Abstract
Purpose
The aim of this study was to determine the frequency and the risk factors of acute and chronic nephrotoxicity in patients who received cisplatin due to malignancy.
Materials and methods
Medical records of all patients who received cisplatin-based chemotherapy regimen between January 2013 and July 2019 were retrospectively evaluated. The data of 203 patients who met the study criteria were examined. The patients were evaluated for acute nephrotoxicity at 48 h and late nephrotoxicity at 3rd month after first course of cisplatin. Early and late nephrotoxicity were defined by NCI CTCAE Version 4.0 criteria.
Results
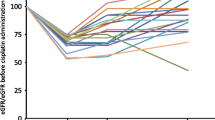
The mean age of the study patients was 56.44 ± 12.69 years, 78.8% were males and 21.2% were females. It is revealed that the incidence of cisplatin-induced acute nephrotoxicity was 9.2% and chronic nephrotoxicity was 37.9%. While the development of acute nephrotoxicity was associated with female gender, history of diabetes mellitus, history of ischemic heart disease and use of antiplatelet drug, the development of chronic nephrotoxicity was associated with older age, female gender and using of diuretics. High serum creatinine, urea and low eGFR value before treatment were found to be associated with both early and late nephrotoxicity (p < 0.05). There was no statistically significant relationship between acute or chronic nephrotoxicity and cumulative dose of cisplatin, hydration or intravenous magnesium supplementation.
Conclusion
High initial serum creatinine value and low initial eGFR are the most important determinants of both early and late nephrotoxicity.
Similar content being viewed by others
References
Barton CD, Pizer B, Jones C, Oni L, Pirmohamed M, Hawcutt DB (2018) Identifying cisplatin-induced kidney damage in paediatric oncology patients. Pediatr Nephrol 33(9):1467–1474
Hanigan MH, Devarajan P (2003) Cisplatin nephrotoxicity: molecular mechanisms. Cancer Ther 1:47–61
Almanric K, Marceau N, Cantin A, Bertin É (2017) Risk factors for nephrotoxicity associated with cisplatin. Can J Hosp Pharm 70(2):99–106
Bodnar L, Wcisle G, Gasowska-Bodnar A, Synowies A, Szarlec-Wcislo K, Szczylik C (2008) Renal protection with magnesium subcarbonate and magnesium sulphate in patients with epithelial ovarian cancer after cisplatin and paclitaxel chemotherapy: a randomised phase II study. Eur J Cancer 44:2608–2614
US Department of Health and Human Services: Common Terminology Criteria for Adverse Events (CTCAE version) 4.0 (2020). https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE-4.03-JUNE
Miyoshi T, Misumi N, Hiraike M, Mihara Y, Nishino T, Tsuruta M et al (2016) Risk factors associated with cisplatin-induced nephrotoxicity in patients with advanced lung cancer. Biol Pharm Bull 39:2009–2014
Motwani SS, McMahon GM, Humphreys BD, Partridge AH, Waikar SS, Curhan GC (2018) Development and validation of a risk prediction model for acute kidney injury after the first course of cisplatin. J Clin Oncol 36:682–688
Lavole A, Daniel S, Baudrin L, Gounant V, Ruppert AM, Epaud C et al (2012) Routine administration of a single dose of cisplatin ≥ 75 mg/m2 after short hydration in an outpatient lung-cancer clinic. Bull Cancer 99:E43-348
Latcha S, Jaimes EA, Patil S, Glezerman IG, Mehta S, Flombaum CD (2016) Long-term renal outcomes after cisplatin treatment. Clin J Am Soc Nephrol 11:1173–1179
De Jongh FE, Van Veen RN, Veltman SJ, De Wit R, Van der Burg ME, Van den Bent MJ et al (2003) Weekly high-dose cisplatin is a feasible treatment option: analysis on prognostic factors for toxicity in 400 patients. Br J Cancer 88:1199–1206
Faig J, Haughton M, Taylor RC, D’Agostino RB Jr, Whelen MJ, Porosnicu Rodriguez KA et al (2018) Retrospective analysis of cisplatin nephrotoxicity in patients with head and neck cancer receiving outpatient treatment with concurrent high-dose cisplatin and radiotherapy. Am J Clin Oncol 41:432–440
De Jongh FE, Verweij J, Loos WJ, De Wit R, De Jonge MJ, Planting AS et al (2001) Body-surface area-based dosing does not increase accuracy of predicting cisplatin exposure. J Clin Oncol 19:3733–3739
Luyckx VA, Shukha K, Brenner BM (2011) Low nephron number and its clinical consequences. Rambam Maimonides Med J 2:e0061
Liu JQ, Cai GY, Wang SY, Song YH, Xia YY, Liang S et al (2018) The characteristics and risk factors for cisplatin-induced acute kidney injury in the elderly. Ther Clin Risk Manag 14:1279–1285
Wen J, Zeng M, Shu Y, Guo D, Sun Y, Guo Z et al (2015) Aging increases the susceptibility of cisplatin-induced nephrotoxicity. Age (Dordr) 37:112
Rosner MH (2013) Acute kidney injury in the elderly. Clin Geriatr Med 29:565–578
Ulldemolins M, Roberts JA, Rello J, Paterson DL, Lipman J (2011) The effects of hypoalbuminaemia on optimizing antibacterial dosing in critically ill patients. Clin Pharmacokinet 50:99–110
van der Vorst MJDL, Neefjes ECW, Toffoli EC, Oosterling-Jansen JEW, Vergeer MR, Leemans CR et al (2019) Incidence and risk factors for acute kidney injury in head and neck cancer patients treated with concurrent chemoradiation with high-dose cisplatin. BMC Cancer 19:1066
Mizuno T, Ishikawa K, Sato W, Koike T, Kushida M, Miyagawa Y et al (2013) The risk factors of severe acute kidney injury induced by cisplatin. Oncology 85:364–369
Bhat ZY, Cadnapaphornchai P, Ginsburg K, Sivagnanam M, Chopra S, Treadway CK et al (2015) Understanding the risk factors and long-term consequences of cisplatin-associated acute kidney injury: an observational cohort study. PLoS ONE 10:e0142225
Thakar CV, Christianson A, Himmelfarb J, Leonard AC (2011) Acute kidney injury episodes and chronic kidney disease risk in diabetes mellitus. Clin J Am Soc Nephrol 6:2567–2572
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kamacı, Ş.Ç., Koçak, G., Yeşilova, A. et al. Evaluation of acute and chronic nephrotoxicity in patients received cisplatin-based chemotherapy: has anything changed over time?. Int Urol Nephrol 54, 1085–1090 (2022). https://doi.org/10.1007/s11255-021-02975-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-021-02975-8