Abstract
Purpose
To investigate the effect of rapamycin on TGFβ1 and MMP1 expression in a rabbit model of urethral stricture.
Methods
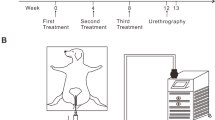
Twenty-four adult New Zealand male rabbits underwent an electrocoagulation of the bulbar urethra with a 13Fr pediatric resectoscope. Then rabbits were randomly divided into three groups: (1) normal control group: normal saline (NS), (2) the vehicle control group: dimethyl sulfoxide (DMSO), and (3) the treatment group: effective-dose rapamycin in DMSO (Ra), with 12, 6, and 6 rabbits in each group, respectively. Drugs were given by urethral irrigation daily for 4 weeks. Urethral tissue was harvested for histological and molecular analyses. TGFβ1 and MMP1 expression levels were evaluated by real-time quantitative PCR and immunohistochemistry.
Results
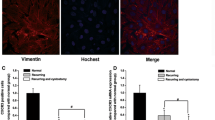
Ten, six, and six rabbits were evaluated finally in Ra, DMSO, and NS group, respectively. Histological examination revealed the distribution of fibrosis and the degree of collagen deposition in the Ra group were smaller and slighter than the two control groups. Collagen content was significantly less in the Ra group than in the DMSO group (P < 0.001) and the NS group (P < 0.001). qRT-PCR analysis showed a higher expression of MMP1 mRNA in the Ra group than in the DMSO group (P < 0.001) and the NS group (P < 0.001). Immunohistochemistry showed the protein levels of MMP1 in the Ra group were significantly increased when compared with the DMSO group (P < 0.01) and the NS group (P < 0.01). On the other hand, no statistical difference could be found between every two groups in both mRNA and protein levels of TGFβ1.
Conclusions
Rapamycin enhances the expression of MMP1 in a rabbit model of urethral stricture, but has no direct effect on the expression of TGFβ1.




Similar content being viewed by others
References
Mundy AR, Andrich DE (2011) Urethral strictures. BJU Int 107(1):6–26. doi:10.1111/j.1464-410X.2010.09800.x
Osman NI, Mangera A, Chapple CR (2013) A systematic review of surgical techniques used in the treatment of female urethral stricture. Eur Urol 64(6):965–973. doi:10.1016/j.eururo.2013.07.038
Hampson LA, McAninch JW, Breyer BN (2014) Male urethral strictures and their management. Nat Rev Urol 11(1):43–50. doi:10.1038/nrurol.2013.275
Meeks JJ, Erickson BA, Granieri MA, Gonzalez CM (2009) Stricture recurrence after urethroplasty: a systematic review. J Urol 182(4):1266–1270. doi:10.1016/j.juro.2009.06.027
Cavalcanti AG, Costa WS, Baskin LS, McAninch JA, Sampaio FJ (2007) A morphometric analysis of bulbar urethral strictures. BJU Int 100(2):397–402. doi:10.1111/j.1464-410X.2007.06904.x
Kajdaniuk D, Marek B, Borgiel-Marek H, Kos-Kudla B (2013) Transforming growth factor beta1 (TGFbeta1) in physiology and pathology. Endokrynol Pol 64(5):384–396. doi:10.5603/EP.2013.0022
Page-McCaw A, Ewald AJ, Werb Z (2007) Matrix metalloproteinases and the regulation of tissue remodelling. Nat Rev Mol Cell Biol 8(3):221–233. doi:10.1038/nrm2125
Da-Silva EA, Sampaio FJ, Dornas MC, Damiao R, Cardoso LE (2002) Extracellular matrix changes in urethral stricture disease. J Urol 168(2):805–807
Chong T, Fu DL, Li HC, Zhang HB, Zhang P, Gan WM, Wang ZM (2011) Rapamycin inhibits formation of urethral stricture in rabbits. J Pharmacol Exp Ther 338(1):47–52. doi:10.1124/jpet.110.178624
Wu MJ, Wen MC, Chiu YT, Chiou YY, Shu KH, Tang MJ (2006) Rapamycin attenuates unilateral ureteral obstruction-induced renal fibrosis. Kidney Int 69(11):2029–2036. doi:10.1038/sj.ki.5000161
Chen G, Chen H, Wang C, Peng Y, Sun L, Liu H, Liu F (2012) Rapamycin ameliorates kidney fibrosis by inhibiting the activation of mTOR signaling in interstitial macrophages and myofibroblasts. PLoS ONE 7(3):e33626. doi:10.1371/journal.pone.0033626
Wang W, Yan J, Wang H, Shi M, Zhang M, Yang W, Peng C, Li H (2014) Rapamycin ameliorates inflammation and fibrosis in the early phase of cirrhotic portal hypertension in rats through inhibition of mTORC1 but not mTORC2. PLoS ONE 9(1):e83908. doi:10.1371/journal.pone.0083908
Poulalhon N, Farge D, Roos N, Tacheau C, Neuzillet C, Michel L, Mauviel A, Verrecchia F (2006) Modulation of collagen and MMP-1 gene expression in fibroblasts by the immunosuppressive drug rapamycin: a direct role as an antifibrotic agent? J Biol Chem 281(44):33045–33052. doi:10.1074/jbc.M606366200
Tamaki Z, Asano Y, Kubo M, Ihn H, Tada Y, Sugaya M, Kadono T, Sato S (2014) Effects of the immunosuppressant rapamycin on the expression of human alpha2(I) collagen and matrix metalloproteinase 1 genes in scleroderma dermal fibroblasts. J Dermatol Sci 74(3):251–259. doi:10.1016/j.jdermsci.2014.02.002
Jaidane M, Ali-El-Dein B, Ounaies A, Hafez AT, Mohsen T, Bazeed M (2003) The use of halofuginone in limiting urethral stricture formation and recurrence: an experimental study in rabbits. J Urol 170(5):2049–2052. doi:10.1097/01.ju.0000091262.01493.e3
Santucci RA, Joyce GF, Wise M (2007) Male urethral stricture disease. J Urol 177(5):1667–1674. doi:10.1016/j.juro.2007.01.041
Steenkamp JW, Heyns CF, de Kock ML (1997) Internal urethrotomy versus dilation as treatment for male urethral strictures: a prospective, randomized comparison. J Urol 157(1):98–101
Ng CF, Chan SY (2007) Re: Hamid Mazdak, Iraj Meshki and Fatemeh Ghassami. Effect of mitomycin C on anterior urethral stricture recurrence after internal urethrotomy. Eur Urol 2007;51:1089–92. Eur Urol 52(3):930. doi:10.1016/j.eururo.2007.05.014
Sangkum P, Yafi FA, Kim H, Bouljihad M, Ranjan M, Datta A, Mandava SH, Sikka SC, Abdel-Mageed AB, Moparty K, Hellstrom WJ (2015) Collagenase Clostridium histolyticum (Xiaflex) for the treatment of urethral stricture disease in a rat model of urethral fibrosis. Urology 86(3):647 e641–646. doi:10.1016/j.urology.2015.06.013
Uyeturk U, Gucuk A, Firat T, Kemahli E, Kukner A, Ozyalvacli ME (2014) Effect of mitomycin, bevacizumab, and 5-fluorouracil to inhibit urethral fibrosis in a rabbit model. J Endourol 28(11):1363–1367. doi:10.1089/end.2014.0420
Krane LS, Gorbachinsky I, Sirintrapun J, Yoo JJ, Atala A, Hodges SJ (2011) Halofuginone-coated urethral catheters prevent periurethral spongiofibrosis in a rat model of urethral injury. J Endourol 25(1):107–112. doi:10.1089/end.2010.0514
Yoshizaki A, Yanaba K, Yoshizaki A, Iwata Y, Komura K, Ogawa F, Takenaka M, Shimizu K, Asano Y, Hasegawa M, Fujimoto M, Sato S (2010) Treatment with rapamycin prevents fibrosis in tight-skin and bleomycin-induced mouse models of systemic sclerosis. Arthritis Rheum 62(8):2476–2487. doi:10.1002/art.27498
Zhu J, Wu J, Frizell E, Liu SL, Bashey R, Rubin R, Norton P, Zern MA (1999) Rapamycin inhibits hepatic stellate cell proliferation in vitro and limits fibrogenesis in an in vivo model of liver fibrosis. Gastroenterology 117(5):1198–1204
Border WA, Noble NA (1994) Transforming growth factor beta in tissue fibrosis. N Engl J Med 331(19):1286–1292. doi:10.1056/nejm199411103311907
Massague J (2012) TGFbeta signalling in context. Nat Rev Mol Cell Biol 13(10):616–630. doi:10.1038/nrm3434
Milani BY, Milani FY, Park DW, Namavari A, Shah J, Amirjamshidi H, Ying H, Djalilian AR (2013) Rapamycin inhibits the production of myofibroblasts and reduces corneal scarring after photorefractive keratectomy. Invest Ophthalmol Vis Sci 54(12):7424–7430. doi:10.1167/iovs.13-12674
Zoncu R, Efeyan A, Sabatini DM (2011) mTOR: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol 12(1):21–35. doi:10.1038/nrm3025
Uitto J, Kouba D (2000) Cytokine modulation of extracellular matrix gene expression: relevance to fibrotic skin diseases. J Dermatol Sci 24(Suppl. 1):S60–S69
Acknowledgments
We appreciate Zhao XG (Xi’an Jiaotong University) for his help with pathological analysis and Li M (Xi’an Jiaotong University) for her help with statistical analysis. And we also appreciate the National Natural Science Foundation of China (No. 30972977) that provided the funds for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no competing interests exist.
Human and animal rights
All experiments were performed according to the guidelines of the committee of animal research at Xi’an Jiaotong University and the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Rights and permissions
About this article
Cite this article
Huang, S.L., Fu, D.L., Li, H.C. et al. The effect of rapamycin on TGFβ1 and MMP1 expression in a rabbit model of urethral stricture. Int Urol Nephrol 48, 717–723 (2016). https://doi.org/10.1007/s11255-016-1227-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-016-1227-x