Abstract
Purpose
Plasma atherogenic index (PAI) was shown to be positively correlated with the presence of malignity in patients with suspicious findings for renal cell cancer and colon cancer in reported studies. In this study, we aimed to evaluate whether there is an association with the presence of malignity in patients PI-RADS 3 prostate lesions and PAI.
Methods
This retrospective study reviewed the data of 139 patients who underwent transrectal ultrasonography-guided systematic and cognitive fusion prostate biopsy for PI-RADS 3 lesions in multiparametric magnetic resonance imaging. The patients were divided to two groups as malign (n = 33) and benign (n = 106). The association between age, body mass index, comorbidities, smoking status, prostate-specific antigen (PSA), PSA density, free/total PSA, prostate weight, lesion diameter, triglyceride value, high-density lipoprotein-cholesterol value, PAI value data and presence of malignity were investigated by descriptive, multivariate and receiver-operating characteristic (ROC) analysis.
Results
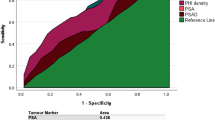
PSA, PSAD, lesion diameter and PAI value were statistically significantly higher in the malignant group compared to the benign group, and the free/total PSA ratio was lower. In multivariate logistic regression analysis, PSA > 9.9 ng/ml, free/total PSA < 12.1%, lesion diameter > 13.5 mm and PAI > 0.13 were identified as independent risk factors for presence of prostate malignancy.
Conclusion
PAI was found to be a predictive parameter for prostate cancer in PI-RADS 3 prostate lesions. Our study can open new thoughts about PAI as metric to assess the prostate cancer risk.

Similar content being viewed by others
Data availability
The data that support the findings of this study are openly available in Figshare Repository at https://figshare.com/s/1b5021c69e26f68be453.
Code availability
Not applicable.
References
Ferlay J, Ervik M, Lam F et al (2020) Global cancer observatory: cancer today. Available online at: https://www.gco.iarc.fr/today/ Accessed 29 Aug 2022.
Culp MB, Soerjomataram I, Efstathiou JA, Bray F, Jemal A (2020) Recent global patterns in prostate cancer incidence and mortality rates. Eur Urol 77:38–52
EAU Guidelines Office (2022) Arnhem, the Netherlands. EAU guidelines. Edn. presented at the EAU Annual Congress Amsterdam
Barkovich EJ, Shankar PR, Westphalen AC (2019) A systematic review of the existing prostate imaging reporting and data system version 2 (PI-RADSv2) literature and subset meta-analysis of PI-RADSv2 categories stratified by Gleason scores. AJR Am J Roentgenol 212:847–854
Eklund M, Jäderling F, Discacciati A et al (2021) MRI-targeted or standard biopsy in prostate cancer screening. N Engl J Med 385:908–920
Houlahan KE, Salmasi A, Sadun TY et al (2019) Molecular hallmarks of multiparametric magnetic resonance imaging visibility in prostate cancer. Eur Urol 76:18–23
Eldred-Evans D, Burak P, Connor MJ et al (2021) Population-based prostate cancer screening with magnetic resonance imaging or ultrasonography: the IP1-PROSTAGRAM study. JAMA Oncol 7:395–402
Kim L, Boxall N, George A et al (2020) Clinical utility and cost modelling of the phi test to triage referrals into image-based diagnostic services for suspected prostate cancer: the PRIM (Phi to RefIne Mri) study. BMC Med 18:1–9
Giambelluca D, Cannella R, Vernuccio F et al (2021) PI-RADS 3 lesions: role of prostate MRI texture analysis in the identification of prostate cancer. Curr Probl Diagn Radiol 50:175–185
Esposito K, Chiodini P, Capuano A et al (2013) Effect of metabolic syndrome and its components on prostate cancer risk: meta-analysis. J Endocrinol Invest 36:132–139
Blanc-Lapierre A, Spence A, Karakiewicz PI, Aprikian A, Saad F, Parent M-É (2015) Metabolic syndrome and prostate cancer risk in a population-based case–control study in Montreal, Canada. BMC Public Health 15:1–11
Vidal AC, Howard LE, Moreira DM, Castro-Santamaria R, Andriole GL, Freedland SJ (2014) obesity increases the risk for high-grade prostate cancer: results from the REDUCE study. Cancer Epidemiol Biomark Prev Cancer Epidemiol 23:2936–2942
Freedland S, Hamilton R, Gerber L et al (2013) Statin use and risk of prostate cancer and high-grade prostate cancer: results from the REDUCE study. Prostate Cancer Prostatic Dis 16:254–259
Munir R, Usman H, Hasnain S, Smans K, Kalbacher H, Zaidi N (2014) Atypical plasma lipid profile in cancer patients: cause or consequence? Biochimie 102:9–18
Pirro M, Ricciuti B, Rader DJ, Catapano AL, Sahebkar A, Banach M (2018) High density lipoprotein cholesterol and cancer: marker or causative? Prog Lipid Res 71:54–69
Fernández-Macías JC, Ochoa-Martínez AC, Varela-Silva JA, Pérez-Maldonado IN (2019) Atherogenic index of plasma: novel predictive biomarker for cardiovascular illnesses. Arch Med Res 50:285–294
Karabay E, Karsiyakali N, Duvar S, Tosun C, Aslan AR, Yucebas OE (2019) Relationship between plasma atherogenic index and final pathology of Bosniak III-IV renal masses: a retrospective, single-center study. BMC Urol 19:1–7
Gundogdu UD, Coban FK (2021) A study of atherogenic plasma and triglyceride-glucose indices and monocyte/HDL-C ratios in colon cancer patients. J Pharm Res Int 33(46):213–217
Weinreb JC, Barentsz JO, Choyke PL et al (2016) PI-RADS prostate imaging–reporting and data system: 2015, version 2. Eur Urol 69:16–40
Wang F-M, Zhang Y (2019) High lipoprotein (a) level is independently associated with adverse clinicopathological features in patients with prostate cancer. Dis Markers 2019:9483935
Kotani K, Sekine Y, Ishikawa S, Ikpot IZ, Suzuki K, Remaley AT (2013) High-density lipoprotein and prostate cancer: an overview. J Epidemiol 23:313–319
Jamnagerwalla J, Howard LE, Allott EH et al (2018) Serum cholesterol and risk of high-grade prostate cancer: results from the REDUCE study. Prostate Cancer Prostatic Dis 21:252–259
Wolny-Rokicka EI, Tukiendorf A, Wydmański J, Zembroń-Łacny A (2017) The comparison and estimation of the prognostic value of lipid profiles in patients with prostate cancer depends on cancer stage advancement. Am J Mens Health 11:1745–1751
Wan F, Qin X, Zhang G et al (2015) Oxidized low-density lipoprotein is associated with advanced-stage prostate cancer. Tumour Biol 36:3573–3582
Funding
The author(s) received no financial support for the research or authorship.
Author information
Authors and Affiliations
Contributions
Conception and design: SS, AK, and ST; data acquisition: KC, EU, and MEP; data analysis and interpretation: YK, KC, and FS; drafting the manuscript: SS and AK; critical revision of the manuscript for scientific and factual content: MEP; statistical analysis: SS; supervision: EO.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Ethical approval
The present study protocol was reviewed and approved by the Institutional Review Board of Ankara City Hospital (approval number: E2-22-2396).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Senel, S., Ceviz, K., Kasap, Y. et al. Efficacy of plasma atherogenic index in predicting malignancy in the presence of Prostate Imaging–Reporting and Data System 3 (PI-RADS 3) prostate lesions. Int Urol Nephrol 55, 255–261 (2023). https://doi.org/10.1007/s11255-022-03409-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-022-03409-9



