Abstract
Purpose
To determine if multiparametric MRI (mpMRI) derived filtration-histogram based texture analysis (TA) can differentiate between different Gleason scores (GS) and the D’Amico risk in prostate cancer.
Methods
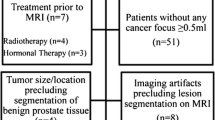
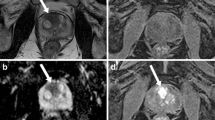
We retrospectively studied patients whose pre-operative 1.5T mpMRI had shown a visible tumour and who subsequently underwent radical prostatectomy (RP). Guided by tumour location from the histopathology report, we drew a region of interest around the dominant visible lesion on a single axial slice on the T2, Apparent Diffusion Coefficient (ADC) map and early arterial phase post-contrast T1 image. We then performed TA with a filtration-histogram software (TexRAD -Feedback Medical Ltd, Cambridge, UK). We correlated GS and D’Amico risk with texture using the Spearman’s rank correlation test.
Results
We had 26 RP patients with an MR-visible tumour. Mean of positive pixels (MPP) on ADC showed a significant negative correlation with GS at coarse texture scales. MPP showed a significant negative correlation with GS without filtration and with medium filtration. MRI contrast texture without filtration showed a significant, negative correlation with D’Amico score. MR T2 texture showed a significant, negative correlation with the D’Amico risk, particularly at textures without filtration, medium texture scales and coarse texture scales.
Conclusion
ADC map mpMRI TA correlated negatively with GS, and T2 and post-contrast images with the D’Amico risk score. These associations may allow for better assessment of disease prognosis and a non-invasive method of follow-up for patients on surveillance. Further, identifying clinically significant prostate cancer is essential to reduce harm from over-diagnosis and over-treatment.


Similar content being viewed by others
Change history
29 May 2019
Unfortunately, in the original article one co-author’s name is missing. The co-author name and affiliation is given as follows.
References
D’Amico A (1998) Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280(11):969
Klotz L, Zhang L, Lam A, Nam R, Mamedov A, Loblaw A (2010) Clinical results of long-term follow-up of a large, active surveillance cohort with localized prostate cancer. J Clin Oncol 28:126–131
Heidenreich A, Bastian PJ, Bellmunt J et al (2014) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol 65:124–137. https://doi.org/10.1016/j.eururo.2013.1009.1046 (Epub 2013 Oct 1016)
Nice.org.uk. (2018). Prostate cancer: diagnosis and management |Guidance and guidelines| NICE. http://www.nice.org.uk/guidance/cg175. Accessed 28 May 2018
Delongchamps N, Rouanne M, Flam T, Beuvon F, Liberatore M, Zerbib M, Cornud F (2011) Multiparametric magnetic resonance imaging for the detection and localization of prostate cancer: combination of T2-weighted, dynamic contrast-enhanced and diffusion-weighted imaging. BJU Int 107(9):1411–1418
Berglund R, Masterson T, Vora K, Eggener S, Eastham J, Guillonneau B (2008) Pathological upgrading and up staging with immediate repeat biopsy in patients eligible for active surveillance. J Urol 180(5):1964–1968
Kasivisvanathan V, Rannikko AS, Borghi M, Panebianco V, Mynderse LA, Vaarala MH et al (2018) MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med 378(19):1767–1777
Sahibzada I, Batura D, Hellawell G (2016) Validating multiparametric MRI for diagnosis and monitoring of prostate cancer in patients for active surveillance. Int Urol Nephrol 48(4):529–533
Barentsz J, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G, Rouviere O, Logager V, Fütterer J (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22(4):746–757
Eliat P, Olivié D, Saïkali S, Carsin B, Saint-Jalmes H, de Certaines J (2012) Can dynamic contrast-enhanced magnetic resonance imaging combined with texture analysis differentiate malignant glioneuronal tumors from other glioblastoma? Neurol Res Int 2012:1–7
Parikh J, Selmi M, Charles-Edwards G, Glendenning J, Ganeshan B, Verma H, Mansi J, Harries M, Tutt A, Goh V (2014) Changes in primary breast cancer heterogeneity may augment midtreatment MR imaging assessment of response to neoadjuvant chemotherapy. Radiology 272(1):100–112
De Cecco C, Ganeshan B, Ciolina M, Rengo M, Meinel F, Musio D, De Felice F, Raffetto N, Tombolini V, Laghi A (2015) Texture analysis as imaging biomarker of tumoral response to neoadjuvant chemoradiotherapy in rectal cancer patients studied with 3-t magnetic resonance. Invest Radiol 50(4):239–245
Ganeshan B, Panayiotou E, Burnand K, Dizdarevic S, Miles K (2011) Tumour heterogeneity in non-small cell lung carcinoma assessed by CT texture analysis: a potential marker of survival. Eur Radiol 22(4):796–802
Miles K, Ganeshan B, Griffiths M, Young R, Chatwin C (2009) Colorectal cancer: texture analysis of portal phase hepatic CT images as a potential marker of survival. Radiology 250(2):444–452
Ganeshan B, Skogen K, Pressney I, Coutroubis D, Miles K (2012) Tumour heterogeneity in oesophageal cancer assessed by CT texture analysis: preliminary evidence of an association with tumour metabolism, stage, and survival. Clin Radiol 67(2):157–164
Davnall F, Yip C, Ljungqvist G, Selmi M, Ng F, Sanghera B, Ganeshan B, Miles K, Cook G, Goh V (2012) Assessment of tumor heterogeneity: an emerging imaging tool for clinical practice? Insights Imaging 3(6):573–589
Miles K, Ganeshan B, Hayball M (2013) CT texture analysis using the filtration-histogram method: what do the measurements mean? Cancer Imaging 13(3):400–406
Donati O, Mazaheri Y, Afaq A, Vargas H, Zheng J, Moskowitz C, Hricak H, Akin O (2014) Prostate cancer aggressiveness: assessment with whole-lesion histogram analysis of the apparent diffusion coefficient. Radiology 271(1):143–152
Ytre-Hauge S, Dybvik J, Lundervold A, Salvesen Ø, Krakstad C, Fasmer K, Werner H, Ganeshan B, Høivik E, Bjørge L, Trovik J, Haldorsen I (2018) Preoperative tumor texture analysis on MRI predicts high-risk disease and reduced survival in endometrial cancer. J Magn Reson Imaging 48(6):1637–1647
Schob S, Meyer H, Dieckow J, Pervinder B, Pazaitis N, Höhn A, Garnov N, Horvath-Rizea D, Hoffmann K, Surov A (2017) Histogram analysis of diffusion weighted imaging at 3T is useful for prediction of lymphatic metastatic spread, proliferative activity, and cellularity in thyroid cancer. Int J Mol Sci 18(4):821
Health Research Authority. (2019). Homepage. https://www.hra.nhs.uk/. Accessed 3 Feb 2019
Pierorazio P, Walsh P, Partin A, Epstein J (2013) Prognostic Gleason grade grouping: data based on the modified Gleason scoring system. BJU Int 111(5):753–760
Dikaios N, Alkalbani J, Sidhu H, Fujiwara T, Abd-Alazeez M, Kirkham A, Allen C, Ahmed H, Emberton M, Freeman A, Halligan S, Taylor S, Atkinson D, Punwani S (2014) Logistic regression model for diagnosis of transition zone prostate cancer on multi-parametric MRI. Eur Radiol 25(2):523–532
Langer D, van der Kwast T, Evans A, Trachtenberg J, Wilson B, Haider M (2009) Prostate cancer detection with multi-parametric MRI: logistic regression analysis of quantitative T2, diffusion-weighted imaging, and dynamic contrast-enhanced MRI. J Magn Reson Imaging 30(2):327–334
Oto A, Kayhan A, Jiang Y, Tretiakova M, Yang C, Antic T, Dahi F, Shalhav A, Karczmar G, Stadler W (2010) Prostate cancer: differentiation of central gland cancer from benign prostatic hyperplasia by using diffusion-weighted and dynamic contrast-enhanced MR imaging. Radiology 257(3):715–723
Wibmer A, Hricak H, Gondo T, Matsumoto K, Veeraraghavan H, Fehr D, Zheng J, Goldman D, Moskowitz C, Fine S, Reuter V, Eastham J, Sala E, Vargas H (2015) Haralick texture analysis of prostate MRI: utility for differentiating non-cancerous prostate from prostate cancer and differentiating prostate cancers with different Gleason scores. Eur Radiol 25(10):2840–2850
Bates A, Miles K (2017) Prostate-specific membrane antigen PET/MRI validation of MR textural analysis for detection of transition zone prostate cancer. Eur Radiol 27(12):5290–5298
Sidhu H, Benigno S, Ganeshan B, Dikaios N, Johnston E, Allen C, Kirkham A, Groves A, Ahmed H, Emberton M, Taylor S, Halligan S, Punwani S (2016) Textural analysis of multiparametric MRI detects transition zone prostate cancer. Eur Radiol 27(6):2348–2358
Barry MJ (2001) Clinical practice prostate-specific-antigen testing for early diagnosis of prostate cancer. N Engl J Med 344(18):1373–1377
Yoon H-J, Kim Y, Chung J, Kim BS (2018) Predicting neo-adjuvant chemotherapy response and progression-free survival of locally advanced breast cancer using textural features of intratumoral heterogeneity on F-18 FDG PET/CT and diffusion-weighted MR imaging. Breast J. https://doi.org/10.1111/tbj.13032
Dickinson L, Ahmed H, Allen C, Barentsz J, Carey B, Futterer J, Heijmink S, Hoskin P, Kirkham A, Padhani A, Persad R, Puech P, Punwani S, Sohaib A, Tombal B, Villers A, van der Meulen J, Emberton M (2011) Magnetic resonance imaging for the detection, localisation, and characterisation of prostate cancer: recommendations from a European consensus meeting. Eur Urol 59(4):477–494
Taira A, Merrick G, Galbreath R, Andreini H, Taubenslag W, Curtis R, Butler W, Adamovich E, Wallner K (2009) Performance of transperineal template-guided mapping biopsy in detecting prostate cancer in the initial and repeat biopsy setting. Prostate Cancer Prostatic Dis 13(1):71–77
Bratan F, Niaf E, Melodelima C, Chesnais A, Souchon R, Mège-Lechevallier F, Colombel M, Rouvière O (2013) Influence of imaging and histological factors on prostate cancer detection and localisation on multiparametric MRI: a prospective study. Eur Radiol 23(7):2019–2029
Lee S, Park K, Choi K, Lim B, Kim J, Lee S, Chung B (2010) Is endorectal coil necessary for the staging of clinically localized prostate cancer? Comparison of non-endorectal versus endorectal MR imaging. World J Urol 28(6):667–672
Langer D, van der Kwast T, Evans A, Sun L, Yaffe M, Trachtenberg J, Haider M (2008) Intermixed normal tissue within prostate cancer: effect on MR imaging measurements of apparent diffusion coefficient and T2—sparse versus dense cancers. Radiology 249(3):900–908
Funding
BG and AA work at University College London Hospital/University College London (UCLH/UCL), which received a proportion of the funding from the UK’s Department of Health’s National Institute of Health Research, Biomedical Research Centre’s funding scheme. AA has also received funding from the London North West Healthcare Charitable Fund and the UCL Experimental Cancer Medicine Centre.
Author information
Authors and Affiliations
Contributions
AA: study conceptualisation. BG: MR image analysis (guidance), statistics and inputs to the manuscript. DB: Study conceptualisation, clinical data collection, manuscript revisions. JS: MR image analysis and manuscript revisions. MH: Initial draft, bibliography and manuscript revisions. SM: Clinical data collection and validation. Control of data and final manuscript approval were undertaken by the last author (study-guarantor).
Corresponding author
Ethics declarations
Conflict of interest
BG is a Director of Feedback Medical Ltd (www.fbkmed.com) and Shareholder of Feedback Plc, Cambridge (which wholly owns Feedback Medical Ltd.), a UK-based medical imaging software company and manufacturer of TexRAD texture analysis research software used in this study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hameed, M., Ganeshan, B., Shur, J. et al. The clinical utility of prostate cancer heterogeneity using texture analysis of multiparametric MRI. Int Urol Nephrol 51, 817–824 (2019). https://doi.org/10.1007/s11255-019-02134-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-019-02134-0