Abstract
Purpose
The study aimed to examine effect of chlorhexidine dressing in the prevention of peritoneal dialysis (PD)-related infection in PD patients.
Methods
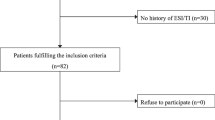
This single-center pilot study recruited 50 incident PD patients to use weekly chlorhexidine dressing. The primary outcome was exit-site/tunnel infection rate. Secondary outcomes were peritonitis rate, time to first PD-related infection, infection-related hospitalization, technique and patient survival, adverse events, and chlorhexidine dressing acceptability. These clinical outcomes were compared with those of a historical cohort (2016–2017) using daily gentamicin cream.
Results
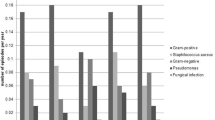
A total of 50 patients were recruited to use chlorhexidine and followed up for one year. The exit-site/tunnel infection rate was 0.09 (95% confidence interval [CI] 0.02–0.22) and peritonitis rate was 0.07 (95% CI 0.01–0.19) episodes per patient-year with chlorhexidine dressing. The 1-year infection-free survival rates for exit-site/tunnel infection and peritonitis were 92% and 94%, respectively. The 1-year technique and patient survival rates were 86% and 96%, respectively. Overall, 12% of participants developed localized contact dermatitis with chlorhexidine. Most participants (73%) reported that chlorhexidine dressings were very acceptable. Compared to a historical cohort using gentamicin cream (n = 238), the chlorhexidine group had similar exit-site/tunnel infection rates (incidence rate ratio [IRR] 0.65, 95% CI 0.22–1.92) but had lower peritonitis rates (IRR 0.24, 95% CI 0.07–0.77), and lower PD infection-related hospitalization rates (IRR 0.21, 95% CI 0.06–0.69) after adjusting for age, sex, race, primary kidney disease, and diabetes mellitus.
Conclusion
Weekly chlorhexidine dressing was associated with acceptable PD-related infection outcomes and was well-accepted by patients, although had a 12% rate of delayed localized contact dermatitis.
Trial registration number and date
The study was registered under www.clinicaltrials.gov with the reference number of NCT03406520 on 23 January 2018.





Similar content being viewed by others
Data availability
Data for this study is available from the corresponding author on appropriate request.
Code availability
Not applicable.
References
Kavanagh D, Prescott GJ, Mactier RA (2004) Peritoneal dialysis-associated peritonitis in Scotland (1999–2002). Nephrol Dial Transplant 19(10):2584–2591
Manera KE, Johnson DW, Craig JC, Shen JI, Ruiz L, Wang AY-M et al (2019) Patient and caregiver priorities for outcomes in peritoneal dialysis. Clin J Am Soc Nephrol. 14(1):74–83
Manera KE, Tong A, Craig JC, Shen J, Jesudason S, Cho Y et al (2019) An international Delphi survey helped develop consensus-based core outcome domains for trials in peritoneal dialysis. Kidney Int 96(3):699–710
Mehrotra R, Singh H (2013) Peritoneal dialysis-associated peritonitis with simultaneous exit-site infection. Clin J Am Soc Nephrol 8(1):126–130
van Diepen ATN, Tomlinson GA, Jassal SV (2012) The association between exit site infection and subsequent peritonitis among peritoneal dialysis patients. Clin J Am Soc Nephrol 7(8):1266–1271
Li PKT, Szeto CC, Piraino B, de Arteaga J, Fan S, Figueiredo AE et al (2016) ISPD peritonitis recommendations: 2016 update on prevention and treatment. Perit Dial Int. 36(5):481–508
Babu T, Rekasius V, Parada JP, Schreckenberger P, Challapalli M (2009) Mupirocin resistance among methicillin-resistant Staphylococcus aureus-colonized patients at admission to a tertiary care medical center. J Clin Microbiol 47(7):2279–2280
Annigeri R, Conly J, Vas S, Dedier H, Prakashan KP, Bargman JM et al (2001) Emergence of mupirocin-resistant Staphylococcus aureus in chronic peritoneal dialysis patients using mupirocin prophylaxis to prevent exit-site infection. Perit Dial Int. 21(6):554–559
Hon PY, Koh TH, Tan TY, Krishnan P, Leong JW-Y, Jureen R et al (2014) Changing molecular epidemiology and high rates of mupirocin resistance among meticillin-resistant Staphylococcus aureus in Singaporean hospitals. J Glob Antimicrob Resist. 2(1):53–55
Al-Hwiesh AK, Abdul-Rahman IS, Al-Muhanna FA, Al-Sulaiman MH, Al-Jondebi MS, Divino-Filho JC (2013) Prevention of peritoneal dialysis catheter infections in Saudi peritoneal dialysis patients: the emergence of high-level mupirocin resistance. Int J Artif Organs 36(7):473–483
Nessim SJ, Jassal SV (2012) Gentamicin-resistant infections in peritoneal dialysis patients using topical gentamicin exit-site prophylaxis: a report of two cases. Perit Dial Int 32(3):339
Pierce DA, Williamson JC, Mauck VS, Russell GB, Palavecino E, Burkart JM (2012) The effect on peritoneal dialysis pathogens of changing topical antibiotic prophylaxis. Perit Dial Int 32(5):525–530
Weinstein RA, Milstone AM, Passaretti CL, Perl TM (2008) Chlorhexidine: expanding the armamentarium for infection control and prevention. Clin Infect Dis 46(2):274–281
Russell AD, Day MJ (1993) Antibacterial activity of chlorhexidine. J Hosp Infect 25(4):229–238
Timsit J-F, Schwebel C, Bouadma L, Geffroy A, Garrouste-Orgeas M, Pease S et al (2009) Chlorhexidine-impregnated sponges and less frequent dressing changes for prevention of catheter-related infections in critically Ill adults. JAMA 301(12):1231
Wei L, Li Y, Li X, Bian L, Wen Z, Li M (2019) Chlorhexidine-impregnated dressing for the prophylaxis of central venous catheter-related complications: a systematic review and meta-analysis. BMC Infect Dis 19(1):429
Safdar N, O’Horo JC, Ghufran A, Bearden A, Didier ME, Chateau D et al (2014) Chlorhexidine-impregnated dressing for prevention of catheter-related bloodstream infection: a meta-analysis*. Crit Care Med 42(7):1703–1713
Shen K, Cho Y, Pascoe EM, Hawley CM, Oliver V, Hughes KM et al (2017) The SIESTA trial: a randomized study investigating the efficacy, safety, and tolerability of acupressure versus sham therapy for improving sleep quality in patients with end-stage kidney disease on hemodialysis. Evidence-Based Complement Altern Med 21(2017):1–10
Hunsley J (1992) Development of the treatment acceptability questionnaire. J Psychopathol Behav Assess 14(1):55–64
Szeto C-C, Li PK-T, Johnson DW, Bernardini J, Dong J, Figueiredo AE et al (2017) ISPD catheter-related infection recommendations: 2017 update. Perit Dial Int. 37(2):141–154
Htay H, Cho Y, Pascoe EM, Darssan D, Nadeau-Fredette A-C, Hawley C et al (2017) Center effects and peritoneal dialysis peritonitis outcomes: analysis of a national registry. Am J Kidney Dis. 71:814–821
Htay H, Seng JJB, Yong MHA, Pang SC, Wu SY, Chan CM et al (2018) Comparison of clinical presentation and outcomes of peritonitis in the elderly and younger peritoneal dialysis patients. Perit Dial Int. 39:163–168
Htay H, Cho Y, Pascoe EM, Darssan D, Hawley C, Clayton PA et al (2017) Outcomes of Corynebacterium peritonitis: a multicenter registry analysis. Perit Dial Int 37(6):619–626
Htay H, Cho Y, Pascoe EM, Hawley C, Clayton PA, Borlace M et al (2018) Outcomes of Acinetobacter peritonitis in peritoneal dialysis patients: a multicenter registry analysis. Perit Dial Int 38(4):257–265
Perl J, Fuller DS, Bieber BA, Boudville N, Kanjanabuch T, Ito Y et al (2020) Peritoneal dialysis-related infection rates and outcomes: results from the peritoneal dialysis outcomes and practice patterns study (PDOPPS). Am J Kidney Dis 76(1):42–53
Htay H, Johnson DW, Wu SY, Oei EL, Foo MWY, Choo JCJ (2017) Comparison of topical chlorhexidine and mupirocin for the prevention of exit-site infection in incident peritoneal dialysis patients. Perit Dial Int 37(3):266–272
Bunke CM, Brier ME, Golper TA (1997) Outcomes of single organism peritonitis in peritoneal dialysis: Gram negatives versus gram positives in the network 9 peritonitis study. Kidney Int 52(2):524–529
Weitz NA, Lauren CT, Weiser JA, LeBoeuf NR, Grossman ME, Biagas K et al (2013) Chlorhexidine gluconate-impregnated central access catheter dressings as a cause of erosive contact dermatitis. JAMA Dermatol 149(2):195
Garland JS, Alex CP, Mueller CD, Otten D, Shivpuri C, Harris MC et al (2001) A randomized trial comparing povidone-iodine to a chlorhexidine gluconate-impregnated dressing for prevention of central venous catheter infections in neonates. Pediatrics 107(6):1431–1436
Acknowledgements
The authors gratefully acknowledge the contributions of Ms Chue Xiuping, a clinical research coordinator, Medical ACP, Singhealth, Pte Ltd, a PD nurse, Ms Kok Su Fong, division of nursing, Singapore General Hospital, and physicians and nurses from peritoneal dialysis unit, Singapore General Hospital.
Funding
The study was supported by Singhealth New Investigator grant (to Htay Htay) and Johnson & Johnson Pte Ltd.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Htay Htay has received consultancy fees and travel sponsorships from AWAK Technologies Pte Ltd. She has also received speaker’s honoraria and travel sponsorships from Baxter Healthcare. Marjorie Foo has received consultancy fees and travel sponsorships from AWAK Technologies Pte Ltd, and travel sponsorships from Baxter Healthcare. David Johnson has received grants from National Health and Medical Research Council of Australia, consultancy fees, research grants, speaker’s honoraria and travel sponsorships from Baxter Healthcare and Fresenius Medical Care, consultancy fees from Astra Zeneca and AWAK, speaker’s honoraria and travel sponsorships from ONO, and travel sponsorships from Amgen. All other authors have no conflict of interest to declare.
Ethics approval
The study was approved by Singhealth Centralized Institutional Review Board (CIRB; reference number 2017/2698).
Consent to participate
Informed consent was obtained from all 50 participants in the pilot study. The CIRB granted a waiver of informed consent for the historical control group as non-interventional study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Htay, H., Choo, J.C.J., Johnson, D.W. et al. Chlorhexidine-impregnated sponge dressing for prevention of catheter exit-site infection in peritoneal dialysis patients: a pilot study. Int Urol Nephrol 53, 803–812 (2021). https://doi.org/10.1007/s11255-020-02674-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-020-02674-w