Abstract
Ferroptosis is a form of iron-dependent, non-apoptotic regulated cell death, which is characterized by the accumulation of lipid hydroperoxides to lethal levels. Ferroptosis recently has been shown to have implications in diverse kidney diseases, such as acute kidney injury, polycystic kidney disease and renal cell carcinoma. This review summarizes current research on ferroptosis, its underlying mechanisms and its role in the progression of different kidney diseases to provide more information regarding treatment and prevention of these destructive diseases.

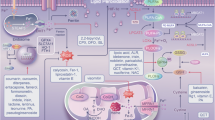
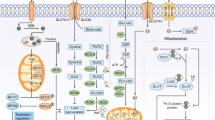
Free iron can interact with ROS, forming a radical and rapid reacts with oxygen to generate a lipid peroxide via the Fenton reaction. Thus, iron import, export, storage, and turnover impact ferroptosis sensitivity. Lipid peroxides and their degradation products are controlled by GSH-based redox reactions. System xc−imports cystine, which is reduced to cysteine and used to synthesize glutathione. GPX4 reduces membrane phospholipid hydroperoxides to suppress ferroptosis. Downstream of NRF2 participated in many ferroptotic process maintaining redox homeostasis, iron/heme metabolism and lipid metabolism

Similar content being viewed by others
References
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS, Morrison B III, Stockwell BR (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149(5):1060–1072. https://doi.org/10.1016/j.cell.2012.03.042
Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, Cheah JH, Clemons PA, Shamji AF, Clish CB, Brown LM, Girotti AW, Cornish VW, Schreiber SL, Stockwell BR (2014) Regulation of ferroptotic cancer cell death by GPX4. Cell 156(1–2):317–331. https://doi.org/10.1016/j.cell.2013.12.010
Cao JY, Dixon SJ (2016) Mechanisms of ferroptosis. Cell Mol Life Sci 73(11–12):2195–2209. https://doi.org/10.1007/s00018-016-2194-1
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascon S, Hatzios SK, Kagan VE, Noel K, Jiang X, Linkermann A, Murphy ME, Overholtzer M, Oyagi A, Pagnussat GC, Park J, Ran Q, Rosenfeld CS, Salnikow K, Tang D, Torti FM, Torti SV, Toyokuni S, Woerpel KA, Zhang DD (2017) Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell 171(2):273–285. https://doi.org/10.1016/j.cell.2017.09.021
Linkermann A, Skouta R, Himmerkus N, Mulay SR, Dewitz C, De Zen F, Prokai A, Zuchtriegel G, Krombach F, Welz PS, Weinlich R, Vanden Berghe T, Vandenabeele P, Pasparakis M, Bleich M, Weinberg JM, Reichel CA, Brasen JH, Kunzendorf U, Anders HJ, Stockwell BR, Green DR, Krautwald S (2014) Synchronized renal tubular cell death involves ferroptosis. Proc Natl Acad Sci U S A 111(47):16836–16841. https://doi.org/10.1073/pnas.1415518111
Eleftheriadis T, Pissas G, Antoniadi G, Liakopoulos V, Stefanidis I (2018) Cell death patterns due to warm ischemia or reperfusion in renal tubular epithelial cells originating from human, mouse, or the native hibernator hamster. Biology (Basel). https://doi.org/10.3390/biology7040048
Tuo QZ, Lei P, Jackman KA, Li XL, Xiong H, Li XL, Liuyang ZY, Roisman L, Zhang ST, Ayton S, Wang Q, Crouch PJ, Ganio K, Wang XC, Pei L, Adlard PA, Lu YM, Cappai R, Wang JZ, Liu R, Bush AI (2017) Tau-mediated iron export prevents ferroptotic damage after ischemic stroke. Mol Psychiatry 22(11):1520–1530. https://doi.org/10.1038/mp.2017.171
Li Q, Han X, Lan X, Gao Y, Wan J, Durham F, Cheng T, Yang J, Wang Z, Jiang C, Ying M, Koehler RC, Stockwell BR, Wang J (2017) Inhibition of neuronal ferroptosis protects hemorrhagic brain. JCI Insight 2(7):e90777. https://doi.org/10.1172/jci.insight.90777
Ma S, Dielschneider RF, Henson ES, Xiao W, Choquette TR, Blankstein AR, Chen Y, Gibson SB (2017) Ferroptosis and autophagy induced cell death occur independently after siramesine and lapatinib treatment in breast cancer cells. PLoS One 12(8):e0182921. https://doi.org/10.1371/journal.pone.0182921
Kinowaki Y, Kurata M, Ishibashi S, Ikeda M, Tatsuzawa A, Yamamoto M, Miura O, Kitagawa M, Yamamoto K (2018) Glutathione peroxidase 4 overexpression inhibits ROS-induced cell death in diffuse large B-cell lymphoma. Lab Invest 98(5):609–619. https://doi.org/10.1038/s41374-017-0008-1
Siew ED, Davenport A (2015) The growth of acute kidney injury: a rising tide or just closer attention to detail? Kidney Int 87(1):46–61. https://doi.org/10.1038/ki.2014.293
Gonsalez SR, Cortes AL, da Silva RC, Lowe J, Prieto MC, da Silva Lara L (2019) Acute kidney injury overview: from basic findings to new prevention and therapy strategies. Pharmacol Ther. https://doi.org/10.1016/j.pharmthera.2019.04.001
Linkermann A, Brasen JH, Darding M, Jin MK, Sanz AB, Heller JO, De Zen F, Weinlich R, Ortiz A, Walczak H, Weinberg JM, Green DR, Kunzendorf U, Krautwald S (2013) Two independent pathways of regulated necrosis mediate ischemia-reperfusion injury. Proc Natl Acad Sci U S A 110(29):12024–12029. https://doi.org/10.1073/pnas.1305538110
Linkermann A, Brasen JH, Himmerkus N, Liu S, Huber TB, Kunzendorf U, Krautwald S (2012) Rip1 (receptor-interacting protein kinase 1) mediates necroptosis and contributes to renal ischemia/reperfusion injury. Kidney Int 81(8):751–761. https://doi.org/10.1038/ki.2011.450
Luedde M, Lutz M, Carter N, Sosna J, Jacoby C, Vucur M, Gautheron J, Roderburg C, Borg N, Reisinger F, Hippe HJ, Linkermann A, Wolf MJ, Rose-John S, Lullmann-Rauch R, Adam D, Flogel U, Heikenwalder M, Luedde T, Frey N (2014) RIP3, a kinase promoting necroptotic cell death, mediates adverse remodelling after myocardial infarction. Cardiovasc Res 103(2):206–216. https://doi.org/10.1093/cvr/cvu146
Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ, Herbach N, Aichler M, Walch A, Eggenhofer E, Basavarajappa D, Radmark O, Kobayashi S, Seibt T, Beck H, Neff F, Esposito I, Wanke R, Forster H, Yefremova O, Heinrichmeyer M, Bornkamm GW, Geissler EK, Thomas SB, Stockwell BR, O’Donnell VB, Kagan VE, Schick JA, Conrad M (2014) Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol 16(12):1180–1191. https://doi.org/10.1038/ncb3064
Muller T, Dewitz C, Schmitz J, Schroder AS, Brasen JH, Stockwell BR, Murphy JM, Kunzendorf U, Krautwald S (2017) Necroptosis and ferroptosis are alternative cell death pathways that operate in acute kidney failure. Cell Mol Life Sci 74(19):3631–3645. https://doi.org/10.1007/s00018-017-2547-4
Schumer M, Colombel MC, Sawczuk IS, Gobé G, Connor J, O’Toole KM, Olsson CA, Wise GJ, Buttyan R (1992) Morphologic, biochemical, and molecular evidence of apoptosis during the reperfusion phase after brief periods of renal ischemia. Am J Pathol 140(4):831–838
Linkermann A, Chen G, Dong G, Kunzendorf U, Krautwald S, Dong Z (2014) Regulated cell death in AKI. J Am Soc Nephrol 25(12):2689–2701. https://doi.org/10.1681/ASN.2014030262
Long DA, Woolf AS, Suda T, Yuan HT (2001) Increased renal angiopoietin-1 expression in folic acid-induced nephrotoxicity in mice. J Am Soc Nephrol 12(12):2721–2731
Stallons LJ, Whitaker RM, Schnellmann RG (2014) Suppressed mitochondrial biogenesis in folic acid-induced acute kidney injury and early fibrosis. Toxicol Lett 224(3):326–332. https://doi.org/10.1016/j.toxlet.2013.11.014
Martin-Sanchez D, Ruiz-Andres O, Poveda J, Carrasco S, Cannata-Ortiz P, Sanchez-Nino MD, Ruiz Ortega M, Egido J, Linkermann A, Ortiz A, Sanz AB (2017) Ferroptosis, but not necroptosis, is important in nephrotoxic folic acid-induced AKI. J Am Soc Nephrol 28(1):218–229. https://doi.org/10.1681/ASN.2015121376
Wen Q, Liu J, Kang R, Zhou B, Tang D (2019) The release and activity of HMGB1 in ferroptosis. Biochem Biophys Res Commun 510(2):278–283. https://doi.org/10.1016/j.bbrc.2019.01.090
Dodson M, de la Vega MR, Cholanians AB, Schmidlin CJ, Chapman E, Zhang DD (2019) Modulating NRF2 in disease: timing is everything. Annu Rev Pharmacol Toxicol 59:555–575. https://doi.org/10.1146/annurev-pharmtox-010818-021856
Nezu M, Souma T, Yu L, Suzuki T, Saigusa D, Ito S, Suzuki N, Yamamoto M (2017) Transcription factor Nrf2 hyperactivation in early-phase renal ischemia-reperfusion injury prevents tubular damage progression. Kidney Int 91(2):387–401. https://doi.org/10.1016/j.kint.2016.08.023
Dodson M, Castro-Portuguez R, Zhang DD (2019) NRF2 plays a critical role in mitigating lipid peroxidation and ferroptosis. Redox Biol. https://doi.org/10.1016/j.redox.2019.101107
Swaminathan S (2018) Iron homeostasis pathways as therapeutic targets in acute kidney injury. Nephron 140(2):156–159. https://doi.org/10.1159/000490808
Adedoyin O, Boddu R, Traylor A, Lever JM, Bolisetty S, George JF, Agarwal A (2018) Heme oxygenase-1 mitigates ferroptosis in renal proximal tubule cells. Am J Physiol Renal Physiol 314(5):F702–F714. https://doi.org/10.1152/ajprenal.00044.2017
Skouta R, Dixon SJ, Wang J, Dunn DE, Orman M, Shimada K, Rosenberg PA, Lo DC, Weinberg JM, Linkermann A, Stockwell BR (2014) Ferrostatins inhibit oxidative lipid damage and cell death in diverse disease models. J Am Chem Soc 136(12):4551–4556. https://doi.org/10.1021/ja411006a
Webster AC, Nagler EV, Morton RL, Masson P (2017) Chronic kidney disease. The Lancet 389(10075):1238–1252. https://doi.org/10.1016/s0140-6736(16)32064-5
Leemans JC, Kors L, Anders HJ, Florquin S (2014) Pattern recognition receptors and the inflammasome in kidney disease. Nat Rev Nephrol 10(7):398–414. https://doi.org/10.1038/nrneph.2014.91
Liu BC, Tang TT, Lv LL, Lan HY (2018) Renal tubule injury: a driving force toward chronic kidney disease. Kidney Int 93(3):568–579. https://doi.org/10.1016/j.kint.2017.09.033
Linkermann A, Stockwell BR, Krautwald S, Anders HJ (2014) Regulated cell death and inflammation: an auto-amplification loop causes organ failure. Nat Rev Immunol 14(11):759–767. https://doi.org/10.1038/nri3743
Galluzzi L, Kepp O, Krautwald S, Kroemer G, Linkermann A (2014) Molecular mechanisms of regulated necrosis. Semin Cell Dev Biol 35:24–32. https://doi.org/10.1016/j.semcdb.2014.02.006
Jiang T, Huang Z, Lin Y, Zhang Z, Fang D, Zhang DD (2010) The protective role of Nrf2 in streptozotocin-induced diabetic nephropathy. Diabetes 59(4):850–860. https://doi.org/10.2337/db09-1342
Tan RJ, Chartoumpekis DV, Rush BM, Zhou D, Fu H, Kensler TW, Liu Y (2016) Keap1 hypomorphism protects against ischemic and obstructive kidney disease. Sci Rep 6:36185. https://doi.org/10.1038/srep36185
de Zeeuw D, Akizawa T, Audhya P, Bakris GL, Chin M, Christ-Schmidt H, Goldsberry A, Houser M, Krauth M, Lambers Heerspink HJ, McMurray JJ, Meyer CJ, Parving HH, Remuzzi G, Toto RD, Vaziri ND, Wanner C, Wittes J, Wrolstad D, Chertow GM, Investigators BT (2013) Bardoxolone methyl in type 2 diabetes and stage 4 chronic kidney disease. N Engl J Med 369(26):2492–2503. https://doi.org/10.1056/NEJMoa1306033
Pergola PE, Raskin P, Toto RD, Meyer CJ, Huff JW, Grossman EB, Krauth M, Ruiz S, Audhya P, Christ-Schmidt H, Wittes J, Warnock DG (2011) Bardoxolone methyl and kidney function in CKD with type 2 diabetes. N Engl J Med 365(4):327–336. https://doi.org/10.1056/NEJMoa1105351
Schnurr K, Belkner J, Ursini F, Schewe T, Kuhn H (1996) The selenoenzyme phospholipid hydroperoxide glutathione peroxidase controls the activity of the 15-lipoxygenase with complex substrates and preserves the specificity of the oxygenation products. J Biol Chem 271(9):4653–4658
Imai H, Narashima K, Arai M, Sakamoto H, Chiba N, Nakagawa Y (1998) Suppression of leukotriene formation in RBL-2H3 cells that overexpressed phospholipid hydroperoxide glutathione peroxidase. J Biol Chem 273(4):1990–1997
Brigelius-Flohe R (2006) Glutathione peroxidases and redox-regulated transcription factors. Biol Chem 387(10–11):1329–1335. https://doi.org/10.1515/BC.2006.166
Li C, Deng X, Xie X, Liu Y, Friedmann Angeli JP, Lai L (2018) Activation of glutathione peroxidase 4 as a novel anti-inflammatory strategy. Front Pharmacol 9:1120. https://doi.org/10.3389/fphar.2018.01120
Banning A, Schnurr K, Bol GF, Kupper D, Muller-Schmehl K, Viita H, Yla-Herttuala S, Brigelius-Flohe R (2004) Inhibition of basal and interleukin-1-induced VCAM-1 expression by phospholipid hydroperoxide glutathione peroxidase and 15-lipoxygenase in rabbit aortic smooth muscle cells. Free Radical Biol Med 36(2):135–144
Wenk J, Schuller J, Hinrichs C, Syrovets T, Azoitei N, Podda M, Wlaschek M, Brenneisen P, Schneider LA, Sabiwalsky A, Peters T, Sulyok S, Dissemond J, Schauen M, Krieg T, Wirth T, Simmet T, Scharffetter-Kochanek K (2004) Overexpression of phospholipid-hydroperoxide glutathione peroxidase in human dermal fibroblasts abrogates UVA irradiation-induced expression of interstitial collagenase/matrix metalloproteinase-1 by suppression of phosphatidylcholine hydroperoxide-mediated NF-κB activation and interleukin-6 release. J Biol Chem 279(44):45634–45642. https://doi.org/10.1074/jbc.M408893200
Kang R, Tang D (2017) Autophagy and ferroptosis—what’s the connection? Curr Pathobiol Rep 5(2):153–159. https://doi.org/10.1007/s40139-017-0139-5
Hou W, Xie Y, Song X, Sun X, Lotze MT, Zeh HJ 3rd, Kang R, Tang D (2016) Autophagy promotes ferroptosis by degradation of ferritin. Autophagy 12(8):1425–1428. https://doi.org/10.1080/15548627.2016.1187366
Bergmann C, Guay-Woodford LM, Harris PC, Horie S, Peters DJM, Torres VE (2018) Polycystic kidney disease. Nat Rev Dis Primers 4(1):50. https://doi.org/10.1038/s41572-018-0047-y
Maser RL, Vassmer D, Magenheimer BS, Calvet JP (2002) Oxidant stress and reduced antioxidant enzyme protection in polycystic kidney disease. J Am Soc Nephrol 13(4):991–999
Kunzelmann K (2016) Ion channels in regulated cell death. Cell Mol Life Sci 73(11–12):2387–2403. https://doi.org/10.1007/s00018-016-2208-z
Hoste S, Chargui A, Belfodil R, Corcelle E, Duranton C, Rubera I, Poujeol C, Mograbi B, Tauc M, Poujeol P (2010) CFTR mediates apoptotic volume decrease and cell death by controlling glutathione efflux and ROS production in cultured mice proximal tubules. Am J Physiol Renal Physiol 298(2):F435–453. https://doi.org/10.1152/ajprenal.00286.2009
Simoes F, Ousingsawat J, Wanitchakool P, Fonseca A, Cabrita I, Benedetto R, Schreiber R, Kunzelmann K (2018) CFTR supports cell death through ROS-dependent activation of TMEM16F (anoctamin 6). Pflugers Arch 470(2):305–314. https://doi.org/10.1007/s00424-017-2065-0
Forschbach V, Goppelt-Struebe M, Kunzelmann K, Schreiber R, Piedagnel R, Kraus A, Eckardt KU, Buchholz B (2015) Anoctamin 6 is localized in the primary cilium of renal tubular cells and is involved in apoptosis-dependent cyst lumen formation. Cell Death Dis 6:e1899. https://doi.org/10.1038/cddis.2015.273
Schreiber R, Buchholz B, Kraus A, Schley G, Scholz J, Ousingsawat J, Kunzelmann K (2019) Lipid peroxidation drives renal cyst growth in vitro through activation of TMEM16A. J Am Soc Nephrol 30(2):228–242. https://doi.org/10.1681/ASN.2018010039
Majmundar AJ, Wong WJ, Simon MC (2010) Hypoxia-inducible factors and the response to hypoxic stress. Mol Cell 40(2):294–309. https://doi.org/10.1016/j.molcel.2010.09.022
Cairns RA, Harris IS, Mak TW (2011) Regulation of cancer cell metabolism. Nat Rev Cancer 11(2):85–95. https://doi.org/10.1038/nrc2981
Gorrini C, Harris IS, Mak TW (2013) Modulation of oxidative stress as an anticancer strategy. Nat Rev Drug Discov 12(12):931–947. https://doi.org/10.1038/nrd4002
Miess H, Dankworth B, Gouw AM, Rosenfeldt M, Schmitz W, Jiang M, Saunders B, Howell M, Downward J, Felsher DW, Peck B, Schulze A (2018) The glutathione redox system is essential to prevent ferroptosis caused by impaired lipid metabolism in clear cell renal cell carcinoma. Oncogene 37(40):5435–5450. https://doi.org/10.1038/s41388-018-0315-z
Yang WH, Ding CC, Sun T, Rupprecht G, Lin CC, Hsu D, Chi JT (2019) The hippo pathway effector TAZ regulates ferroptosis in renal cell carcinoma. Cell Rep 28(10):2501–2508.e2504. https://doi.org/10.1016/j.celrep.2019.07.107
Kerins MJ, Milligan J, Wohlschlegel JA, Ooi A (2018) Fumarate hydratase inactivation in hereditary leiomyomatosis and renal cell cancer is synthetic lethal with ferroptosis induction. Cancer Sci 109(9):2757–2766. https://doi.org/10.1111/cas.13701
Hangauer MJ, Viswanathan VS, Ryan MJ, Bole D, Eaton JK, Matov A, Galeas J, Dhruv HD, Berens ME, Schreiber SL, McCormick F, McManus MT (2017) Drug-tolerant persister cancer cells are vulnerable to GPX4 inhibition. Nature 551(7679):247–250. https://doi.org/10.1038/nature24297
Luo M, Wu L, Zhang K, Wang H, Zhang T, Gutierrez L, O’Connell D, Zhang P, Li Y, Gao T, Ren W, Yang Y (2018) miR-137 regulates ferroptosis by targeting glutamine transporter SLC1A5 in melanoma. Cell Death Differ 25(8):1457–1472. https://doi.org/10.1038/s41418-017-0053-8
Zhang K, Wu L, Zhang P, Luo M, Du J, Gao T, O’Connell D, Wang G, Wang H, Yang Y (2018) miR-9 regulates ferroptosis by targeting glutamic-oxaloacetic transaminase GOT1 in melanoma. Mol Carcinog 57(11):1566–1576. https://doi.org/10.1002/mc.22878
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tang, S., Xiao, X. Ferroptosis and kidney diseases. Int Urol Nephrol 52, 497–503 (2020). https://doi.org/10.1007/s11255-019-02335-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-019-02335-7