Abstract
Background
TRAF-interacting protein (TRAIP) is a RING-type E3 ubiquitin ligase, which has been implicated in various cellular processes and participated in various cancers as an oncogene. However, the function and potential mechanism of TRAIP in prostate cancer (PCa) have not been investigated so far.
Methods
Public TGCA data were used to evaluate the expression profile of TRAIP in prostatic tumors. The relative expression of TRAIP and TRAF2 in PCa tissues and tumor cell lines was detected by qPCR, western blot, and IHC staining. Next, TRAIP knockdown and overexpression plasmids were constructed and transfected into PCa cell lines. Moreover, cell proliferation, invasion, migration, and apoptosis were measured by colony formation, Transwell, wound healing, and flow cytometry assays. Subsequently, cell cycle and signaling pathway-related proteins were tested by western blot. Finally, the effect of TRAIP on PCa was measured based on the nude mouse xenograft model.
Results
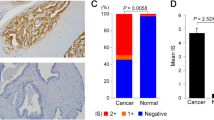
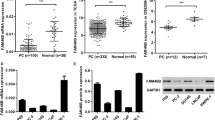
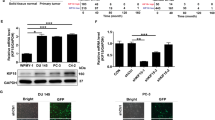
TRAIP was significantly upregulated in PCa tissues and tumor cell lines. In addition, TRAIP promoted cell proliferation, invasion, and migration of PCa cell lines. Such an oncogenic property was mediated by the cell cycle arrest and the inhibition of apoptosis, as indicated by different functional assays and the expression of cell cycle and apoptosis regulatory proteins in cultured cells. Moreover, TRAIP combined with TRAF2 to activate PI3K/AKT pathway. Finally, TRAIP depletion suppressed the growth of tumors and cell proliferation in vivo.
Conclusions
Our study first revealed that TRAIP promoted tumor progression and identified it as a potential therapeutic target for PCa patients in the future.





Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available within the paper, and any raw data can be obtained from the corresponding author upon request.
References
Liu M, Huang Q, A J, Li L, Li X, Zhang Z, Dong JT (2021) The cardiac glycoside deslanoside exerts anticancer activity in prostate cancer cells by modulating multiple signaling pathways. Cancers (Basel) 13 (22). https://doi.org/10.3390/cancers13225809
Sekhoacha M, Riet K, Motloung P, Gumenku L, Adegoke A, Mashele S (2022) Prostate cancer review: genetics, diagnosis, treatment options, and alternative approaches. Molecules 27 (17). https://doi.org/10.3390/molecules27175730
Guo Z, Zeng Y, Chen Y, Liu M, Chen S, Yao M, Zhang P, Zhong F, Jiang K, He S, Yuan G (2020) TRAIP promotes malignant behaviors and correlates with poor prognosis in liver cancer. Biomed Pharmacother 124:109857. https://doi.org/10.1016/j.biopha.2020.109857
Feng W, Guo Y, Huang J, Deng Y, Zang J, Huen MS (2016) TRAIP regulates replication fork recovery and progression via PCNA. Cell Discov 2:16016. https://doi.org/10.1038/celldisc.2016.16
Zheng Y, Jia H, Wang P, Liu L, Chen Z, Xing X, Wang J, Tan X, Wang C (2023) Silencing TRAIP suppresses cell proliferation and migration/invasion of triple negative breast cancer via RB-E2F signaling and EMT. Cancer Gene Ther 30(1):74–84. https://doi.org/10.1038/s41417-022-00517-7
Zong X, Xiao X, Shen B, Jiang Q, Wang H, Lu Z, Wang F, Jin M, Min J, Wang F, Wang Y (2021) The N6-methyladenosine RNA-binding protein YTHDF1 modulates the translation of TRAF6 to mediate the intestinal immune response. Nucleic Acids Res 49(10):5537–5552. https://doi.org/10.1093/nar/gkab343
Xiao X, Cheng Y, Fu J, Lu Z, Wang F, Jin M, Zong X, Wang Y (2021) Gut immunity and microbiota dysbiosis are associated with altered bile acid metabolism in LPS-challenged piglets. Oxiditive Medicine and Cellular Longevity 2021:6634821. https://doi.org/10.1155/2021/6634821
Xiao X, Song D, Cheng Y, Hu Y, Wang F, Lu Z, Wang Y (2019) Biogenic nanoselenium particles activate Nrf2-ARE pathway by phosphorylating p38, ERK1/2, and AKT on IPEC-J2 cells. J Cell Physiol 234(7):11227–11234. https://doi.org/10.1002/jcp.27773
Zong X, Cheng Y, Xiao X, Fu J, Wang F, Lu Z, Wang Y, Jin M (2021) Protective effects of sulfated polysaccharide from Enterobacter cloacae Z0206 against DSS-induced intestinal injury via DNA methylation. Int J Biol Macromol 183:861–869. https://doi.org/10.1016/j.ijbiomac.2021.04.182
Li QJ, Wang M, Hu YQ, Zhao ES, Li J, Ren LL, Wang M, Xu YD, Liang Q, Zhang D, Lai YR, Liu SY, Peng XS, Zhu CM, Ye LP (2021) MYBL2 disrupts the Hippo-YAP pathway and confers castration resistance and metastatic potential in prostate cancer. Theranostics 11(12):5794–5812. https://doi.org/10.7150/thno.56604
Pan JS, Liu F, Xiao XL, Xu RH, Dai L, Zhu MZ, Xu HC, Xu YX, Zhao AG, Zhou WJ, Dang YQ, Ji G (2022) METTL3 promotes colorectal carcinoma progression by regulating the m6A-CRB3-Hippo axis. J Exp Clin Canc Res 41 (1). ARTN 19 https://doi.org/10.1186/s13046-021-02227-8
Xiao X, Cheng Y, Song D, Li X, Hu Y, Lu Z, Wang F, Wang Y (2019) Selenium-enriched Bacillus paralicheniformis SR14 attenuates H(2)O(2)-induced oxidative damage in porcine jejunum epithelial cells via the MAPK pathway. Appl Microbiol Biotechnol 103(15):6231–6243. https://doi.org/10.1007/s00253-019-09922-9
Si ZZ, Yu L, Jing HY, Wu L, Wang XD (2021) Oncogenic lncRNA ZNF561-AS1 is essential for colorectal cancer proliferation and survival through regulation of miR-26a-3p/miR-128–5p-SRSF6 axis. J Exp Clin Canc Res 40 (1). https://doi.org/10.1186/s13046-021-01882-1
Chen H, Zhou L, Wu X, Li R, Wen J, Sha J, Wen X (2016) The PI3K/AKT pathway in the pathogenesis of prostate cancer. Front Biosci (Landmark Ed) 21(5):1084–1091. https://doi.org/10.2741/4443
Karimian A, Mir SM, Parsian H, Refieyan S, Mirza-Aghazadeh-Attari M, Yousefi B, Majidinia M (2019) Crosstalk between Phosphoinositide 3-kinase/Akt signaling pathway with DNA damage response and oxidative stress in cancer. J Cell Biochem 120(6):10248–10272. https://doi.org/10.1002/jcb.28309
Ward AB, Mir H, Kapur N, Gales DN, Carriere PP, Singh S (2018) Quercetin inhibits prostate cancer by attenuating cell survival and inhibiting anti-apoptotic pathways. World J Surg Oncol 16(1):108. https://doi.org/10.1186/s12957-018-1400-z
De Blasio C, Verma N, Moretti M, Cialfi S, Zonfrilli A, Franchitto M, Truglio F, De Smaele E, Ichijo H, Naguro I, Screpanti I, Talora C (2021) Functional cooperation between ASK1 and p21(Waf1/Cip1) in the balance of cell-cycle arrest, cell death and tumorigenesis of stressed keratinocytes. Cell Death Discovery 7 (1). https://doi.org/10.1038/s41420-021-00459-3
Liu J, Peng Y, Wei W (2022) Cell cycle on the crossroad of tumorigenesis and cancer therapy. Trends Cell Biol 32(1):30–44. https://doi.org/10.1016/j.tcb.2021.07.001
Kloeber JA, Lou Z (2022) Critical DNA damaging pathways in tumorigenesis. Semin Cancer Biol 85:164–184. https://doi.org/10.1016/j.semcancer.2021.04.012
Matthews HK, Bertoli C, de Bruin RAM (2022) Cell cycle control in cancer. Nat Rev Mol Cell Biol 23(1):74–88. https://doi.org/10.1038/s41580-021-00404-3
Almeida S, Ryser S, Obarzanek-Fojt M, Hohl D, Huber M (2011) The TRAF-interacting protein (TRIP) is a regulator of keratinocyte proliferation. J Investig Dermatol 131(2):349–357. https://doi.org/10.1038/jid.2010.329
Deng TG, Yan GB, Song X, Xie L, Zhou Y, Li JL, Hu XX, Li Z, Hu J, Zhang YB, Zhang H, Sun Y, Feng PF, Wei D, Hu B, Liu J, Tan WH, Ye M (2018) Deubiquitylation and stabilization of p21 by USP11 is critical for cell-cycle progression and DNA damage responses. Proc Natl Acad Sci USA 115(18):4678–4683. https://doi.org/10.1073/pnas.1714938115
Jaiswal SK, Raj S, DePamphilis ML (2021) Developmental Acquisition of p53 Functions. GENES 12 (11). https://doi.org/10.3390/genes12111675
Bhat EA, Kim CM, Kim S, Park HH (2018) In Vitro Inhibitory Mechanism Effect of TRAIP on the Function of TRAF2 Revealed by Characterization of Interaction Domains. Int J Mol Sci 19 (8). https://doi.org/10.3390/ijms19082457
Acknowledgements
Not applicable.
Funding
No funding was used in this study.
Author information
Authors and Affiliations
Contributions
YG and CL designed the experiments, carried them out, analyzed and interpreted the data, and prepared the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors state that there are no conflicts of interest to disclose.
Ethics approval
Ethical approval was obtained from the Ethics Committee of Anhui Second People's Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gu, Y., Liang, C. TRAIP suppressed apoptosis and cell cycle to promote prostate cancer proliferation via TRAF2–PI3K–AKT pathway activation. Int Urol Nephrol 56, 1639–1648 (2024). https://doi.org/10.1007/s11255-023-03890-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-023-03890-w