan. Bulletin of the Kyoto University Forests 57:60–75
Garnier E, Cortez J, Billès G, Navas M-L, Roumet C, Debussche M, Laurent G, Blanchard A, Aubry D, Bellmann A (2004) Plant functional markers capture ecosystem properties during secondary succession. Ecology 85:2630–2637
Grime JP, Hunt R (1975) Relative growth-rate: its range and adaptive significance in a local flora. J Ecol 63:393–422
Hansen MC, Ptapov PV, Moore R, Hancher M, Turubanova SA, Tyukavina A, Thau D, Stehman SV, Goetz SJ, Loveland TR, Kommareddy A, Egorov A, Chini L, CJustice CO, Townshend JRG (2013) High-resolution global maps of of 21st-century forest cover change. Science 342:850–853. https://doi.org/10.1126/science.1244693
Jennings SB, Brown ND, Sheil D (1999) Assessing forest canopies and understorey illumination: canopy closure, canopy cover and other measures. Forestry 72:59–74
Johnson DM, McCulloh KA, Woodruff DR, Meinzer FC (2012) Hydraulic safety margins and embolism reversal in stems and leaves: why are conifers and angiosperms so different? Plant Sci 195:48–53
Jung V, Violle C, Mondy C, Hoffmann L, Muller S (2010) Intraspecific variability and trait-based community assembly. J Ecol 98:1134–1140. https://doi.org/10.1111/j.1365-2745.2010.01687.x
Keenan TF, Niinemets Ü (2016) Global leaf trait estimates biased due to plasticity in the shade. Nat Plants 3:16201
Kikuzawa K, Onoda Y, Wright IJ, Reich PB (2013) Mechanisms underlying global temperature-related patterns in leaf longevity. Glob Ecol Biogeogr 22:982–993. https://doi.org/10.1111/geb.12042
King DA, Davies SJ, Tan S, Noor NSMD (2006) The role of wood density and stem support costs in the growth and mortality of tropical trees. J Ecol 94:670–680. https://doi.org/10.1111/j.1365-2745.2006.01112.x
Kitajima K, Poorter L (2010) Tissue-level leaf toughness, but not lamina thickness, predicts sapling leaf lifespan and shade tolerance of tropical tree species. New Phytol 186:708–721. https://doi.org/10.1111/j.1469-8137.2010.03212.x
Kohyama T, Takada T (2009) The stratification theory for plant coexistence promoted by one-sided competition. J Ecol 97:463–471. https://doi.org/10.1111/j.1365-2745.2009.01490.x
Kröber W, Böhnke M, Welk E, Wirth C, Bruelheide H (2012) Leaf trait-environment relationships in a subtropical broadleaved forest in South-East China. PLoS ONE. https://doi.org/10.1371/journal.pone.0035742
Kunstler G, Lavergne S, Courbaud B, Thuiller W, Vieilledent G, Zimmermann NE, Kattge J, Coomes DA (2012) Competitive interactions between forest trees are driven by species’ trait hierarchy, not phylogenetic or functional similarity: implications for forest community assembly. Ecol Lett 15:831–840. https://doi.org/10.1111/j.1461-0248.2012.01803.x
Lajoie G, Vellend M (2015) Understanding context dependence in the contribution of intraspecific variation to community trait–environment matching. Ecology 96:2912–2922
Laliberté E, Legendre P (2010) A distance-based framework for measuring functional diversity from multiple traits. Ecology 91:299–305
Lohbeck M, Poorter L, Paz H, Pla L, van Breugel M, Martínez-Ramos M, Bongers F (2012) Functional diversity changes during tropical forest succession. Perspect Plant Ecol Evol Syst 14:89–96. https://doi.org/10.1016/j.ppees.2011.10.002
Lohbeck M, Lebrija-Trejos E, Martínez-Ramos M, Meave JA, Poorter L, Bongers F (2015) Functional trait strategies of trees in dry and wet tropical forests are similar but differ in their consequences for succession. PLoS ONE 10:1–15. https://doi.org/10.1371/journal.pone.0123741
Lusk CH, Reich PB, Montgomery RA, Ackerly DD, Cavender-Bares J (2008) Why are evergreen leaves so contrary about shade? Trends Ecol Evol 23:299–303
Lusk CH, Onoda Y, Kooyman R, Gutierrez-Giron A (2010) Reconciling species-level vs plastic responses of evergreen leaf structure to light gradients: shade leaves punch above their weight. New Phytol 186:429–438. https://doi.org/10.1111/j.1469-8137.2010.03202.x
Mason NWH, Mouillot D, Lee WG, Wilson JB (2005) Functional richness, functional evenness and functional divergence: the primary components of functional diversity. Oikos 111:112–118. https://doi.org/10.1111/j.0030-1299.2005.13886.x
Matsuo T, Martínez-Ramos M, Bongers F, van der Sande MT, Poorter L (2021) Forest structure drives changes in light heterogeneity during tropical secondary forest succession. J Ecol 109:2871–2884. https://doi.org/10.1111/1365-2745.13680
Matsuo T, Hiura T, Onoda Y (2022) Vertical and horizontal light heterogeneity along gradients of secondary succession in cool and warm temperate forests. J Veg Sci 33:e13135. https://doi.org/10.1111/jvs.13135
Mayfield MM, Levine JM (2010) Opposing effects of competitive exclusion on the phylogenetic structure of communities. Ecol Lett 13:1085–1093. https://doi.org/10.1111/j.1461-0248.2010.01509.x
McGill BJ, Enquist BJ, Weiher E, Westoby M (2006) Rebuilding community ecology from functional traits. Trends Ecol Evol 21:178–185. https://doi.org/10.1016/j.tree.2006.02.002
Norden N, Angarita HA, Bongers F, Martínez-Ramos M, Granzow-de la Cerda I, Van Breugel M, Lebrija-Trejos E, Meave JA, Vandermeer J, Williamson GB (2015) Successional dynamics in Neotropical forests are as uncertain as they are predictable. Proc Natl Acad Sci 112:8013–8018
Onoda Y, Schieving F, Anten NPR (2008) Effects of light and nutrient availability on leaf mechanical properties of Plantago major: a conceptual approach. Ann Bot 101:727–736. https://doi.org/10.1093/aob/mcn013
Onoda Y, Saluñga JB, Akutsu K, Aiba S, Yahara T, Anten NPR, Salunga JB, Akutsu K, Aiba S, Yahara T, Anten NPR (2014) Trade-off between light interception efficiency and light use efficiency: implications for species coexistence in one-sided light competition. J Ecol 102:167–175. https://doi.org/10.1111/1365-2745.12184
Oohata S (1991) A study to estimate the forest biomass: a non-cutting method to use the piled up data. Bull Kyoto Univ for 63:23–36
Pérez-Harguindeguy N, Díaz S, Garnier E, Lavorel S, Poorter H, Jaureguiberry P, Bret-Harte MS, Cornwell WK, Craine JM, Gurvich DE, Urcelay C, Veneklaas EJ, Reich PB, Poorter L, Wright IJ, Ray P, Enrico L, Pausas JG, de Vos AC, Buchmann N, Funes G, Quétier F, Hodgson JG, Thompson K, Morgan HD, ter Steege H, Sack L, Blonder B, Poschlod P, Vaieretti MV, Conti G, Staver AC, Aquino S, Cornelissen JHC (2013) New handbook for standardised measurement of plant functional traits worldwide. Aust J Bot 61:167. https://doi.org/10.1071/bt12225
Poorter H, Niinemets Ü, Poorter L, Wright IJ, Villar R, Niinemets U, Poorter L, Wright IJ, Villar R (2009) Causes and consequences of variation in leaf mass per area (LMA):a meta-analysis. New Phytol 182:565–588. https://doi.org/10.1111/j.1469-8137.2009.02830.x
Poorter L, Rozendaal DMA, Bongers F, de Almeida-Cortez JS, Almeyda Zambrano AM, Álvarez FS, Andrade JL, Villa LFA, Balvanera P, Becknell JM, Bentos TV, Bhaskar R, Boukili V, Brancalion PHS, Broadbent EN, César RG, Chave J, Chazdon RL, Colletta GD, Craven D, de Jong BHJ, Denslow JS, Dent DH, DeWalt SJ, García ED, Dupuy JM, Durán SM, Espírito Santo MM, Fandiño MC, Fernandes GW, Finegan B, Moser VG, Hall JS, Hernández-Stefanoni JL, Jakovac CC, Junqueira AB, Kennard D, Lebrija-Trejos E, Letcher SG, Lohbeck M, Lopez OR, Marín-Spiotta E, Martínez-Ramos M, Martins SV, Massoca PES, Meave JA, Mesquita R, Mora F, de Souza MV, Müller SC, Muñoz R, Muscarella R, de Oliveira Neto SN, Nunes YRF, Ochoa-Gaona S, Paz H, Peña-Claros M, Piotto D, Ruíz J, Sanaphre-Villanueva L, Sanchez-Azofeifa A, Schwartz NB, Steininger MK, Thomas WW, Toledo M, Uriarte M, Utrera LP, van Breugel M, van der Sande MT, van der Wal H, Veloso MDM, Vester HFM, Vieira ICG, Villa PM, Williamson GB, Wright SJ, Zanini KJ, Zimmerman JK, Westoby M (2019) Wet and dry tropical forests show opposite successional pathways in wood density but converge over time. Nat Ecol Evol 3:928–934. https://doi.org/10.1038/s41559-019-0882-6
Pulsford SA, Lindenmayer DB, Driscoll DA (2016) A succession of theories: purging redundancy from disturbance theory. Biol Rev Camb Philos Soc 91:148–167. https://doi.org/10.1111/brv.12163
Reich PB (2014) The world-wide ‘fast–slow’plant economics spectrum: a traits manifesto. J Ecol 102:275–301
Rozendaal DMA, Hurtado VH, Poorter L (2006) Plasticity in leaf traits of 38 tropical tree species in response to light; relationships with light demand and adult stature. Funct Ecol 20:207–216. https://doi.org/10.1111/j.1365-2435.2006.01105.x
Rueffler C, Van Dooren TJM, Leimar O, Abrams PA (2006) Disruptive selection and then what? Trends Ecol Evol 21:238–245
Schönbeck L, Lohbeck M, Bongers F, Martínez Ramos M, Sterck F (2015) How do light and water acquisition strategies affect species selection during secondary succession in moist tropical forests? Forests. https://doi.org/10.3390/f6062047
Shipley B (2010) From plant traits to vegetation structure: chance and selection in the assembly of ecological communities. Cambridge University Press, Cambridge
Siefert A, Violle C, Chalmandrier L, Albert CH, Taudiere A, Fajardo A, Aarssen LW, Baraloto C, Carlucci MB, Cianciaruso MV, de Dantas VL, de Bello F, Duarte LD, Fonseca CR, Freschet GT, Gaucherand S, Gross N, Hikosaka K, Jackson B, Jung V, Kamiyama C, Katabuchi M, Kembel SW, Kichenin E, Kraft NJ, Lagerstrom A, Bagousse-Pinguet YL, Li Y, Mason N, Messier J, Nakashizuka T, Overton JM, Peltzer DA, Perez-Ramos IM, Pillar VD, Prentice HC, Richardson S, Sasaki T, Schamp BS, Schob C, Shipley B, Sundqvist M, Sykes MT, Vandewalle M, Wardle DA (2015) A global meta-analysis of the relative extent of intraspecific trait variation in plant communities. Ecol Lett 18:1406–1419. https://doi.org/10.1111/ele.12508
Spasojevic MJ, Suding KN (2012) Inferring community assembly mechanisms from functional diversity patterns: the importance of multiple assembly processes. J Ecol 100:652–661. https://doi.org/10.1111/j.1365-2745.2011.01945.x
Sterck F, Markesteijn L, Schieving F, Poorter L (2011) Functional traits determine trade-offs and niches in a tropical forest community. Proc Natl Acad Sci 108:20627–20632
Villéger S, Mason NWH, Mouillot D (2008) New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 89:2290–2301. https://doi.org/10.1890/07-1206.1
Violle C, Navas M, Vile D, Kazakou E, Fortunel C, Hummel I, Garnier E (2007) Let the concept of trait be functional! Oikos 116:882–892
Violle C, Enquist BJ, McGill BJ, Jiang L, Albert CH, Hulshof C, Jung V, Messier J (2012) The return of the variance: intraspecific variability in community ecology. Trends Ecol Evol 27:244–252. https://doi.org/10.1016/j.tree.2011.11.014
Walters MB, Reich PB (1999) Low-light carbon balance and shade tolerance in the seedlings of woody plants: do winter deciduous and broad-leaved evergreen species differ? New Phytol 143:143–154
Weedon JT, Cornwell WK, Cornelissen JH, Zanne AE, Wirth C, Coomes DA (2009) Global meta-analysis of wood decomposition rates: a role for trait variation among tree species? Ecol Lett 12:45–56. https://doi.org/10.1111/j.1461-0248.2008.01259.x
Westoby M, Falster DS, Moles AT, Vesk PA, Wright IJ (2002) Plant ecological strategies: some leading dimensions of variation between species. Annu Rev Ecol Syst 33:125–159. https://doi.org/10.1146/annurev.ecolsys.33.010802.150452
Whittaker RH (1953) A consideration of climax theory: the climax as a population and pattern. Ecol Monogr 23:41–78
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F, Cavender-Bares J, Chapin T, Cornelissen JHC, Diemer M, Flexas J, Garnier E, Groom PK, Gulias J, Hikosaka K, Lamont BB, Lee T, Lee W, Lusk C, Midgley JJ, Navas ML, Niinemets U, Oleksyn J, Osada N, Poorter H, Cornellssen JHC, Diemer M, Flexas J, Garnier E, Groom PK, Gulias J, Hikosaka K, Lamont BB, Lee T, Lee W, Lusk C, Midgley JJ, Navas ML, Niinemets Ü, Oleksyn J, Osada H, Poorter H, Pool P, Prior L, Pyankov VI, Roumet C, Thomas SC, Tjoelker MG, Veneklaas EJ, Villar R et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827. https://doi.org/10.1038/nature02403
Acknowledgements
We would like to thank Naoko Tokuchi, Hisashi Hasegawa, Tomoyuki Nakagawa and Masaru Okuda for their logistical support for our research in the Wakayama Research Forest of Kyoto University and would also like to thank the land owners for their permission to conduct this research in their forests. We would also like to thank Daiki Yokoyama, Kiyonori Nakashima, Yuki Tsujii, Waka Saito, Yukihiro Yoshida, Syoko Oguchi, Mana Mukai, Gaku Amada and Atushi Kakimoto for their help during the fieldwork and Michimasa Yamazaki for his help with the NC analyser. We are also grateful to Masahiro Aiba and Madelon Lohbeck for their constructive comments on the early draft.
Funding
This study was partly supported by JSPS KAKENHI #26711025 and by the Environment Research and Technology Development Fund (S-9) of the MOE of Japan.
Author information
Authors and Affiliations
Contributions
YN, KK and YO conceived the ideas and designed methodology. YN, YO and TI collected the data. YN analysed the data. YN, YO and TM led the writing of the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Daniel Potts.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nomura, Y., Matsuo, T., Ichie, T. et al. Quantifying functional trait assembly along a temperate successional gradient with consideration of intraspecific variations and functional groups. Plant Ecol 224, 669–682 (2023). https://doi.org/10.1007/s11258-023-01329-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-023-01329-x