Abstract
Purpose
Progressive decline in lean mass and gain of fat mass are common in patients treated with peritoneal dialysis (PD). It is unclear whether body composition or its longitudinal changes contribute to increased mortality among prevalent PD patients.
Methods
This was a retrospective cohort study. Body composition was assessed using bioelectrical impedance spectroscopy (BIS). Lean and fat tissue indices were derived from lean and fat mass indexed to height-squared, respectively. The patient’s baseline BIS results were used to explore its mortality risk prediction for the entire cohort. Among patients with subsequent BIS measurements, changes of lean and fat mass over time were also examined with survival outcomes.
Results
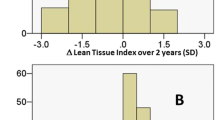
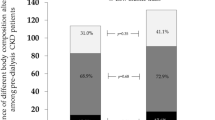
Among all participants (n = 555, PD vintage 38 months), higher baseline lean tissue index was associated with lower mortality after adjusting for fat tissue index and confounders (HR 0.90; 95% CI 0.84–0.97, p = 0.01). However, this association was no longer significance after the final adjustment with serum albumin level (p = 0.06). A total of 136 patients had repeated BIS data. After the interval of 10.1 months during two consecutive BIS measurements, there was a strong inverse correlation between the percentage changes of lean and fat tissue indices (r = − 0.73, p < 0.001). The longitudinal changes in lean mass, either high or low categories, were not significantly associated with all-cause mortality. In contrast, patients who were classified as having low values of fat tissue index (below median) from baseline to the next BIS measurements had a lower odds of death in the univariable (HR 0.32; 95% CI 0.12–0.84, p = 0.02) but not in the adjusted models.
Conclusion
Among prevalent PD patients, higher baseline lean mass was independently associated with better survival. However, the longitudinal changes in lean mass were not significantly associated with mortality. In contrast, the maintenance of low fat status over time appeared to be associated with a lower likelihood of death among PD population.



Similar content being viewed by others
References
Johansen KL, Lee C (2015) Body composition in chronic kidney disease. Curr Opin Nephrol Hypertens 24:268–275
Doshi M, Streja E, Rhee CM, Park J, Ravel VA, Soohoo M et al (2016) Examining the robustness of the obesity paradox in maintenance hemodialysis patients: a marginal structural model analysis. Nephrol Dial Transplant 31:1310–1319
Ahmadi SF, Zahmatkesh G, Streja E, Mehrotra R, Rhee CM, Kovesdy CP et al (2016) Association of body mass index with mortality in peritoneal dialysis patients: a systematic review and meta-analysis. Perit Dial Int 36:315–325
Liu J, Zeng X, Hong HG, Li Y, Fu P (2017) The association between body mass index and mortality among Asian peritoneal dialysis patients: a meta-analysis. PLoS ONE 12:e0172369
Carrero JJ, Johansen KL, Lindholm B, Stenvinkel P, Cuppari L, Avesani CM (2016) Screening for muscle wasting and dysfunction in patients with chronic kidney disease. Kidney Int 90:53–66
Carrero JJ, Stenvinkel P, Cuppari L, Ikizler TA, Kalantar-Zadeh K, Kaysen G et al (2013) Etiology of the protein-energy wasting syndrome in chronic kidney disease: a consensus statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J Ren Nutr 23:77–90
Lo WK (2016) Metabolic syndrome and obesity in peritoneal dialysis. Kidney Res Clin Pract 35:10–14
Marcelli D, Usvyat LA, Kotanko P, Bayh I, Canaud B, Etter M et al (2015) Body composition and survival in dialysis patients: results from an international cohort study. Clin J Am Soc Nephrol 10:1192–1200
Zhang H, Tao X (2019) Evaluation of body composition monitoring for assessment of nutritional status in hemodialysis patients. Ren Fail 41:377–383
Parthasarathy R, Oei E, Fan SL (2019) Clinical value of body composition monitor to evaluate lean and fat tissue mass in peritoneal dialysis. Eur J Clin Nutr 73:1520–1528
Kim C, Kim JK, Lee HS, Kim SG, Song YR (2020) Longitudinal changes in body composition are associated with all-cause mortality in patients on peritoneal dialysis. Clin Nutr. https://doi.org/10.1016/j.clnu.2020.04.034
Pellicano R, Strauss BJ, Polkinghorne KR, Kerr PG (2011) Longitudinal body composition changes due to dialysis. Clin J Am Soc Nephrol 6:1668–1675
Bergström J, Heimbürger O, Lindholm B (1998) Calculation of the protein equivalent of total nitrogen appearance from urea appearance. Which formulas should be used? Perit Dial Int 18:467–473
Kopple JD, Wolfson M, Chertow GM (2000) K/DOQI nutrition in chronic renal failure. Am J Kidney Dis 35(6 Suppl 2):s17–s18
Gilmore J (2006) KDOQI Clinical practice guidelines and clinical practice recommendations for 2006 updates: hemodialysis adequacy, peritoneal dialysis adequacy and vascualr access. Am J Kidney Dis 48(suppl 1):S1–S322
Park J, Mehrotra R, Rhee CM, Molnar MZ, Lukowsky LR, Patel SS et al (2013) Serum creatinine level, a surrogate of muscle mass, predicts mortality in peritoneal dialysis patients. Nephrol Dial Transplant 28:2146–2155
Churchill DN, Thorpe KE, Nolph KD, Keshaviah PR, Oreopoulos DG. Adequacy of dialysis and nutrition in continuous peritoneal dialysis: association with clinical outcomes Canada-USA (CANUSA) Peritoneal Dialysis Study Group. J Am Soc Nephrol 1996; 7: 198–207.
Malgorzewicz S, Chmielewski M, Kaczkan M, Borek P, Lichodziejewska-Niemierko M, Rutkowski B (2016) Nutritional predictors of mortality in prevalent peritoneal dialysis patients. Acta Biochim Pol 63:111–115
Ikizler TA, Burrowes JD, Byham-Gray LD, Campbell KL, Carrero JJ, Chan W, et al. KDOQI Nutrition in CKD Guideline Work Group. KDOQI clinical practice guideline for nutrition in CKD: 2020 update. Am J Kidney Dis 2020;76(3):S1-S107.
Carrero JJ, Wanner C (2016) clinical monitoring of protein-energy wasting in chronic kidney disease: moving from body size to body composition. J Ren Nutr 26:63–64
Marrón B, Remón C, Pérez-Fontán M, Quirós P, Ortíz A. Benefits of preserving residual renal function in peritoneal dialysis. Kidney Int Suppl. 2008; 1: S42–51.
Mehrotra R, Duong U, Jiwakanon S, Kovesdy CP, Moran J, Kopple JD et al (2011) Serum albumin as a predictor of mortality in peritoneal dialysis: comparisons with hemodialysis. Am J Kidney Dis 58:418–428
Choi SJ, Kim NR, Hong SA, Lee WB, Park MY, Kim JK et al (2011) Changes in body fat mass in patients after starting peritoneal dialysis. Perit Dial Int 31:67–73
Choi SJ, Kim EJ, Park MY, Kim JK, Hwang SD (2014) Does body fat mass define survival in patients starting peritoneal dialysis? Perit Dial Int 34:376–382
Kim JK, Park HC, Song YR, Kim HJ, Moon SJ, Kim SG (2019) Effects of excessive body fat accumulation on long-term outcomes during peritoneal dialysis. Perit Dial Int 39:268–275
Guida B, Trio R, Di Maro M, Memoli A, Di Lauro T, Belfiore A et al (2019) Prevalence of obesity and obesity-associated muscle wasting in patients on peritoneal dialysis. Nutr Metab Cardiovasc Dis 29:1390–1399
Honda H, Qureshi AR, Axelsson J, Heimburger O, Suliman ME, Barany P et al (2007) Obese sarcopenia in patients with end-stage renal disease is associated with inflammation and increased mortality. Am J Clin Nutr 86:633–638
Mehrotra R (2013) Nutritional issues in peritoneal dialysis patients: how do they differ from that of patients undergoing hemodialysis? J Ren Nutr 23:237–240
Caron-Lienert RS, Figueiredo AE, da Costa BP, Bombardelli CF, Pizzato AC, Conti A et al (2014) Evaluation of body composition and fluid volume using a body composition monitor: does intraperitoneal fluid matter? Perit Dial Int 34:456–458
Funding
None.
Author information
Authors and Affiliations
Contributions
Conceptualization: PK; data curation: TK, PC, and SK; formal analysis and interpretation: PK, KP, KT, and TK; writing original draft and editing: PK; supervision and mentorship: SE. Each author contributed important intellectual content during manuscript drafting. All the authors made substantial contributions, read, and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
This work has been made possible in part by a Special Task Force for Activating Research (STAR) in Renal Nutrition, Chulalongkorn University funded grant to Dr. Kittiskulnam. Dr. Kanjanabuch reports grants from National Research Council of Thailand, grants from Rachadaphiseksompot Endorsement Fund, Chulalongkorn University, grants from Thailand Research Foundation, outside the submitted work and receiving consultancy fees from VISTERRA as a country investigator and speaking honoraria from Astra Zeneca and Baxter Healthcare. All the authors declare no conflict of interests.
Ethical approval
This study was performed in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards (IRB No. 406/63). This article does not contain any data obtained directly from human (only document extraction) and there were no additional clinical and laboratory studies in the included patients. Informed consent was not obtained in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kittiskulnam, P., Chuengsaman, P., Katesomboon, S. et al. Association analysis of body composition with survival among prevalent peritoneal dialysis patients. Int Urol Nephrol 54, 437–446 (2022). https://doi.org/10.1007/s11255-021-02923-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-021-02923-6