Abstract
Objectives
We aimed to evaluate the role of nephrin and podocalyxin in determining the intervals between shock wave lithotripsy (SWL) sessions and how soon the kidney damage was recovered.
Methods
This work was a prospective study that included 30 patients with unilateral kidney stones. The patients’ midflow urine samples were collected before SWL and 1 h, 1 day and 1 week after the procedure. Nephrin and podocalyxin levels in the urine samples were measured by the enzyme-linked immunosorbent assay method.
Results
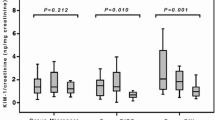
Among the 30 patients who underwent SWL, 19 were males and 11 were females. The mean age of the SWL group was 34.7 ± 13.2. Both biomarkers did not correlate with age, creatinine values, body mass index, stone side, stone size, energy, frequency and shock numbers. Nephrin and podocalyxin levels were significantly higher at the pre-SWL point (p < 0.05). After the procedure, a significant decrease was observed in both biomarker levels (p < 0.05). At the end of first day, these levels started to increase progressively up to the end of the first week (p > 0.05).
Conclusions
Nephrin and podocalyxin may help to determine early period kidney damage associated with SWL. Post-SWL podocalyxin and nephrin values may be used to determine the interval between SWL sessions.

Similar content being viewed by others
Abbreviations
- BMI:
-
Body mass index
- SWL:
-
Shock wave lithotripsy
- SD:
-
Slit diaphragm
- HESW:
-
High-energy shock waves
- ERPF:
-
Effective renal plasma flow
- ROS:
-
Reactive oxygen species
- RIRS:
-
Retrograde intrarenal surgery
References
Romero V, Akpinar H, Assimos DG (2010) Kidney stones: a global picture of prevalence, incidence, and associated risk factors. Rev Urol 12(2–3):e86–e96
Cohen TD, Durrani AF, Brown SA, Ferraro R, Preminger GM (1998) Lipid peroxidation induced by shockwave lithotripsy. J Endourol 12(3):229–232
Willis LR, Evan AP, Connors BA, Blomgren P, Fineberg NS, Lingeman JE (1999) Relationship between kidney size, renal injury, and renal impairment induced by shock wave lithotripsy. J Am Soc Nephrol 10(8):1753–1762
Lingeman JE, McAteer JA, Kempson SA, Evan AP (1988) Bioeffects of extracorporeal shockwave lithotripsy: strategy for research and treatment. Urol Clin N Am 15:507
Recker F, Hofmann W, Bex A, Tscholl R (1992) Quantitative determination of urinary marker proteins: a model to detect intrarenal bioeffects after extracorporeal lithotripsy. J Urol 148(3):1000–1006
Mundel P, Shankland SJ (2002) Podocyte biology and response to injury. J Am Soc Nephrol. 13:3005–3015
Camici M (2007) Urinary detection of podocyte injury. Biomed Pharmacother 61(5):245–249
Kerjaschki D, Sharkey DJ, Farquhar MG (1984) Identification and characterization of podocalyxin–the major sialoprotein of the renal glomerular epithelial cell. J Cell Biol 98(4):1591–1596
Kerosuo L, Juvonen E, Alitalo R, Gylling M, Kerjaschki D, Miettinen A (2004) Podocalyxin in human haematopoietic cells. Br J Haematol 124:809–818
Kershaw DB, Beck SG, Wharram BL, Wiggins JE, Goyal M, Thomas PE, Wiggins RC (1997) Molecular cloning and characterization of human podocalyxin-like protein. Orthologous relationship to rabbit PCLP1 and rat podocalyxin. J Biol Chem 272:15708–15714
Casey G, Neville PJ, Liu X, Plummer SJ, Cicek MS, Krumroy LM, Curran AP, McGreevy MR, Catalona WJ, Klein EA, Witte JS (2006) Podocalyxin variants and risk of prostate cancer and tumor aggressiveness. Hum Mol Genet 15:735–741
Larrucea S, Butta N, Arias-Salgado EG, Alonso-Martin S, Ayuso MS, Parrilla R (2008) Expression of podocalyxin enhances the adherence, migration, and intercellular communication of cells. Exp Cell Res 314:2004–2015
Kestilä M, Lenkkeri U, Männikkö M, Lamerdin J, McCready P, Putaala H, Ruotsalainen V, Morita T, Nissinen M et al (1998) Positionally cloned gene for a novel glomerular protein—nephrin—is mutated in congenital nephrotic syndrome. Mol Cell 1:575–582
Aström E, Rinta-Valkama J, Gylling M, Ahola H, Miettinen A, Timonen T, Holthöfer H (2006) Nephrin in human lymphoid tissues. Cell Mol Life Sci 63:498–504
Karalezli G, Gögüş O, Bedük Y, Köküuslu C, Sarica K, Kutsal O (1993) Histopathologic effects of extracorporeal shock wave lithotripsy on rabbit kidney. Urol Res 21(1):67–70
Sarica K, Ozer G, Soygür T, Yaman O, Ozer E, Ustün H, Yaman LS, Göğüş O (1997) Preservation of shock-wave-induced renal histologic changes by dermatan sulfate. Urology 49(1):145–150
Krysiewicz S (1992) Complications of renal extracorporeal shock wave lithotripsy reviewed. Urol Radiol 13(3):139–145
Biri H, Oztürk HS, Büyükkoçak S, Kaçmaz M, Cimen MY, Unal D, Birey M, Bozkirli I, Durak I (1998) Antioxidant defense potential of rabbit renal tissues after ESWL: protective effects of antioxidant vitamins. Nephron 79(2):181–185
Kaude JV, Williams CM, Miliner MR, Scott KN, Finlayson B (1985) Renal morphology and function immediately after extracorporeal shock-wave lithotripsy. AJR Am J Roentgenol 145(2):305–313
Bomanji J, Boddy S, Britton KE, Nimmon CC, Whitfield HN (1987) Radionuclide evaluation pre and post-extracorporeal shock wave lithotripsy for renal calculi. J Nucl Med 28(8):1284–1289
Sarıca K, Kosar A, Yaman O, Bedük Y, Durak I, Göğüş O, Kavukçu M (1996) Evaluation of ischemia after SWL: detection of free oxygen radical scavenger enzymes in renal parenchyma subjected to high-energy shock waves. Urol Int 57(4):221–223
Neal DE Jr, Kaack MB, Harmon EP, Puyau F, Morvant A, Richardson E, Thomas R (1991) Renin production after experimental extracorporeal shock wave lithotripsy: a primate model. J Urol 146(2 (Pt 2)):548–550
Strohmaier WL, Carl AM, Wilbert DM, Bichler KH (1996) Effects of extracorporeal shock wave lithotripsy on plasma concentrations of endothelin and renin in humans. J Urol 155(1):48–51
Yurdakul T, Kulaksizoglu H, Pişkin MM, Avunduk MC, Ertemli E, Gokçe G, Barişkaner H, Byükbaş S, Kocabas V (2010) Combination antioxidant effect of α-tocoferol and erdosteine in ischemia-reperfusion injury in rat model. Int Urol Nephrol 42(3):647–655
Thamilselvan S, Byer KJ, Hackett RL, Khan SR (2000) Free radical scavengers, catalase and superoxide dismutase provide protection from oxalate-associated injury to LLC-PK1 and MDCK cells. J Urol 164(1):224–229
Zhu B, Li XF, Zhu XL, Lin Y, Zhong S, Zhu CF, Tang XL, Hu YQ, Cheng XX, Wang YJ (2014) ELISA analysis of urinary nephrin and podocalyxin standardized by aquaporin-2 in adult patients with nephrotic syndrome. J Nephrol 27(4):411–417
Matovinović MS (2009) Podocyte ınjury in glomerular diseases. eJIFCC 20(1):21–27
Whiteside C, Porutis K, Cameron R, Thomson J (1989) Glomerular epithelial detachment not reduced charge density correlates with proteinuria in adriamycin and puromycin nephrosis. Lab Investig 61:650–660
Hara M, Yamamoto T, Yanagihara T, Takada T, Itoh M, Adachi Y, Yoshizumi A, Kawasaki K, Kihara I (1995) Urinary excretion of podocalyxin indicates glomerular epithelial cell injuries in glomerulonephritis. Nephron 69:397–403
Andersson M, Nilsson U, Hjalmarsson C, Haraldsson B, Nystrom JS (2007) Mild renal ischemia-reperfusion reduces charge and size selectivity of the glomerular barrier. Am J Physiol Renal Physiol 292:F1802–F1809
Koop K, Eikmans M, Baelde HJ, Kawachi H, De Heer E, Paul LC, Bruijn JA (2003) Expression of podocyte-associated molecules in acquired human kidney diseases. J Am Soc Nephrol 14(8):2063–2071
Jim B, Ghanta M, Qipo A, Fan Y, Chuang PY, Cohen HW, Abadi M, Thomas DB, He JC (2012) Dysregulated nephrin in diabetic nephropathy of type 2 diabetes: a cross sectional study. PLoS One 7(5):e36041
Hara A, Wada T, Furuichi K, Sakai N, Kawachi H, Shimizu F, Shibuya M, Matsushima K, Yokoyama H, Egashira K, Kaneko S (2006) Blockade of VEGF accelerates proteinuria, via decrease in nephrin expression in rat crescentic glomerulonephritis. Kidney Int 69(11):1986–1995
Shao Y, Connors BA, Evan AP, Willis LR, Lıfshıtz DA, Lıngeman JE (2003) Morphological changes induced in the pig kidney by extracorporeal shockwave lithotripsy: nephron injury. Anat Rec A Discov Mol Cell Evol Biol 275(1):979–989
Wang Y, Zhao S, Loyd S, Groome LJ (2012) Increased urinary excretion of nephrin, podocalyxin, and βig-h3 in women with preeclampsia. Am J Physiol Renal Physiol 302(9):F1084–F1089
Obermüller N, Geiger H, Weipert C, Urbschat A (2014) Current developments in early diagnosis of acute kidney injury. Int Urol Nephrol 46(1):1–7
Sirota JC, Klawitter J, Edelstein CL (2011) Biomarkers of acute kidney injury. J Toxicol 2011:328120
Aydin HR, Irkilata L, Aydin M, Daggulli M, Taskin MH, Demirel HC, Adanur S, Moral C, Atilla MK, Sancaktutar AA (2016) The application of kidney injury molecule-1 to determine the duration between shock wave lithotripsy sessions. J Endourol 30(1):109–113
Turk C, Petrik A, Seitz C, Skolarikos A, Thomas K, Neisius A et al (2018) Guidelines on urolithiasis (limited text update March 2017), pp 1–81. http://uroweb.org. 13 Mar 2017
Kira VM, Fagundes DJ, Bandeira CO, Fagundes AT, Ortiz V (2007) The repeated extracorporeal shockwaves and the renal parenchyma injury on normal and diabetic rats. Acta Cir Bras 22(4):285–290
Author information
Authors and Affiliations
Contributions
KH: project development, data collection, data analysis, manuscript writing. BF: project development, data collection. AN: data collection, manuscript editing. Kİ: data collection, manuscript editing. ŞE: project development, data collection, data analysis, manuscript editing. SK: data analysis, manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare to have no conflicts of interest. There was no funding for this study.
Research involving human participants and/or animals
This article does not contain any studies with animals performed by any of the authors.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study protocol (2018/01-01) was approved by the ethics committee of the Health Sciences University Erzurum Regional Education and Research Hospital.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kocatürk, H., Atasoy, N., Bedir, F. et al. Evaluation of the urinary podocalyxin and nephrin excretion levels to determine a safe time interval between two sessions of SWL for renal stones: a non randomized exploratory study. Int Urol Nephrol 51, 1727–1734 (2019). https://doi.org/10.1007/s11255-019-02229-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-019-02229-8