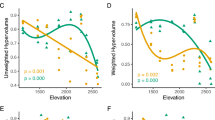
Abstract
For decades, evolutionary plant biologists have been interested in how females reap a fitness advantage and are thus maintained in gynodioecious plants. Recent attention has focused on the role of multi-species interactions in the maintenance of females, wherein the role of arbuscular mycorrhizal fungi (AMF) is virtually unknown. AMF form symbiotic associations with plants, often resulting in enhanced nutrient acquisition and tolerance to biotic and abiotic stress. Depending on the degree to which females and hermaphrodites rely on their AMF partners, AMF could differentially affect plant tolerance to stress. We conducted a field experiment to examine (1) if females and hermaphrodites of the gynodioecious herb, Polemonium foliosissimum, differentially associate with AMF, and (2) if there was a difference in AMF colonization between the sexes after imposing stress by simulating herbivore damage. First, we compared mycorrhizal colonization between the sexes under ambient (i.e., undamaged) field conditions. Then, for a second set of plants, we simulated herbivory by clipping plants and compared mycorrhizal colonization, leaf phosphorus concentration [P], and fruit and seed production between clipped and control plants. Mycorrhizal colonization did not differ between sexes under ambient field conditions or under the stress imposed by clipping. Clipping did, however, influence both percent root length and vesicle colonization in a plant-size-dependent manner. Overall, colonization and vesicle number increased with plant size for clipped plants, but decreased with size for non-clipped plants. Leaf [P] did not differ between the sexes nor was influenced by clipping, and leaf [P] was not associated with mycorrhizal colonization. Furthermore, neither fruit set nor seed production was associated with mycorrhizal colonization. Our findings suggest that the sexes of P. foliosissimum do not differentially associate with nor differentially garner benefit from mycorrhizal fungi. However, a complete accounting of the costs and benefits of AMF for gynodioecious species must encompass the full life-history of the plant.



Similar content being viewed by others
References
Alexander T, Meier R, Toth R, Weber HC (1988) Dynamics of arbuscule development and degeneration in mycorrhizas of Triticum aestivum L. and Avena sativa L. with references to Zea mays L. New Phytol 110:363–370
Alexander T, Toth R, Meier R, Weber HC (1989) Dynamics of arbuscule development and degeneration in onion, bean, and tomato with reference to vesicular–arbuscular mycorrhizae in grasses. Can J Bot 67:2505–2513
Anway JC (1968) The systematic botany and taxonomy of Polemonium foliosissimum A. Gray (Polemoneaceae). Am Midl Nat 79:458–475
Ashman T-L (2000) Pollinator selectivity and its implications for the evolution of dioecy and sexual dimorphism. Ecology 81:2577–2591
Ashman T-L (2006) The evolution of separate sexes: a focus on the ecological context. In: Harder LD, Barrett SCH (eds) The ecology and evolution of flowers. Oxford University Press, Oxford, pp 419–465
Ashman T-L, Diefenderfer C (2001) Sex ratio represents a unique context for selection on attractive traits: consequences for the evolution of sexual dimorphism. Am Nat 157:334–347
Ashman T-L, Penet L (2007) Direct and indirect effects of a sex-biased antagonist on male and female fertility: consequences for reproductive trait evolution in a gender-dimorphic plant. Am Nat 169:595–608
Asikainen E, Mutikainen P (2005) Pollen and resources limitation in a gynodioecious species. Am J Bot 92:487–494
Barr, B Gothic Weather, Long-term Weather Data (1974–2015) Rocky Mountain Biology Laboratory. http://www.gothicwx.org/long-term-snow.html. Accessed 15 Mar 2015
Barto EK, Rillig MC (2010) Does herbivory really suppress mycorrhiza? A meta-analysis. J Ecol 98:745–753
Bennett AE, Bever JD (2007) Mycorrhizal species differentially alter plant growth and response to herbivory. Ecology 88:210–218
Biermann B, Linderman RG (1983) Use of vesicular–arbuscular mycorrhizal roots, intraradical vesicles, and extraradical vesicles as inoculum. New Phytol 95:97–105
Borowicz VA (2013) The impact of arbuscular mycorrhizal fungi on plant growth following herbivory: a search for patter. Acta Oecol 52:1–9
Botham R, Collin CL, Ashman T-L (2009) Plant-mycorrhizal fungus interactions affect the expression of inbreeding depression in wild strawberry. Int J Plant Sci 2:143–150
Brody AK (1997) Effects of pollinators, herbivores, and seed predators on flowering phenology. Ecology 78:1624–1631
Brundrett MC (2009) Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320:37–77
Brundrett MC, Kendrick B (1990) The roots of mycorrhizas of herbaceous woodland plants. I. Quantitative aspects of morphology. New Phytol 114:457–468
Brundrett MC, Piché Y, Peterson RL (1985) A developmental study of the early stages in vesicular–arbuscular mycorrhiza formation. Can J Bot 63:184–194
Casper BB, Forseth IN, Wait DA (2006) A stage-based study of drought response in Cryptantha flava (Boraginaceae): gas exchange, water use efficiency, and whole plant performance. Am J Bot 93:977–987
Charlesworth B, Charlesworth D (1978) A model for the evolution of dioecy and gynodioecy. Am Nat 112:975–997
Clarke G, Brody AK (2015) Gender inequity in pre-dispersal seed predation contributes to female seed set advantage in a gynodioecious species. Ecology 16(6):1186–1195
Collin CL, Ashman T-L (2010) Root fungi in wild strawberry: root colonization depends on host inbreeding. Evol Ecol Res 12:477–490
Collin CL, Pennings PS, Rueffler C, Widmer A, Shykoff JA (2002) Natural enemies and sex: how seed predation and pathogens contribute to sex-differential reproductive success in a gynodioecious plant. Oecolgia 131:94–102
Core Team R (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Cornelissen JHC, Lavorel S, Garnier E, Dias S, Buchmann N, Gurvich DE, Reich PB, ter Steege H, Morgan HD, van der Heijden MGA, Pausas JG, Poorter H (2003) A handbook of protocols for standardized and easy measurement of plant functional traits worldwide. Aust J Bot 51:335–380
Cox G, Tinker PB (1976) Translocation and transfer of nutrients in vesicular–arbuscular mycorrhizas. I. The arbuscule and phosphorus transfer: a quantitative ultrastructural study. New Phytol 77:371–378
de Mendiburu (2014) Agricolae: Statistical Procedures for Agricultural Research. R package version 1.2-1
Donovan LA, Ehleringer JR (1992) Contrasting water-use patterns among size and life-history classes of a semi-arid shrub. Funct Ecol 6:482–488
Dufay M, Billard E (2012) How much better are females? The occurrence of female advantage, its proximal causes and its variation within and among gynodioecious species. Ann Bot 109:505–519
Eppley SM, Mercer CA, Haaning C, Graves CB (2009) Sex-specific variation in the interaction between Distichlis spicata (Poaceae) and mycorrhizal fungi. Am J Bot 96:1967–1973
Fitter AH, Graves JD, Watkins NK, Robinson D (1998) Carbon transfer between plants and its control in networks of arbuscular mycorrhizas. Funct Ecol 12:406–412
Gange AC, Smith AK (2005) Arbuscular mycorrhizal fungi influence visitation rates of pollinating insects. Ecol Entomol 30:600–606
Gehring CA, Whitham TG (1994) Interactions between aboveground herbivores and the mycorrhizal mutualists of plants. Trends Ecol Evol 7:251–255
Gehring CA, Whitham TG (2002) Mycorrhizae-herbivore interactions: population and community consequences. In: van der Heijden MGA, Sanders IR (eds) Mycorrhizal Ecology. Springer, Heidelberg, pp 295–320
González JB (2014) Investigations into host-specific interactions and local adaptation in the mycorrhizal symbiosis. MSc Thesis, University of Vermont
Heinemeyer A, Fitter AH (2004) Impact of temperature on the arbuscular mycorrhizal (AM) symbiosis: growth responses of the host plant and its AM fungal partner. J Exp Bot 55:525–534
Jakobsen I, Rosendahl L (1990) Carbon flow into soil and external hyphae from roots of mycorrhizal cucumber plants. New Phytol 115:77–83
Johnson NC, Graham JH, Smith FA (1997) Functioning of mycorrhizal associations along the mutualism–parasitism continuum. New Phytol 135:575–585
Jones MD, Smith SE (2004) Exploring functional definitions of mycorrhizas: are mycorrhizas always mutualisms? Can J Bot 82:1089–1109
Klironomos JN, McCune J, Moutoglis P (2004) Species of arbuscular mycorrhizal fungi affect mycorrhizal responses to simulated herbivory. Appl Soil Ecol 26:133–141
Knight TM, Steets JA, Vamosi JC, Mazer SJ, Burd M, Campbell DR, Dudash MR, Johnston MO, Mitchell RJ, Ashman T-L (2005) Pollen limitation of plant reproduction: pattern and process. Ann Rev Ecol Evol S 36:467–497
Koide RT (1991) Tansley review no. 29. Nutrient supply, nutrient demand and plant response to mycorrhizal infection. New Phytol 117:365–386
Kula A, Hartnett D, Wilson G (2005) Effects of mycorrhizal symbiosis on tallgrass prairie plant-herbivore interactions. Ecol Lett 8:61–69
Lewis D (1941) Male sterility in natural populations of hermaphrodite plants. The equilibrium between females and hermaphrodites to be expected with different types of inheritance. New Phytol 40:56–63
Liu A, Wang B, Hamel C (2004) Arbuscular mycorrhiza colonization and development at suboptimal root zone temperatures. Mycorrhiza 14:93–101
Lu X, Koide RT (1994) The effects of mycorrhizal infection on components of plant growth and reproduction. New Phytol 128:211–218
Marshall M, Ganders FR (2001) Sex-biased seed predation and the maintenance of females in a gynodioecious plant. Am J Bot 88:1437–1443
Maschinski J, Whitham TG (1989) The continuum of plant responses to herbivory: the influence of plant association, nutrient availability, and timing. Am Nat 143:1–19
McCall AC, Barr CM (2012) Why do florivores prefer hermaphrodites over females in Nemophila menziesii (Boraginaceae)? Oecologia 170:147–157
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular–arbuscular mycorrhizal fungi. New Phytol 115:495–501
Miller RM, Miller SP, Jastrow JD, Rivetta CB (2002) Mycorrhizal mediated feedbacks influence net carbon gain and nutrient uptake in Andropogon gerardii. New Phytol 155:149–162
Neumann E, George E (2010) Nutrient uptake: the arbuscular mycorrhiza fungal symbiosis as a plant nutrient acquisition strategy. In: Koltai H, Kapulnik Y (eds) Arbuscular mycorrhizas: physiology and function. Springer, New York, pp 137–167
Newsham KK, Fitter AH, Watkinson AR (1995) Arbuscular mycorrhiza protect an annual grass from root pathogenic fungi in the field. J Ecol 83:991–1000
Parniske M (2008) Arbuscular mycorrhiza: the mother of plant root endosymbioses. Nat Rev Microbiol 6:763–775
Richards AJ (1997) Plant breeding systems, 2nd edn. Chapman and Hall Inc, New York
Schenck NC, Graham SO, Green NE (1975) Temperature and light effect on contamination and spore germination of vesicular–arbuscular mycorrhizal fungi. Mycologia 67:1189–1192
Shykoff JA, Kolokotronis S-O, Collin CL, López-Villavicencio M (2003) Effects of male sterility on reproductive traits in gynodioecious plants: a meta-analysis. Oecolgia 135:1–9
Smith SE, Read D (2008) Mycorrhizal symbiosis, 3rd edn. Academic Press, Amsterdam
Spigler RB, Ashman T-L (2012) Gynodioecy to dioecy: are we there yet? Ann Bot 109:531–543
Tommerup IC (1983) Temperature relations of spore germination and hyphal growth of vesicular–arbuscular mycorrhizal fungi in soil. T Brit Mycol Soc 81:381–387
van der Heijden MGA, Horton TR (2009) Socialism in soil? The importance of mycorrhizal fungal networks for facilitation in natural ecosystems. J Ecol 97:1139–1150
Varga S (2010) Review. Effects of arbuscular mycorrhizas on reproductive traits in sexually dimorphic plants. Span J Agric Res 8(S1):S11–S24
Varga S, Kytöviita M-M (2008) Sex-specific responses to mycorrhiza in a dioecious species. Am J Bot 95:1225–1232
Varga S, Kytöviita M-M (2010a) Gender dimorphism and mycorrhizal symbiosis affect floral visitors and reproductive output in Geranium sylvaticum. Funct Ecol 24:750–758
Varga S, Kytöviita M-M (2010b) Mycorrhizal benefit differs among the sexes in a gynodioecious species. Ecology 91:2583–2593
Varga S, Kytöviita M-M, Siikamäki P (2009) Sexual differences in response to simulated herbivory in the gynodioecious herb Geranium sylvaticum. Plant Ecol 202:325–336
Varga S, Vega-Frutis R, Kytöviita M-M (2013) Transgenerational effects of plant sex and arbuscular mycorrhizal symbiosis. New Phytol 199:812–821
Vega-Frutis R, Guevara R (2009) Different arbuscular mycorrhizal interactions in male and female plants of wild Carica papaya L. Plant Soil 322:165–176
Vega-Frutis R, Guevara R (2013) Greater mycorrhizal colonization of unisexual morphs than of hermaphroditic morphs of Jacaratia mexicana during flowering and fruiting in central Mexico. Symbiosis 59:173–181
Vega-Frutis R, Munguía-Rosas MA, Varga S, Kytöviita M-M (2013) Sex-specific patterns of antagonistic and mutualistic biotic interactions in dioecious and gynodioecious plants. Perspect Plant Ecol 15:45–55
Vierheilig H, Coughlan AP, Wyss U, Piché Y (1998) Ink and vinegar, a simple staining technique for arbuscular-mycorrhizal fungi. Appl Environ Microb 64:5004–5007
Warton DI, Hui FKC (2011) The arcsine is asinine: the analysis of proportions in ecology. Ecology 92:3–10
Zhang YW, Yang CF, Zhao JM, Guo YH (2009) Selective nectar robbing in a gynodioecious plant (Glechoma longituba) enhances female advantage. J Evolut Biol 22:527–535
Zimmerman M (1980a) Reproduction in Polemonium: competition for pollinators. Ecology 61:497–501
Zimmerman M (1980b) Reproduction in Polemonium: pre-dispersal seed predation. Ecology 61:502–506
Acknowledgments
Special thanks to the staff and scientists at RMBL and members of the Brody and Gotelli Labs. We thank Joe Morton and Renee Petipas for helpful comments during the design of the study. We thank Jeanne Harris and Jim Vigoreaux, and two anonymous reviewers for constructive criticism of earlier versions of this manuscript, and Alan Howard and Ruth Mickey for help with the statistical analysis. JBG was supported by a National Science Foundation REU grant to RMBL (DBI 0753774), a University of Vermont College of Agriculture and Life Sciences FUSE grant and an Honors College grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Gavin Kernaghan.
Rights and permissions
About this article
Cite this article
González, J.B., Clarke, G.L. & Brody, A.K. Lack of sex-specific differences in mycorrhizal associations and response to herbivory in the gynodioecious herb, Polemonium foliosissimum . Plant Ecol 216, 951–962 (2015). https://doi.org/10.1007/s11258-015-0481-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-015-0481-9