Abstract
Purpose
Hyperuricemia is common among patients with chronic kidney disease (CKD). In the general population, hyperuricemia is associated with secondary hyperparathyroidism (SHPT), in a mechanism that involves vitamin D metabolism. Data for patients with CKD, however, are scarce. We aimed to evaluate the relationship between hyperuricemia and mineral and bone metabolism, particularly hyperparathyroidism.
Methods
This is a retrospective study that included 922 adult patients with stages 3, 4, or 5 CKD, not on dialysis. Clinical, demographic, and biochemical data were collected from charts and included uric acid, parathyroid hormone (PTH), 25(OH)-vitamin D, calcium, phosphate, renal function (estimated glomerular filtration rate—eGFR), and medications such as allopurinol, furosemide, and cholecalciferol. SHPT was defined as PTH > 65 pg/ml.
Results
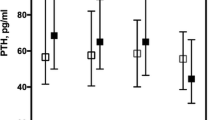
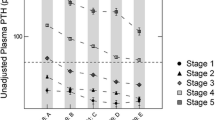
Our patients were mostly Caucasian women, with a mean age of 64 ± 16 years. SHPT and hyperuricemia were observed in 70% and 62.4% of patients, respectively. Patients with SHPT presented higher levels of uric acid (7.2 ± 1.8 vs. 6.6 ± 1.7 mg/dL, p = 0.0001) and a higher frequency of hyperuricemia (66% vs. 33%, p = 0.0001). Patients with hyperuricemia were mostly female, with lower eGFR, higher phosphate, and higher PTH. The risk of hypovitaminosis D was higher among patients with SHPT (69.7% vs. 53.1%, p = 0.0001). Hyperuricemia remained independently associated with hyperparathyroidism, (p = 0.033) even after adjustments for eGFR, calcium, phosphate, hypovitaminosis D, and use of allopurinol, calcitriol, furosemide, and cholecalciferol.
Conclusion
Hyperuricemia seems to be a contributing factor for SHPT in patients with CKD. The mechanisms behind this finding have yet to be elucidated.


Similar content being viewed by others
Availability of data and materials
Data are available for reviewers and authors if requested.
Abbreviations
- CKD:
-
Chronic kidney disease
- eGFR:
-
Estimated glomerular filtration rate
- FGF-23:
-
Fibroblast growth factor 23
- PTH:
-
Parathyroid hormone
- SHPT:
-
Secondary hyperparathyroidism
- UA:
-
Uric acid
References
Miyaoka T, Mochizuki T, Takei T, Tsuchiya K, Nitta K (2014) Serum uric acid levels and long-term outcomes in chronic kidney disease. Heart Vessel 29(4):504–512. https://doi.org/10.1007/s00380-013-0396-0
Jeon HJ, Oh J, Shin DH (2019) Urate-lowering agents for asymptomatic hyperuricemia in stage 3–4 chronic kidney disease: controversial role of kidney function. PLoS ONE 14(6):e0218510. https://doi.org/10.1371/journal.pone.0218510
Sampson AL, Singer RF, Walters GD (2017) Uric acid lowering therapies for preventing or delaying the progression of chronic kidney disease. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD009460.pub2
Doria A, Galecki AT, Spino C, Pop-Busui R, Cherney DZ, Lingvay I, Parsa A, Rossing P, Sigal RJ, Afkarian M, Aronson R, Caramori ML, Crandall JP, de Boer IH, Elliott TG, Goldfine AB, Haw JS, Hirsch IB, Karger AB, Maahs DM, McGill JB, Molitch ME, Perkins BA, Polsky S, Pragnell M, Robiner WN, Rosas SE, Senior P, Tuttle KR, Umpierrez GE, Wallia A, Weinstock RS, Wu C, Mauer M (2020) Group PS serum urate lowering with allopurinol and kidney function in type 1 diabetes. N Engl J Med 382(26):2493–2503. https://doi.org/10.1056/NEJMoa1916624
Badve SV, Pascoe EM, Tiku A, Boudville N, Brown FG, Cass A, Clarke P, Dalbeth N, Day RO, de Zoysa JR, Douglas B, Faull R, Harris DC, Hawley CM, Jones GRD, Kanellis J, Palmer SC, Perkovic V, Rangan GK, Reidlinger D, Robison L, Walker RJ, Walters G, Johnson DW, Investigators C-FS (2020) Effects of allopurinol on the progression of chronic kidney disease. N Engl J Med 382(26):2504–2513. https://doi.org/10.1056/NEJMoa1915833
Alemzadeh R, Kichler J (2016) Uric acid-induced Inflammation is mediated by the parathyroid hormone:25-hydroxyvitamin D ratio in obese adolescents. Metab Syndr Relat Disord 14(3):167–174. https://doi.org/10.1089/met.2015.0099
Ponvilawan B, Charoenngam N, Ungprasert P (2020) Primary hyperparathyroidism is associated with a higher level of serum uric acid: a systematic review and meta-analysis. Int J Rheum Dis 23(2):174–180. https://doi.org/10.1111/1756-185X.13740
Peng H, Li H, Li C, Chao X, Zhang Q, Zhang Y (2013) Association between vitamin D insufficiency and elevated serum uric acid among middle-aged and elderly Chinese Han women. PLoS ONE 8(4):e61159. https://doi.org/10.1371/journal.pone.0061159
Yilmaz H, Kaya M, Sahin M, Delibasi T (2012) Is vitamin D status a predictor glycaemic regulation and cardiac complication in type 2 diabetes mellitus patients? Diabetes Metab Syndr 6(1):28–31. https://doi.org/10.1016/j.dsx.2012.05.007
Kidney disease: improving global outcomes CKDMBDWG, (2009) KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl 113:S1-130. https://doi.org/10.1038/ki.2009.188
Vanholder R, Patel S, Hsu CH (1993) Effect of uric acid on plasma levels of 125(OH)2D in renal failure. J Am Soc Nephrol 4(4):1035–1038
Mohammed A, Marie MA, Abdulazim DO, Hassan M, Shaker O, Ayeldeen G, Salem MM, Sharaf El Din UA (2020) On behalf of the egyptian vascular calcification G serum urate lowering therapy using allopurinol improves serum 25 hydroxy vitamin D in Stage 3–5 CKD patients: a pilot study. Nephron. https://doi.org/10.1159/000512340
Andrews ES, Perrenoud L, Nowak KL, You Z, Pasch A, Chonchol M, Kendrick J, Jalal D (2018) Examining the effects of uric acid-lowering on markers vascular of calcification and CKD-MBD; a post-hoc analysis of a randomized clinical trial. PLoS ONE 13(10):e0205831. https://doi.org/10.1371/journal.pone.0205831
Kim Y, Kang J, Kim GT (2018) Prevalence of hyperuricemia and its associated factors in the general korean population: an analysis of a population-based nationally representative sample. Clin Rheumatol 37(9):2529–2538. https://doi.org/10.1007/s10067-018-4130-2
Zhu Y, Pandya BJ, Choi HK (2011) Prevalence of gout and hyperuricemia in the US general population: the national health and nutrition examination survey 2007–2008. Arthritis Rheum 63(10):3136–3141. https://doi.org/10.1002/art.30520
Song P, Wang H, Xia W, Chang X, Wang M, An L (2018) Prevalence and correlates of hyperuricemia in the middle-aged and older adults in China. Sci Rep 8(1):4314. https://doi.org/10.1038/s41598-018-22570-9
MacFarlane LA, Kim SC (2014) Gout: a review of nonmodifiable and modifiable risk factors. Rheum Dis Clin North Am 40(4):581–604. https://doi.org/10.1016/j.rdc.2014.07.002
Trifiro G, Morabito P, Cavagna L, Ferrajolo C, Pecchioli S, Simonetti M, Bianchini E, Medea G, Cricelli C, Caputi AP, Mazzaglia G (2013) Epidemiology of gout and hyperuricaemia in Italy during the years 2005–2009: a nationwide population-based study. Ann Rheum Dis 72(5):694–700. https://doi.org/10.1136/annrheumdis-2011-201254
Unger MD, Cuppari L, Titan SM, Magalhaes MC, Sassaki AL, dos Reis LM, Jorgetti V, Moyses RM (2010) Vitamin D status in a sunny country: where has the sun gone? Clin Nutr 29(6):784–788. https://doi.org/10.1016/j.clnu.2010.06.009
Machado H, Martins CSW, Jorgetti V, Elias RM, Moyses RMA (2020) Chronic kidney disease is a main confounding factor for 25-vitamin D measurement. J Bras Nefrol 42(1):94–98. https://doi.org/10.1590/2175-8239-jbn-2019-0053
Thakkinstian A, Anothaisintawee T, Chailurkit L, Ratanachaiwong W, Yamwong S, Sritara P, Ongphiphadhanakul B (2015) Potential causal associations between vitamin D and uric acid: bidirectional mediation analysis. Sci Rep 5:14528. https://doi.org/10.1038/srep14528
Toussaint ND, Ruderman I (2017) What is the role of vitamin D supplementation on vascular health in CKD? Clin J Am Soc Nephrol 12(9):1377–1379. https://doi.org/10.2215/CJN.07170717
Kaseda R, Hosojima M, Sato H, Saito A (2011) Role of megalin and cubilin in the metabolism of vitamin D(3). Ther Apher Dial 15(Suppl 1):14–17. https://doi.org/10.1111/j.1744-9987.2011.00920.x
Christensen EI, Willnow TE (1999) Essential role of megalin in renal proximal tubule for vitamin homeostasis. J Am Soc Nephrol 10(10):2224–2236
Negrea L (2019) Active vitamin D in chronic kidney disease: getting right back where we started from? Kidney Dis (Basel) 5(2):59–68. https://doi.org/10.1159/000495138
Pike JW, Christakos S (2017) Biology and mechanisms of action of the vitamin D hormone. Endocrinol Metab Clin North Am 46(4):815–843. https://doi.org/10.1016/j.ecl.2017.07.001
Dusso AS, Brown AJ (1998) Mechanism of vitamin D action and its regulation. Am J Kidney Dis 32(2 Suppl 2):S13-24. https://doi.org/10.1053/ajkd.1998.v32.pm9808140
Chen W, Roncal-Jimenez C, Lanaspa M, Gerard S, Chonchol M, Johnson RJ, Jalal D (2014) Uric acid suppresses 1 alpha hydroxylase in vitro and in vivo. Metabolism 63(1):150–160. https://doi.org/10.1016/j.metabol.2013.09.018
Scott JT, Dixon AS, Bywaters EG (1964) Association of hyperuricaemia and gout with hyperparathyroidism. Br Med J 1(5390):1070–1073. https://doi.org/10.1136/bmj.1.5390.1070
Valdemarsson S, Lindblom P, Bergenfelz A (1998) Metabolic abnormalities related to cardiovascular risk in primary hyperparathyroidism: effects of surgical treatment. J Intern Med 244(3):241–249. https://doi.org/10.1046/j.1365-2796.1998.00366.x
Miller PD, Schwartz EN, Chen P, Misurski DA, Krege JH (2007) Teriparatide in postmenopausal women with osteoporosis and mild or moderate renal impairment. Osteoporos Int 18(1):59–68. https://doi.org/10.1007/s00198-006-0189-8
Hashimoto Y, Wakayama N, Miyauchi A (2007) Therapeutic agents for disorders of bone and calcium metabolism–teriparatide, parathyroid hormone (1–34) as a daily subcutaneous injection. Clin Calcium 17(1):48–55
Hui JY, Choi JW, Mount DB, Zhu Y, Zhang Y, Choi HK (2012) The independent association between parathyroid hormone levels and hyperuricemia: a national population study. Arthritis Res Ther 14(2):R56. https://doi.org/10.1186/ar3769
Chin KY, Nirwana SI, Ngah WZ (2015) Significant association between parathyroid hormone and uric acid level in men. Clin Interv Aging 10:1377–1380. https://doi.org/10.2147/CIA.S90233
Acknowledgements
We would like to thank Fatima Libanio for her support in data collection from the electronic chart.
Funding
The author(s) RMAM and RME disclosed receipt of support from CNPQ (Conselho Nacional de Desenvolvimento Científico e Tecnológico), grant numbers 303545/2020–8 and 305106/2018–0, respectively). This financial support had no role in study design; collection, analysis, and interpretation of data; writing the report; and the decision to submit the report for publication.
Author information
Authors and Affiliations
Contributions
TEC and RME conceived the idea; TEC and JCL collected the data; RMAM and RME supervised the research; TEC, JCL, and VAC give important intellectual contribution; all authors interpreted the data and discussed the results; TEC, RMAM, and RME analyzed the data; TEC and RME drafted the manuscript; all authors read and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
Ethical approval for this study was obtained from *Comissão de Ética para análise de projetos de pesquisa—CAPpesq (45163715.4.0000.0068).
Consent to participate
Written informed consent was not obtained from subjects since was waived by the Research Ethical Board.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Costa, T.E.M., Lauar, J.C., Innecchi, M.L.R. et al. Hyperuricemia is associated with secondary hyperparathyroidism in patients with chronic kidney disease. Int Urol Nephrol 54, 2255–2261 (2022). https://doi.org/10.1007/s11255-022-03116-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-022-03116-5