Abstract
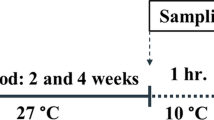
In order to increase the low-temperature resistance of Nile tilapia, the purpose of this study was to determine the potential effect of ω3 fatty acid incorporation in Oreochromis niloticus diet. To perform this, two experimental diets containing soybean oil (D1) and cod liver oil (D2) have been supplied to juvenile tilapia for 30 days. According to our results, similar improvements in the two diets have been recorded for growth performance of O. niloticus including the final body mass, specific growth rate, and feed conversion ratio. Our results showed that fish fed with diet D2 promoted high polyunsaturated fatty acids mainly n-3 series (PUFA (n-3)) percent, highlighting the increased levels of docosahexaenoic (DHA) and eicosapentaenoic (EPA) as well as the activation of their conversion enzyme ratios D5D and D6D desaturases. The second objective was to assess the effect of the two experimental diets on low water temperature tolerance. This was done by exposing juvenile fish at the end of the first experiment to 16, 14, 12, 10, and 8 °C for 12 h, 24 h, and 48 h. The sub-lethal LT50 of O. niloticus fed with diet D1 was 10.6, 11.4, and 13 °C respectively, after 12 h, 24 h, and 48 h. This pattern was commonly observed for O. niloticus fed with D2, showing that the subLT50 were 10.3, 11.1, and 12 °C during the same period. These results demonstrate that O. niloticus juveniles fed with diet D2 are more tolerant to low temperatures than those fed with diet D1.





Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
Abbreviations
- ARA:
-
Arachidonic acid
- D5D:
-
Δ5-Desaturase
- D6D:
-
Δ6-Desaturase
- D9D:
-
Δ9-Desaturase = stearoyl-CoA-desaturase
- DPA:
-
Docosapentaenoic acid
- DHA:
-
Docosahexaenoic acid
- EPA:
-
Eicosapentaenoic acid
- ELOVL6:
-
Elongase
- LA:
-
Linoleic acid
- ND:
-
None determined
- MUFA:
-
Monounsaturated fatty acids
- PUFA:
-
Polyunsaturated fatty acids
- SFA:
-
Saturated fatty acids
- subLT:
-
Sub-lethal temperatures
- subLT50 :
-
Sub-lethal temperatures 50
References
Al-Souti, A., Al-Sabahi, J., Soussi. B and Goddard, S., 2012. The effects of fish oil-enriched diets on growth, feed conversion and fatty acid content of red hybrid tilapia, Oreochromis sp, Food Chemistry,133,723-727.https://doi.org/10.1016/j.foodchem.2012.01.080.
Atwood, H. L., Tomasso, R., Webb, K. and Gatlin, D.M., 2003. Low temperature tolerance of Nile tilapia, 0reochromis niloticus: effects of environmental and dietary factors, Aquaatic Research , 34 (3), 241-251.https://doi.org/10.1046/j.1365-2109.2003.00811.x
Azaza, M.S., Dhraief, M.N. and Kraiem, M.M., 2008. Effects of water temperature on growth and sex ratio of juvenile Nile tilapia Oreochromis niloticus (Linnaeus) reared in geothermal waters in southern Tunisia. Journal of Thermal Biology, 33, 98–105. https://doi.org/10.1016/j.jtherbio.2007.05.007
Azaza, M.S., Mensi F., Wassim K., Abdelmouleh A., Brini B. and, Kraim M.M., 2009. Nutritional evaluation of waste date fruit as partial substitute for soybean meal in practical diets of juvenile Nile tilapia, Oreochromis niloticus L, Aquaculture Nutrition, 15, 262-272.
Azaza M.S., Legendre M., Kraim M.M. and Baras E., 2010. Size-dependent effects of fluctuating thermal regimes on the growth and size heterogeneity of Nile tilapia, Journal of Fish Biology, 76 (3), 669-683.
Bahurmiz, O.M. and Ng, W.K., 2007. Effects of dietary palm oil source on growth, tissue fatty acid composition and nutrient digestibility of red hybrid tilapia, Oreochromis sp., raised from stocking to marketable size, Aquaculture, 262, 382–392. https://doi.org/10.1016/j.aquaculture.2006.11.023
Barthet, V.J., 2008. (n-7) and (n-9) cis-monounsaturated fatty acid contents of 12 brassica species , Phytochemistry, 69(2), 411-411. https://doi.org/10.1016/j.phytochem.2007.08.016
Bell, J.G. and Sargen, J.R., 2003.Arachidonic acid in aquaculture feeds: current status and future opportunities, Aquaculture, 218, 491-499. doi: https://doi.org/10.1016/S0044-8486(02)00370-8.
Cecchi, G, Basini, S., andCastano, C., 1985. Methanolyse rapide des huiles en solvant, Revue française des corps gras, 4 , 163–164.
Charo-Karisa, H., Komen, H., Rezk, M. A., Ponzoni, R.W., Van Arendonk, J. and Bovenhuis, H., 2006 Heritability estimates and response to selection for growth of Nile tilapia (Oreochromis niloticus) in low-input earthen ponds, Aquaculture, 261(2), 479-486.
Cnaani, A., Hallerman, E.M., Ron, M., Weller, J. I., Indelman, M., Kashi, Y., Gall, G.A.E. and Hulata, G., 2003. Detection of a chromosomal region with two quantitative trait loci, affecting cold tolerance and fish size, in an F2 tilapia hybrid, Aquaculture, 223,117- 128. https://doi.org/10.1016/S0044-8486(03)00163-7
Cou, B.S.and Shiau, S.Y., 2001. Effect of Dietary Cod Liver Oil on Growth and Fatty Acids of Juvenile Hybrid tilapia, North American Journal of Aquaculture, 63, 277-284. https://doi.org/10.1577/15488454(2001)063<0277:EODCLO>2.0.CO;2
Corrêa, C.F., Nobrega, O.R., Block, J.M. and Fracalossi, D.M., 2018. Mixes of plant oils as fish oil substitutes for Nile tilapia at optimal and cold suboptimal temperature.Aquaculture, 497,82-90. https://doi.org/10.1016/j.aquaculture.2018.07.034
Da Costa, F., Robert, R., Quéré, C. and Wikfors, G., Soudant, P., 2015. Essential Fatty acid assimilation and synthesis in larvae of the bivalve Crassostrea gigas. Lipids, 50(5), 503-511. https://doi.org/10.1007/BF00004290.
Dhillon, R. S. and Fox, M. G., 2007. Growth-independent effects of temperature on age and size at maturity in Japanese Medaka (Oryzias latipes). Copeia, http://doi.org/10.1643/CI-02-098R1
FAO., 2014. The state of world fisheries and aquaculture p. 223.
Folch, J., Lees, M. and Stanley, G., 1957. A simple method of the isolation and purification of total lipids from animal tissues, Journal of Biological Chemistry, 226, 497- 509. https://doi.org/10.1016/S0021-9258(18)64849-5
FAO’s Fisheries and Aquaculture Department., 2020 Statistical Collections Capture Production and Aquaculture Production Datasets 1950–2019 (Release Date: March 2020). Available online: https://www.fao.org/shery/topic/16073. Accessed 25 March 2020
Flodmark , L. E. W., Vollestad, L. A. and Foresth, T.,2004. Performance of juvenile brown trout exposed to fluctuating water level and temperature. Journal of Fish Biology, 65, 460–470. https://doi.org/10.1111/j.1095-8649.2004.00463.x.
HMSO, U.K., 1994. Nutritional aspects of cardiovascular disease (report on health and social subject’s No. 46), London: HMSO.
Hofer SC, and Watts SA, 2002. Cold tolerance in genetically male tilapia (GMT registered), Oreochromis niloticus.World Aquaculture, 33,19–21
Kanazawa, A., Teshima, S., Sakamoto, M. and Awal, M.A., 1980. Requirement of Tilapia zilii for essential fatty acids. Bulletin of the Japanese Society for the Science of Fish , 46, 1353-1356. https://doi.org/10.2331/suisan.49.1127
Kotronen, A., Laakso, T.S., Westerbacka, J., Kiviluoto, T., Arola, J., Ruskeepää, A.L., Järvinen, H.Y. and Oresic; M., 2010. Comparison of lipid and fatty acid composition of the liver, subcutaneous and intra-abdominal adipose tissue, and serum, Obesity, 18(5), 937–944. https://doi.org/10.1038/oby.2009.326
Kraїem, M.M., Duvernay, J., 1981. Comparaison des températures limites de nage chez deux populations d’Ombres Communs Thymalus thymalus (L.), d’origine différente (Bavière et Scandinavie), Cybium, 5 (3), 45–49.
Li, S., Li, C., Madan,D., Florabelle,G. and Rex. D., 2002. Cold tolerance of three strains of Nile tilapia, Oreochromis niloticus, in China. Aquaculture, 213(1–4),123-129.
Li, E., Lim, C., Klesius, P.H. and Welker, T.L., 2013. Growth, body fatty acid composition, immune response, and resistance to Streptococcus iniae of hybrid tilapia, Oreochromis niloticus × Oreochromis Aureus, fed diets containing various levels of linoleic and linolenic acids. Journal of World Aquaculture Society, 44, 42–55. https://doi.org/10.1111/jwas.12014.
Lim, C., Yildirim-Aksoy, M. and Klesius, P. 2011. Lipid and fatty acid requirements of tilapias, North American Journal of aquaculture, 73, 188–193. https://doi.org/10.1080/15222055.2011.579032
Lu, D.L., Ma, Q., Wang, J., Li, L.Y., Han, S. L., Limbu, S.M., Li, D.L., Chen, L.Q., Zhang, M.L. and Du, Z.Y., 2019. Fasting enhances cold resistance in fish through stimulating lipid catabolism and autophagy. Journal of Physiology, 597(6), 1585–1603. https://doi.org/10.1113/JP277091
Marques, A., Teixeira, B., Barrento, S., Anacleto, P., Carvalho, M.L .and Nunes, M.L., 2010. Chemical composition of Atlantic spider crab Maja brachydactyla: human health implication. Journal of Food Composition and Analysis, 23, 230-237.
Montero, D., Robaina, L., Caballero, M.J., Ginés, R. and Izquierdo, M.S., 2005. Growth, feed utilization and flesh quality of European sea bass (Dicentrarchus labrax) fed diets containing vegetable oils: a time-course study on the effect of a re-feeding period with a 100% fish oil diet, Aquaculture, 248, 121-134.
National Research Council (NRC)., 2011. Nutrient Requirement of Fish. Committee on Animal Nutrition, Board on Agriculture,( National Academic Press. Washington, DC), pp, 102–134.
Nemova, N.N., Fokina, N.N., Nefedova, Z.A., Ruokolainen, T.R. and Bakhmet, I.N., 2013. Modifications of gill lipid composition in littoral and cultured blue mussels Mytilus edulis L. under the influence of ambient salinity, Polar Record, 49 (250), 272–277. https://doi.org/10.1017/S0032247412000629
Ng, W.K., Lim, P.K. and Sidek, H., 2001. The influence of a dietary lipid source on growth, muscle fatty acid composition and erythrocyte osmotic fragility of hybrid tilapia, Fish Physiology Biochemistry, 25, 301-310. https://doi.org/10.1023/A:1023271901111
Pandit, N. P. and Nakamura, M., 2010. Effect of High Temperature on Survival, Growth and Feed Conversion Ratio of Nile Tilapia, Oreochromis Niloticus, Our Nature, 8, 219-224. https://doi.org/10.3126/on.v8i1.4331
Rioux, V., Catheline, D., Bouriel, M., Legrand, P., 2005. Dietary myristic acid at physiologically relevant levels increases the tissue content of C20:5 n-3 and C20:3 n-6 in the rat. Reproduction Nutrition and Developpement, 45, 599-612. https://doi.org/10.1051/rnd:2005048
Rabei, A., Hichami, A., Beldi, H., Bellenger, S., Khan, N.A. and. Soltani,.N., 2018. Fatty acid composition, enzyme activities and metallothioneins in Donax trunculus (Mollusca, Bivalvia) from polluted and reference sites in the Gulf of Annaba (Algeria): pattern of recovery during transplantation. Environmental Pollution, 237, 900–907. https://doi.org/10.1016/j.envpol.2018.01.041
Senadheera, S.D., Turchini, G.M., Thanuthong. and T., Francis, D.S., 2011. Effects of dietary α-linolenic acid (18:3n-3)/linoleic acid (18:2n-6) ratio on fatty acid metabolism in Murray cod (Maccullochella peelii peelii). Journal of Agriculture Food Chemistry, 59, 1020-1030. https://doi.org/10.1021/jf104242y
Sigurd, O.H. and Sigurd O.S., 2008.The effect of temperature and fish size on growth, feed intake, food conversion efficiency and stomach evacuation rate of Atlantic salmon post-smolts. Aquaculture, 283 (1-4), 36-42. https://doi.org/10.1016/j.aquaculture.2008.06.042
Stickney, R.R. and McGeachin, R.B. 1983. Responses of Tilapia aurea to Semipurified Diets of Differing Fatty Acid Composition. In: L. Fishelson and Z. Yaron (Eds.), Proceedings of the International Symposium on Tilapia in Aquaculture, (Tel Aviv University Press,Tel Aviv, Israel), 346–355.
Takeuchi, Y., Yahag, N., Izumida, Y., Nishi, M., Kubota, M., Teraoka, Y., Yamamoto,T., Matsuzaka, T., Nakagawa, Y., Sekiya,M., Iizuka, Y., Ohashi, K., Yamada N., Kadowaki, T. and Shimano, H., 2010. Polyunsaturated fatty acids selectively suppress sterol regulatory element-binding protein-1 through proteolytic processing and autoloop regulatory circuit, Journal of Biology Chemistry, 285, 11681-11691
Teoh, C.Y., Turchini, G. M. and Ng, W.K., 2011. Genetically improved farmed Nile tilapia and red hybrid tilapia showed differences in fatty acid metabolism when fed diets with added fish oil or a vegetable oil blend, Aquaculture, 312, 126-136. https://doi.org/10.1016/j.aquaculture.2010.12.018
Tocher, D.R., 2010. Fatty acid requirements in ontogeny of marine and freshwater fish, Aquaculture Research, 41, 717-732. https://doi.org/10.1111/j.1365-2109.2008.02150.x.
Author information
Authors and Affiliations
Contributions
IC (investigation; visualization; writing—original draft; writing—review and editing); FG (investigation; visualization; writing—original draft; writing—review and editing); SB (investigation); SH (formal analysis); MEC (resources, funding acquisition); MSA (conceptualization, methodology, supervision).
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chetoui, I., Ghribi, F., Bejaoui, S. et al. Incorporation of ω3 fatty acids in the diets of Nile tilapia juvenile (Oreochromis niloticus L.): effects on growth performance, fatty acid composition, and tolerance to low temperature. Trop Anim Health Prod 54, 401 (2022). https://doi.org/10.1007/s11250-022-03394-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-022-03394-2