Abstract
Purpose
Accumulating evidence suggests that a relationship exists between serum uric acid (UA) and the progression of chronic kidney disease (CKD), but information regarding idiopathic membranous nephropathy (IMN) is limited.
Methods
Patients with renal biopsy-confirmed diagnosis of IMN between 2009 and 2017 were identified. The demographic and clinical data recorded at the time of renal biopsy were considered the baseline values. The included cases were separated into three groups based on tertiles of the baseline serum UA level, and the relationship between serum UA and poor renal outcome was investigated by receiver operating characteristic (ROC) and time-event analyses. The primary endpoint was poor renal outcome, which was defined as a decrease in the estimated glomerular filtration rate to 50% of the baseline level or progression to end-stage renal disease during the follow-up.
Results
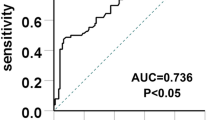
Of 989 cases, 572 eligible patients were included. During a median of 18 months of follow-up, 45 (7.9%) patients progressed to the primary endpoint. Both baseline serum UA and time-averaged UA levels could be used for discrimination of renal outcomes, but the difference was not significant (p value = 0.6). Our Cox regression analysis further demonstrated that baseline serum UA was an independent predictor of poor renal outcome in IMN patients, and subgroup analysis revealed a gender difference in the predictive effect of serum UA.
Conclusions
Our study demonstrated that baseline serum UA was an independent predictor of poor renal outcome in patients with IMN, and a gender difference in the predictive effect was observed in our cohort.



Similar content being viewed by others
Abbreviations
- UA:
-
Uric acid
- CI:
-
Confidence interval
- HR:
-
Hazard ratio
- MN:
-
Membranous nephropathy
- ESRD:
-
End-stage renal disease
- CKD:
-
Chronic kidney disease
- BP:
-
Blood pressure
- eGFR:
-
Estimated glomerular filtration rate
- SD:
-
Standard deviation
- Scr:
-
Serum creatinine
- SBP:
-
Systolic blood pressure
- DBP:
-
Diastolic blood pressure
References
Makker SP, Tramontano A (2011) Idiopathic membranous nephropathy: an autoimmune disease. Semin Nephrol 31(4):333–340. https://doi.org/10.1016/j.semnephrol.2011.06.004
Beck LH Jr, Bonegio RG, Lambeau G, Beck DM, Powell DW, Cummins TD, Klein JB, Salant DJ (2009) M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N Engl J Med 361(1):11–21. https://doi.org/10.1056/NEJMoa0810457
Tomas NM, Beck LH Jr, Meyer-Schwesinger C, Seitz-Polski B, Ma H, Zahner G, Dolla G, Hoxha E, Helmchen U, Dabert-Gay AS, Debayle D, Merchant M, Klein J, Salant DJ, Stahl RA, Lambeau G (2014) Thrombospondin type-1 domain-containing 7A in idiopathic membranous nephropathy. N Engl J Med 371(24):2277–2287. https://doi.org/10.1056/NEJMoa1409354
Wolf G (2006) Renal injury due to renin–angiotensin–aldosterone system activation of the transforming growth factor-β pathway. Kidney Int 70(11):1914–1919
Shiiki H, Saito T, Nishitani Y, Mitarai T, Yorioka N, Yoshimura A, Yokoyama H, Nishi S, Tomino Y, Kurokawa K (2004) Prognosis and risk factors for idiopathic membranous nephropathy with nephrotic syndrome in Japan. Kidney Int 65(4):1400–1407
Cattran D (2005) Management of membranous nephropathy: when and what for treatment. J Am Soc Nephrol 16(5):1188–1194
Schieppati A, Mosconi L, Perna A, Mecca G, Bertani T, Garattini S, Remuzzi G (1993) Prognosis of untreated patients with idiopathic membranous nephropathy. N Engl J Med 329(2):85–89
Sprangers B, Bomback AS, Cohen SD, Radhakrishnan J, Valeri A, Markowitz GS, D’Agati V, Appel GB (2012) Idiopathic membranous nephropathy: clinical and histologic prognostic features and treatment patterns over time at a tertiary referral center. Am J Nephrol 36(1):78–89
Reichert LJ, Koene RA, Wetzels JF (1998) Prognostic factors in idiopathic membranous nephropathy. Am J Kidney Dis 31(1):1–11
Segal PE, Choi MJ (2012) Recent advances and prognosis in idiopathic membranous nephropathy. Adv Chronic Kidney Dis 19(2):114–119. https://doi.org/10.1053/j.ackd.2012.01.007
Kanellis J, Kang D-H (2005) Uric acid as a mediator of endothelial dysfunction, inflammation, and vascular disease. In: Seminars in nephrology, vol 1, 2005. Elsevier, New York, pp 39–42
Meotti FC, Jameson GN, Turner R, Harwood DT, Stockwell S, Rees MD, Thomas SR, Kettle AJ (2011) Urate as a physiological substrate for myeloperoxidase: implications for hyperuricemia and inflammation. J Biol Chem. https://doi.org/10.1074/jbc.M110.172460
Pagidipati NJ, Hess CN, Clare RM, Akerblom A, Tricoci P, Wojdyla D, Keenan RT, James S, Held C, Mahaffey KW (2017) An examination of the relationship between serum uric acid level, a clinical history of gout, and cardiovascular outcomes among patients with acute coronary syndrome. Am Heart J 187:53–61
Nodera M, Suzuki H, Matsumoto Y, Kamioka M, Kaneshiro T, Yoshihisa A, Ohira T, Takeishi Y (2018) Association between serum uric acid level and ventricular tachyarrhythmia in heart failure patients with implantable cardioverter-defibrillator. Cardiology 140(1):47–51
Chang H-Y, Lee P-H, Lei C-C, Hsu Y-C, Chang H-H, Tung C-W, Lin C-L, Yang H-F, Lu L-C, Jong M-C (2010) Hyperuricemia as an independent risk factor of chronic kidney disease in middle-aged and elderly population. Am J Med Sci 339(6):509–515
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J, Ckd EPI (2009) A new equation to estimate glomerular filtration rate. Ann Intern Med 150(9):604–612
Levey AS, Eckardt K-U, Tsukamoto Y, Levin A, Coresh J, Rossert J, Zeeuw DD, Hostetter TH, Lameire N, Eknoyan G (2005) Definition and classification of chronic kidney disease: a position statement from kidney disease: improving global outcomes (KDIGO). Kidney Int 67(6):2089–2100
Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, Sanchez J-C, Müller M (2011) pROC: an open-source package for R and S + to analyze and compare ROC curves. BMC Bioinform 12(1):77. https://doi.org/10.1186/1471-2105-12-77
Wickham H (2009) ggplot2: elegant graphics for data analysis. Springer, New York
Team RC (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Yoshida K (2019) tableone: create ‘Table 1’ to describe baseline characteristics. R package version 0.10.0. https://CRAN.R-project.org/package=tableone
Uchida S, Kumagai T, Chang WX, Tamura Y, Shibata S (2018) Time to target uric acid to retard chronic kidney disease progression. In: Treviño-Becerra A, Iseki K (eds) Uric acid in chronic kidney disease, vol 192. Karger Publishers, Basel, pp 56–68
Eleftheriadis T, Golphinopoulos S, Pissas G, Stefanidis I (2017) Asymptomatic hyperuricemia and chronic kidney disease: narrative review of a treatment controversial. J Adv Res 8(5):555–560. https://doi.org/10.1016/j.jare.2017.05.001
Ramirez-Sandoval JC, Sanchez-Lozada LG, Madero M (2017) Uric acid, vascular stiffness, and chronic kidney disease: is there a link? Blood Purif 43(1–3):189–195. https://doi.org/10.1159/000452726
Johnson RJ, Nakagawa T, Jalal D, Sánchez-Lozada LG, Kang D-H, Ritz E (2013) Uric acid and chronic kidney disease: which is chasing which? Nephrol Dial Transplant 28(9):2221–2228
Mwasongwe SE, Fülöp T, Katz R, Musani SK, Sims M, Correa A, Flessner MF, Young BA (2018) Relation of uric acid level to rapid kidney function decline and development of kidney disease: the Jackson Heart Study. J Clin Hypertens 20(4):775–783
Lee JW, Lee KH (2019) Comparison of renoprotective effects of febuxostat and allopurinol in hyperuricemic patients with chronic kidney disease. Int Urol Nephrol. https://doi.org/10.1007/s11255-018-2051-2
Sampson AL, Singer RF, Walters GD (2017) Uric acid lowering therapies for preventing or delaying the progression of chronic kidney disease. Cochrane Database Syst Rev 10:CD009460. https://doi.org/10.1002/14651858.cd009460.pub2
Sanchez-Lozada LG, Soto V, Tapia E, Avila-Casado C, Sautin YY, Nakagawa T, Franco M, Rodriguez-Iturbe B, Johnson RJ (2008) Role of oxidative stress in the renal abnormalities induced by experimental hyperuricemia. Am J Physiol Renal Physiol 295(4):F1134–F1141. https://doi.org/10.1152/ajprenal.00104.2008
Sanchez-Lozada LG, Tapia E, Santamaria J, Avila-Casado C, Soto V, Nepomuceno T, Rodriguez-Iturbe B, Johnson RJ, Herrera-Acosta J (2005) Mild hyperuricemia induces vasoconstriction and maintains glomerular hypertension in normal and remnant kidney rats. Kidney Int 67(1):237–247. https://doi.org/10.1111/j.1523-1755.2005.00074.x
Nakagawa T, Mazzali M, Kang DH, Kanellis J, Watanabe S, Sanchez-Lozada LG, Rodriguez-Iturbe B, Herrera-Acosta J, Johnson RJ (2003) Hyperuricemia causes glomerular hypertrophy in the rat. Am J Nephrol 23(1):2–7. https://doi.org/10.1159/000066303
Zhang J, Chen C, Zhou Q, Zheng S, Lv Y, Zhang J, You X, Li Z, Zhou Z, Pan M (2017) Elevated serum fibrinogen level is an independent risk factor for IgA nephropathy. Oncotarget 8(58):99125–99135. https://doi.org/10.18632/oncotarget.21702
Kawabe M, Sato A, Hoshi T, Sakai S, Hiraya D, Watabe H, Kakefuda Y, Ishibashi M, Abe D, Takeyasu N (2016) Gender differences in the association between serum uric acid and prognosis in patients with acute coronary syndrome. J Cardiol 67(2):170–176
Culleton BF, Larson MG, Kannel WB, Levy D (1999) Serum uric acid and risk for cardiovascular disease and death: the Framingham Heart Study. Ann Intern Med 131:7–13
Ben Salem C, Slim R, Fathallah N, Hmouda H (2016) Drug-induced hyperuricaemia and gout. Rheumatology 56(5):679–688
Uchida S, Chang WX, Ota T, Tamura Y, Shiraishi T, Kumagai T, Shibata S, Fujigaki Y, Hosoyamada M, Kaneko K (2015) Targeting uric acid and the inhibition of progression to end-stage renal disease—a propensity score analysis. PLoS One 10(12):e0145506
Acknowledgements
The authors would like to thank their colleagues at the Department of Nephrology, the First and Second Affiliated Hospitals of Wenzhou Medical University for their support and assistance during the study period.
Funding
This study was supported by the Wenzhou Municipal Science and Technology Bureau under Grant Y20170300 to Min Pan and the Natural Science Foundation of Zhejiang Province under Grant LY14H050006 to Fan Lin.
Author information
Authors and Affiliations
Contributions
All authors contributed significant intellectual content to this manuscript as follows: principal investigators, conceived and designed the study: ZJ, PM, and LGY; assessed the study, extracted data, and performed statistical analyses: ZJ, ZJN, YXH, and LD; drafted the manuscript: ZJ; performed a critical review of the manuscript: LF and LGY. All authors have read the manuscript and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Ethical approval
The procedure was approved by the Ethics Committee of the First Affiliated Hospital of Wenzhou Medical University.
Informed consent
This study was performed after obtaining written informed consent from all patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, J., Pan, M., Zhang, J. et al. Serum uric acid is an independent predictor of renal outcomes in patients with idiopathic membranous nephropathy. Int Urol Nephrol 51, 1797–1804 (2019). https://doi.org/10.1007/s11255-019-02254-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-019-02254-7