Abstract
Introduction
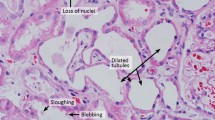
Indinavir, a protease inhibitor that is commonly used to treat HIV infection, may cause crystal formation within the renal tubules when urine pH is above 3.5. Crystallization in the urine may lead to intrarenal crystal deposition and acute renal failure (ARF).
Aim
To establish the beneficial urological management of acute renal failure caused by indinavir treatment of HIV/AIDS patients.
Patients––methods
Five HIV positive patients (four men, one woman) with a mean age of 32 years (range 28–36 years) were referred to our Department of Urology from an AIDS outpatient Clinic, because of the development of postrenal acute renal failure with continuously elevated creatinine and urea plasma levels after indinavir therapy. Among the initial therapeutic maneuvers, indinavir administration was interrupted for 1 week while bilateral double-J ureteral stents were inserted in all the HIV/AIDS patients, during the first 24–72 h to secure upper-tract drainage. Concurrently urine has been acidified by oral administration of the amino acid l-methionine and oral fluid intake was increased.
Results
All the patients responded well to the treatment and their renal function was effortlessly restored to normal within a few days.
Conclusion
HIV-positive patients receiving indinavir therapy might be complicated by acute renal failure, mainly due to intrarenal crystal deposition (tubules) or urolithiasis (postrenal obstruction). This adverse effect may simply manage by the discontinuation of indinavir administration, urine acidification, as well as the possible early insertion of bilateral double-J ureteral stents.
Similar content being viewed by others
References
Gendle DL, Stoller ML, Jarrett TW et al (1997) Protease inhibitor-induced urolithiasis. Urology 50:508–511
CrixivanR package insert. Merck and Co., lnc; U.S. Human health 1996
Salahuddin S, Hsu YS, Bucholz NP et al (2001) Is indinavir crystalluria anindicator for indinavir stone formation? AIDS 15(8):1079–1080
Grady BR, Munch LC, Hoven AD et al (1997) Urolithiasis associated with theprotease inhibitor indinavir. Urology 50(4):513–518
Chen IW, Vastag KJ, Lin JH (1995) High-performance liquid chromatographic determination of a potent and selective HIV protease inhibitor (L-735, 524) in rat, dog and monkey plasma. J chromatography B 672:111–117
Daudon M, Estepa L, Viard JP et al (1997) Urinary stones in HIV-1-positive patients treated with indinavir. The Lancet 349:1294–1295
Mascolini M (1996) The rolling uncertainties of antiprotease prescribing. J Int Assoc Physicians AIDS Care 2:6
Reilly RF, Tray K, Perazella MA (2001) Indinavir nephropathy revisited: a pattern of insidious renal failure with identifiable risk factors. Am J Kidney Dis 38(4):E23
Kopp JB, Miller KD, Mican JA (1997) Crystalluria and urinary tract abnormalities associated with indinavir. Ann Intern Med 127:119–125
Izzedine H, Launay-Vacher V, Deray G (2005) Antiviral drug-induced nephrotoxicity. Am J Kidney Dis 45:5
Mouratoff JG, Tokumoto J, Olson JL, Chertow GM (2000) Acute renal failure with interstitial nephritis in a patient with AIDS. Am J Kidney Dis 35(3):557–561
Berns JS, Cohen RM, Silverman M, Turner J (1997) Acute renal failure due to indinavir crystalluria and nephrolithiasis: report of two cases. Am J Kidney Dis 30:558–560
Kalaitzis C, Touloupidis S, Patris E, Lehrich K, Kuntz RM (2004) Indinavir-Urolithiasis bei HIV-positiven Patienten. Behandlung und Prophylaxe. Der Urologe A (43), Heft 2:168–171
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalaitzis, C., Passadakis, P., Giannakopoulos, S. et al. Urological management of indinavir-associated acute renal failure in HIV-positive patients. Int Urol Nephrol 39, 743–746 (2007). https://doi.org/10.1007/s11255-006-9154-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-006-9154-x