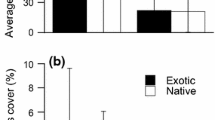
Abstract
Competition and herbivory can interact to influence the recovery of plant communities from disturbance. Previous attention has focused on the roles of dominant plant species in structuring plant communities, leaving the roles of subordinate species often overlooked. In this study, we examined how manipulating the density of a subordinate plant species, Solanum carolinense, and its insect herbivores influenced an old-field plant community in northern Florida following a disturbance. Five years following the disturbance, the initial densities of S. carolinense planted at the start of the experiment negatively influenced total plant cover and species diversity, and the cover of some grasses (e.g., Paspalum urvillei) and forbs (e.g., Rubus trivalis). Selectively removing herbivores from S. carolinense increased S. carolinense abundance (both stem densities and cover), increased the total cover of plants in the surrounding plant community, and affected plant community composition. Some plant species increased (e.g., Digitaria ciliaris, Solidago altissima) and others decreased (e.g., Paspalum notatum, Cynodon dactylon) in cover in response to herbivore removal. Herbivore effects on plant community metrics did not depend on S. carolinense density (no significant herbivory by density interaction), suggesting that even at low densities, a reduction of S. carolinense herbivores can influence the rest of the plant community. The recovery of the plant community was context dependent, depending on site- and plot-level differences in underlying environmental conditions and pre-disturbance plant community composition. We demonstrate that the density of and herbivory on a single subordinate plant species can affect the structure of an entire plant community.



Similar content being viewed by others
References
Armesto JJ, Pickett STA (1985) Experiments on disturbance in old-field plant communities: impact on species richness and abundance. Ecology 66:230–240
Bach CE (1994) Effects of a specialist herbivore (Altica subplicata) on Salix cordata and sand dune succession. Ecol Monogr 64:423–445
Carson WP, Root RB (1999) Top-down effects of insect herbivores during early succession: influence on biomass and plant dominance. Oecologia 121:260–272
Carson WP, Root RB (2000) Herbivory and plant species coexistence: community regulation by an outbreaking phytophagous insect. Ecol Monogr 70:73–99
Chase JM, Abrams PA, Grover JP, Diehl S, Chesson P, Holt RD, Richards SA, Nisbet RM, Case TJ (2002) The interaction between predation and competition: a review and synthesis. Ecol Lett 5:302–315
Clements FE (1938) Nature and structure of the climax. J Ecol 24:252–282
Connell JH (1983) On the prevalence and relative importance of interspecific competition—evidence from field experiments. Am Nat 122:661–696
Connell JH, Slayter R (1977) Mechanisms of succession in natural communities and their role in community stability and organization. Am Nat 111:1119–1144
Cramer VA, Hobbs RJ, Standish RJ (2008) What’s new about old fields? Land abandonment and ecosystem assembly. Trends Ecol Evol 23:104–112
Crawley MJ (1989) Insect herbivores and plant-population dynamics. Annu Rev Entomol 34:531–564
Crawley MJ, Pacala SW (1991) Herbivores, plant parasites, and plant diversity. In: Toft CA, Aeschlimann A, Bolis L (eds) Parasite-host associations: coexistence or conflict?. Oxford University Press, Oxford, pp 157–173
Diamond JM (1975) Assembly of species communities. In: Cody ML, Diamond JM (eds) Ecology and evolution of communities. Harvard University Press, Cambridge, pp 342–444
Firbank LG, Watkinson AR (1990) On the effects of competition: from monocultures to mixtures. In: Grace JB, Tilman D (eds) Perspectives on plant competition. The Blackburn Press, Caldwell, pp 165–192
Foster DF, Swanson F, Aber J, Burke I, Brokaw N, Tilman D, Knapp A (2003) The importance of land-use legacies to ecology and conservation. Bioscience 53:77–88
Gedan KB, Crain CM, Bertness MD (2009) Small-mammal herbivore control of secondary succession in New England tidal marshes. Ecology 90:430–440
Gleason HA (1927) Further views on the succession concept. Ecology 8:299–326
Gunasekara AS, Rubin AL, Goh K, Spurlock F, Tjeerdema RS (2008) Environmental fate and toxicology of Carbaryl. Rev Environ Contam Toxicol 196:95–121
Hahn PG, Orrock JL (2015) Land-use legacies and present fire regimes interact to mediate herbivory by altering the neighboring plant community. Oikos 124:497–506
Hairston NG, Smith FE, Slobodkin LB (1960) Community structure, population control and competition. Am Nat 94:421–425
Hambäck PA, Beckerman AP (2003) Herbivory and plant resource competition: a review of two interacting interactions. Oikos 101:26–37
Hervé M (2015) RVAideMemoire: diverse basic statistical and graphical functions package. Version 0.9-45-2. http://cran.r-project.org/web/packages/RVAideMemoire/index.html
Huntly N (1991) Herbivores and the dynamics of communities and ecosystems. Annu Rev Ecol Syst 22:477–503
Inouye BD (2001) Response surface experimental designs for investigating interspecific competition. Ecology 82:2696–2706
Jin S, Yang L, Danielson P, Homer C, Fry J, Xian G (2013) A comprehensive change detection method for updating the National Land Cover Database to circa 2011. Remote Sens Environ 132:159–175
Keddy PA (1990) Competitive hierarchies and centrifugal organization in plant communities. In: Grace JB, Tilman D (eds) Perspectives on Plant Competition. The Blackburn Press, Caldwell, pp 265–290
Kim TN (2012) Community-level consequences of plant–herbivore interactions. PhD Dissertation, Florida State University
Kim TN, Underwood N (2015) A full picture of plant neighborhood effects on herbivory: damage is both density and frequency dependent. Ecology 96:1431–1437
Kim TN, Underwood N, Inouye BD (2013) Insect herbivores change the outcome of plant competition through both inter- and intraspecific processes. Ecology 94:1753–1763
Lau JA, Strauss SY (2005) Insect herbivores drive important indirect effects of exotic plants on native communities. Ecology 86:2900–2997
Louda SM, Keeler KH, Holt RD (1990) Herbivore influences on plant performance and competitive interactions. In: Grace JB, Tilman D (eds) Perspectives on plant competition. The Blackburn Press, Caldwell, pp 413–444
Murthy N, Raghu K (1990) Effect of carbaryl and 1-naphthol on seedling growth of barley, and on growth and nodulation of groundnut in two soils. Proc Plant Sci 100:11–15
R Development Core Team (2014) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Schmidtlein S, Zimmermann P, Schüpferling R, Weiß C (2007) Mapping the floristic continuum: ordination space position estimated from imaging spectroscopy. J Veg Sci 18:131–140
Simms EL, Rausher MD (1987) Costs and benefits of plant-resistance to herbivory. Am Nat 130:570–581
Underwood N, Halpern S (2012) Insect herbivores, density dependence, and the performance of the perennial herb Solanum carolinense. Ecology 93:1026–1035
Wise MJ (2007) The herbivores of Solanum carolinense (Horsenettle) in Northern Virginia: natural history and damage assessment. Southeast Nat 6:505–522
Wise MJ, Sacchi CF (1996) Impact of two specialist insect herbivores on reproduction of horse nettle, Solanum carolinense. Oecologia 108:328–337
Acknowledgments
We thank J. Simonis, J. Fort, C. Venner, J. Hines, and numerous REU students for helping to establish and maintain the project over the 5 years. We thank the staff at the University of Florida North Florida Research and Education Center for their logistical support. Comments from Joshua Grinath greatly improved this manuscript. This project was funded by NSF DEB-0717221 to N. Underwood, and NSF DEB-0716922 and NRI, CSREES, USDA Grant 2006-35320-16686 to S. Halpern.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Lauchlan Frase.
Appendices
Appendix 1
See Fig. 4.
For logistical reasons, plot size varied with density (Spearman r s = −0.948) but to avoid completely confounding density with plot area, we used two plot sizes for each density treatment. Although this does present the possibility of spurious correlations with density and plot size, there was enough variation between the two variables to decouple their effects on plant responses. However, to ensure that spurious correlations were not present, we re-ran GLMs (see “Data analysis” in Methods), replacing the significant variable (e.g., density) with the other correlated variable (e.g., plot size) and compared model results and AICc values. If the replacement did not yield the same results and the other variable did not appear significant, we conclude that it was not a spurious correlation and that the effects are real. In all cases, the density and area effects represented in the paper were real
Appendix 2
Index of prior plant composition.
Plant composition prior to disturbance was estimated from high-resolution digital orthographic quarter quad imagery acquired in March 2004, 3 years before the start of the experiment. Different plant species, but also variation in soil and vegetation water content, can reflect light of different intensities across a range of the electromagnetic (EM) spectrum, which forms a basis for all land cover classifications (e.g., Jin et al. 2013). Accordingly, we assume that the digital number values corresponding to each 1 m2 pixel represents the average spectral radiance of the individual plants within that area, and therefore the composition of plant species. Moreover, we assume underlying environmental variation potentially driving differences in prior plant cover (e.g., soil type or hydrological properties) are incorporated within radiance values. We included the three bands from the image (green, blue, and infrared) in a principal components analysis (PCA) in order to generate an image that represents spectral radiance from multiple ranges of the EM spectrum on a single band. Ordination techniques such as this can be used to characterize vegetation communities in a continuous fashion rather than forming discrete habitat classes (Schmidtlein et al. 2007). We overlaid the plot boundaries on the PCA image product and, for each plot, calculated the mean of the first-axis PCA scores (Fig. 5). The first axis explained 71.6 % of the variation in the original three-band image. The mean for each plot was used in subsequent analyses as an index of prior plant cover.
See Fig. 5.
PCA images illustrating differences in prior plant cover among plots in the west (a) and east (b) sites. Plot positions are outlined in white and ranged in sizes from 1.6 to 98.4 m2. The five initial planting densities of S. carolinense within each plot were 0.65, 2.77, 11.11, 22.68, and 30.86 stems/m2. This figure depicts all three axes as a false color composite with PCA axis 1 (71.6 %) coded as red, PCA axis 2 (25.7 %) coded as blue, and PCA axis 3 (2.7 %) coded as green, but only the mean of the first-axis scores corresponding to areas within each plot was used as a covariate in our analyses “prior plant cover”
Appendix 3
See Table 2.
Appendix 4
See Table 3.
Rights and permissions
About this article
Cite this article
Kim, T.N., Spiesman, B.J., Buchanan, A.L. et al. Selective manipulation of a non-dominant plant and its herbivores affects an old-field plant community. Plant Ecol 216, 1029–1045 (2015). https://doi.org/10.1007/s11258-015-0487-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-015-0487-3