Abstract
Background
Gremlin1 belongs to the superfamily members of transforming growth factor (TGF)-β1, playing a profibrotic role in chronic kidney diseases (CKD) and the transition from the late stage of acute kidney injury (AKI) to CKD, but the effect it plays in the early stage of AKI is unclear. This study aimed to investigate the role of Gremlin1on apoptosis in renal tubular epithelial cells under ischemia–reperfusion (I/R) induction.
Methods
We detected Gremlin1 and TGF-β1 expression in the kidneys of mice undergoing renal ischemia–reperfusion injury bilaterally. We induced apoptosis through depletion and reperfusion of oxygen and serum in human kidney tubular epithelial cells (HKCs), mimicking I/R injury in vivo, and detected the role and pathways of Gremlin1 and TGF-β1on HKCs injury.
Results
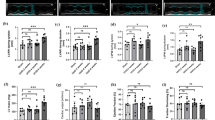
Mice undergoing bilateral I/R surgery presented AKI with a significant increase in serum creatinine, obvious renal tubular injuries, and increased macrophage cell and T-cell infiltration in interstitial areas. Gremlin1 expression was significantly increased along with TGF-β1 in the kidneys of AKI mice compared to sham mice. Exogenous Gremlin1 inhibited I/R-induced caspase3 expression in HKCs, which was blocked by a VEGFR2 kinase inhibitor III (SU5416). TGF-β1 also inhibited I/R-induced cell apoptosis in HKCs but had no synergic effect with Gremlin1. The TGF-β1’s inhibitory effect could be blocked by the TGF-β1 type I receptor (activin receptor-like kinase 5, and ALK5)-specific inhibitor SB431542.
Conclusions
Gremlin1 and TGF- β1 protect kidney tubular epithelial cells from ischemia–reperfusion-induced apoptosis through VEGFR2 and Smad2 signaling pathways.







Similar content being viewed by others
References
Che M, Li Y, Liang X, Xie B, Xue S, Qian J et al (2011) Prevalence of acute kidney injury following cardiac surgery and related risk factors in Chinese patients. Nephron Clin Pract 117(4):c305–c311
Hobson CE, Yavas S, Segal MS, Schold JD, Tribble CG, Layon AJ et al (2009) Acute kidney injury is associated with increased long-term mortality after cardiothoracic surgery. Circulation 119(18):2444–2453
Karkouti K, Wijeysundera DN, Yau TM, Callum JL, Cheng DC, Crowther M et al (2009) Acute kidney injury after cardiac surgery: focus on modifiable risk factors. Circulation 119(4):495–502
Oeal JB, Shaw AD, Billings FT (2016) Acute kidney injury following cardiac surgery: current understanding and future directions. Crit Care 20(1):187
Negi S, Koreeda D, Kobayashi S, Yano T, Tatsuta K, Mima T et al (2018) Acute kidney injury: epidemiology, outcomes, complications, and therapeutic strategies. Semin Dial 31(5):519–527
Bonventre JV, Weinberg JM (2003) Recent advances in the pathophysiology of ischemic acute renal failure. J Am Soc Nephrol 14(8):2199–2210
Sharfuddin AA, Molitoris BA (2011) Pathophysiology of ischemic acute kidney injury. Nat Rev Nephrol 7(4):189–200
Bove T, Zangrillo A, Guarracino F, Alvaro G, Persi B, Maglioni E et al (2014) Effect of fenoldopam on use of renal replacement therapy among patients with acute kidney injury after cardiac surgery: a randomized clinical trial. JAMA 312(21):2244–2253
Michos O, Panman L, Vintersten K, Beier K, Zeller R, Zuniga A (2004) Gremlin-mediated BMP antagonism induces the epithelial-mesenchymal feedback signaling controlling metanephric kidney and limb organogenesis. Development 131(14):3401–3410
Marquez-Exposito L, Lavoz C, Rodrigues-Diez RR, Rayego-Mateos S, Orejudo M, Cantero-Navarro E et al (2018) Gremlin regulates tubular epithelial to mesenchymal transition via VEGFR2: potential role in renal fibrosis. Front Pharmacol 9:1195
Carvajal G, Droguett A, Burgos ME, Aros C, Ardiles L, Flores C et al (2008) Gremlin: a novel mediator of epithelial mesenchymal transition and fibrosis in chronic allograft nephropathy. Transpl Proc 40(3):734–739
Marchant V, Droguett A, Valderrama G, Burgos ME, Carpio D, Kerr B et al (2015) Tubular overexpression of Gremlin in transgenic mice aggravates renal damage in diabetic nephropathy. Am J Physiol Renal Physiol 309(6):F559–F568
Wang XB, Zhu H, Song W, Su JH (2018) Gremlin regulates podocyte apoptosis via transforming growth factor-beta (TGF-beta) pathway in diabetic nephropathy. Med Sci Monit Int Med J Exp Clin Res 24:183–189
Staloch D, Gao X, Liu K, Xu M, Feng X, Aronson JF et al (2015) Gremlin is a key pro-fibrogenic factor in chronic pancreatitis. J Mol Med 93(10):1085–1093
Bhatia V, Cao Y, Ko TC, Falzon M (2016) Parathyroid hormone-related protein interacts with the transforming growth factor-beta/bone morphogenetic protein-2/gremlin signaling pathway to regulate proinflammatory and profibrotic mediators in pancreatic acinar and stellate cells. Pancreas 45(5):659–670
Myllarniemi M, Lindholm P, Ryynanen MJ, Kliment CR, Salmenkivi K, Keski-Oja J et al (2008) Gremlin-mediated decrease in bone morphogenetic protein signaling promotes pulmonary fibrosis. Am J Respir Crit Care Med 177(3):321–329
Church RH, Ali I, Tate M, Lavin D, Krishnakumar A, Kok HM et al (2017) Gremlin1 plays a key role in kidney development and renal fibrosis. Am J Physiol Renal Physiol 312(6):F1141–F1157
Ma T, Huang C, Xu Q, Yang Y, Liu Y, Meng X et al (2017) Suppression of BMP-7 by histone deacetylase 2 promoted apoptosis of renal tubular epithelial cells in acute kidney injury. Cell Death Dis 8(10):3139
Ma T, Huang C, Meng X, Li X, Zhang Y, Ji S et al (2016) A potential adjuvant chemotherapeutics, 18beta-glycyrrhetinic acid, inhibits renal tubular epithelial cells apoptosis via enhancing BMP-7 epigenetically through targeting HDAC2. Sci Rep 6:25396
Wei R, Zhang R, Xie Y, Shen L, Chen F (2015) Hydrogen suppresses hypoxia/reoxygenation-induced cell death in hippocampal neurons through reducing oxidative stress. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharm 36(2):585–598
Brooks C, Wei Q, Cho SG, Dong Z (2009) Regulation of mitochondrial dynamics in acute kidney injury in cell culture and rodent models. J Clin Investig 119(5):1275–1285
Gao X, Cao Y, Staloch DA, Gonzales MA, Aronson JF, Chao C et al (2014) Bone morphogenetic protein signaling protects against cerulein-induced pancreatic fibrosis. PLoS ONE 9(2):e89114
Dolan V, Murphy M, Sadlier D, Lappin D, Doran P, Godson C et al (2005) Expression of gremlin, a bone morphogenetic protein antagonist, in human diabetic nephropathy. Am J Kidney Dis 45(6):1034–1039
Mezzano S, Droguett A, Lavoz C, Krall P, Egido J, Ruiz-Ortega M (2018) Gremlin and renal diseases: ready to jump the fence to clinical utility? Nephrol Dial Transplant 33(5):735–741
Droguett A, Krall P, Burgos ME, Valderrama G, Carpio D, Ardiles L et al (2014) Tubular overexpression of gremlin induces renal damage susceptibility in mice. PLoS ONE 9(7):e101879
Rodrigues-Diez R, Lavoz C, Carvajal G, Rayego-Mateos S, Rodrigues Diez RR, Ortiz A et al (2012) Gremlin is a downstream profibrotic mediator of transforming growth factor-beta in cultured renal cells. Nephron Exp Nephrol 122(1–2):62–74
Rodrigues-Diez R, Rodrigues-Diez RR, Lavoz C, Carvajal G, Droguett A, Garcia-Redondo AB et al (2014) Gremlin activates the Smad pathway linked to epithelial mesenchymal transdifferentiation in cultured tubular epithelial cells. Biomed Res Int 2014:802841
Molitoris BA (1999) Acute renal failure. Drugs Today (Barc) 35(9):659–666
Havasi A, Borkan SC (2011) Apoptosis and acute kidney injury. Kidney Int 80(1):29–40
Lavoz C, Alique M, Rodrigues-Diez R, Pato J, Keri G, Mezzano S et al (2015) Gremlin regulates renal inflammation via the vascular endothelial growth factor receptor 2 pathway. J Pathol 236(4):407–420
Priante G, Gianesello L, Ceol M, Del Prete D, Anglani F (2019) Cell death in the kidney. Int J Mol Sci 20(14):3598
Brazil DP, Church RH, Surae S, Godson C, Martin F (2015) BMP signalling: agony and antagony in the family. Trends Cell Biol 25(5):249–264
Farkas L, Farkas D, Gauldie J, Warburton D, Shi W, Kolb M (2011) Transient overexpression of Gremlin results in epithelial activation and reversible fibrosis in rat lungs. Am J Respir Cell Mol Biol 44(6):870–878
Soofi A, Zhang P, Dressler GR (2013) Kielin/chordin-like protein attenuates both acute and chronic renal injury. J Am Soc Nephrol 24(6):897–905
Liao WJ, Lin H, Cheng CF, Ka SM, Chen A, Yang RB (2019) SCUBE1-enhanced bone morphogenetic protein signaling protects against renal ischemia-reperfusion injury. Biochim Biophys Acta 1865(2):329–338
Zhen-Qiang F, Bing-Wei Y, Yong-Liang L, Xiang-Wei W, Shan-Hong Y, Yuan-Ning Z et al (2012) Localized expression of human BMP-7 by BM-MSCs enhances renal repair in an in vivo model of ischemia-reperfusion injury. Genes Cells Devoted Mol Cell Mech 17(1):53–64
Ji C, Huang JW, Xu QY, Zhang J, Lin MT, Tu Y et al (2016) Gremlin inhibits UV-induced skin cell damages via activating VEGFR2-Nrf2 signaling. Oncotarget 7(51):84748–84757
Huang H, Huang H, Li Y, Liu M, Shi Y, Chi Y et al (2013) Gremlin induces cell proliferation and extra cellular matrix accumulation in mouse mesangial cells exposed to high glucose via the ERK1/2 pathway. BMC Nephrol 14:33
Guan Q, Nguan CY, Du C (2010) Expression of transforming growth factor-beta1 limits renal ischemia-reperfusion injury. Transplantation 89(11):1320–1327
Yang Q, Ren GL, Wei B, Jin J, Huang XR, Shao W et al (2019) Conditional knockout of TGF-betaRII /Smad2 signals protects against acute renal injury by alleviating cell necroptosis, apoptosis and inflammation. Theranostics 9(26):8277–8293
Acknowledgements
The authors would like to thank Wei Cui, Sa Liu, and Limin Zhao at the Beijing Institute of Heart, Lung and Blood Vessel Diseases for technical support.
Funding
This work was supported by a grant from Beijing Municipal Natural Science Foundation (7162055).
Author information
Authors and Affiliations
Contributions
XG: experimental design, data generation and interpretation, manuscript draft, revision, approval, and submission. LH: experimental design, data generation, and interpretation. XY: experimental design, data generation, and interpretation. LM: experimental design, data interpretation, manuscript revision, and approval.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, X., Han, L., Yao, X. et al. Gremlin1 and TGF-β1 protect kidney tubular epithelial cells from ischemia–reperfusion injury through different pathways. Int Urol Nephrol 54, 1311–1321 (2022). https://doi.org/10.1007/s11255-021-03010-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-021-03010-6