Abstract
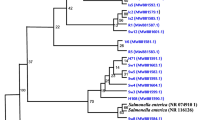
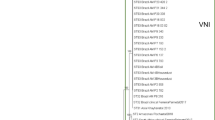
In this study, the prevalence of ESBL/pAmpC-producing Escherichia coli and their molecular characterization from cloacal swab samples were investigated. All samples were obtained from broiler flocks that are located in Hatay, Adana, and Mersin provinces of Turkey. Antimicrobial susceptibilities of the isolates were determined by disk diffusion method following the CLSI criteria. Genetic mechanisms mediating resistance in ESBL/pAmpC-producing E. coli isolates were identified by polymerase chain reaction (PCR) and followed by DNA sequencing. Phylogenetic groups and plasmid replicon types of the isolates were also investigated by PCR. The clonal relationship of selected isolates was investigated by enterobacterial repetitive intergenic consensus (ERIC)-PCR and multilocus sequence typing (MLST) method. Of 430 cloacal swab samples, 154 (35.8%) were positive for ESBL/pAmpC-producing E. coli. The ESBL/pAmpC type beta-lactamases were as follows: CMY-2 (n = 46), CMY-2 + TEM-1b (n = 63), SHV-12 (n = 5), SHV-12 + TEM-1b (n = 12), CTX-M-3 (n = 14), CTX-M-3 + TEM-1b (n = 1), CTX-M-15 (n = 4), CTX-M-15 + TEM-1b (n = 4), and CTX-M-1 (n = 3). Moreover, various rates of resistance to different antimicrobials were determined such as nalidixic acid (92.9%), ciprofloxacin (76%), sulfamethoxazole-trimethoprim (78.6%), tetracycline (73.4%), streptomycin (52.6%), chloramphenicol (44.2%), kanamycin (27.9%), tobramycin (24.7%), gentamicin (19.5%), and amikacin (0.6%). Furthermore, 148 (96.1%) isolates were found to be MDR. The ESBL/pAmpC-producing isolates were distributed into the following phylogroups: E (n = 61), B1 (n = 30), F (n = 20), A (n = 19), B2 (n = 11), D (n = 10), and C (n = 3). ERIC-PCR analysis showed 51 unrelated patterns. Out of the 28 selected isolates, the following sequence types (STs) were detected: ST354 (n = 3), ST114 (n = 3), ST5696 (n = 2), ST156 (n = 2), ST174 (n = 2), ST362 (n = 2), ST157 (n = 2), ST5114 (n = 2), ST6635, ST539, ST457, ST1640, ST95, ST5843, ST1158, ST10, ST648, and ST4248. The results of the current study revealed that broilers in Turkey are important reservoir of ESBL/pAmpC-producing E. coli, which suggest that these agents have a great potential of transmission to humans by food chain or direct contact.


Similar content being viewed by others
References
Ahmed, A.M., Motoi, Y., Sato, M., Maruyama, A., Watanabe, H., Fukumoto, Y., Shimamoto, T., 2007. Zoo animals as a reservoir of gram negative bacteria harboring integrons and antimicrobial resistance genes. Appl Environ Microbiol. 73, 6686–6690.
Aslantaş, Ö., Yilmaz, E.Ş., 2017. Prevalence and molecular characterization of extended-spectrum β-lactamase (ESBL) and plasmidic AmpC β-lactamase (pAmpC) producing Escherichia coli in dogs. J Vet Med Sci. 79, 1024–1030.
Aslantaş, Ö., Elmacıoğlu, S., Yılmaz, E.Ş., 2017. Prevalence and Characterization of ESBL- and AmpC-producing Escherichia coli from Cattle. Kafkas Univ Vet Fak Derg. 23, 63–67.
Bailey, J.K., Pinyon, J.L., Anantham, S., Hall R.M., 2010. Distribution of human commensal Escherichia coli phylogenetic groups. J Clin Microbiol. 48, 3455–3456.
Balouiri, M., Sadiki, M., Ibnsouda, S.K., 2016. Methods for in vitro evaluating antimicrobial activity: A review. J Pharm Anal. 6, 71–79.
Başaran Kahraman, B., Diren Sığırcı, B., Çelik, B., Gümüş, B., Metiner, K., Adıgüzel M.C, Bağcıgil, A.F., İkiz, S., Özgür, N.Y., Ak, S., 2016. Detection of extended-spectrum β-lactamase and AmpC β-lactamase producing Escherichia coli isolates from chickens. Kafkas Univ Vet Fak Derg. 22, 591–596.
Börjesson, S., Bengtsson, B., Jernberg, C., Englund, S., 2013. Spread of extended-spectrum beta-lactamase producing Escherichia coli isolates in Swedish broilers mediated by an incl plasmid carrying bla(CTX-M-1). Acta Vet. Scand. 55, 3.
Bortolaia, V., Guardabassi, L., Trevisani, M., Bisgaard, M., Venturi, L., Bojesen, A.M., 2010. High diversity of extended-spectrum beta-lactamases in Escherichia coli isolates from Italian broiler flocks. Antimicrob Agents Chemother. 54, 1623–1626.
Carattoli, A., Bertini, A., Villa, L., Falbo, V., Hopkins, K.L., Threlfall, E.J., 2005. Identification of plasmids by PCR-based replicon typing. J. Microbiol. Methods. 63, 219–228.
Carattoli, A., Zankari, E., García-Fernández, A., Voldby Larsen, M., Lund, O., Villa, L., Møller Aarestrup, F., Hasman, H., 2014. In silico detection and typing of plasmids using PlasmidFinder and plasmid multilocus sequence typing. Antimicrob. Agents Chemother. 58, 3895–3903.
Castellanos, L.R., Donado-Godoy, P., León, M., Clavijo, V., Arevalo, A., Bernal, J.F., Timmerman, A.J., Mevius, D.J., Wagenaar, J.A., Hordijk, J., 2017. High heterogeneity of Escherichia coli sequence types harbouring ESBL/AmpC genes on IncI1 Plasmids in the Colombian poultry chain. PLoS One. 12, e0170777.
Clermont, O., Bonacorsi, S., Bingen, E., 2000. Rapid and simple determination of the Escherichia coli phylogenetic group. Appl Environ Microbiol. 66, 4555–4558.
Clermont, O., Christenson, J.K., Denamur, E., Gordon, D.M. 2013. The Clermont Escherichia coli phylo-typing method revisited: improvement of specificity and detection of new phylo-groups. Environ Microbiol Rep. 5, 58–65.
Clinical and Laboratory Standard Institute (CLSI): Performance Standards for Antimicrobial Susceptibility Testing; Nineteenth Informational Supplements. CLSI Document M100-S19. Wayne, PA, USA, 2012.
Costa, D., Vinué, L., Poeta, P., Coelho, A.C., Matos, M., Sáenz, Y., Somalo, S., Zarazaga, M., Rodrigues, J., Torres, C., 2009. Prevalence of extended-spectrum beta-lactamase-producing Escherichia coli isolates in faecal samples of broilers. Vet Microbiol. 138, 339–344.
Day, M.J., Rodríguez, I., van Essen-Zandbergen, A., Dierikx, C., Kadlec, K., Schink, A.K., Wu, G., Chattaway, M.A., DoNascimento, V., Wain, J., Helmuth, R., Guerra, B., Schwarz, S., Threlfall, J., Woodward, M.J., Coldham, N., Mevius, D., Woodford, N., 2016. Diversity of STs, plasmids and ESBL genes among Escherichia coli from humans, animals and food in Germany, the Netherlands and the UK. J Antimicrob Chemother. 71, 1178–1182.
Dierikx, C.M., van der Goot, J.A., Smith, H.E., Kant, A., Mevius, D.J. 2013. Presence of ESBL/AmpC-producing Escherichia coli in the broiler production pyramid: a descriptive study. PLoS One. 8, e79005.
EFSA, ECDC, 2018. The European Union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2016. EFSA J. 16, 5182.
Ewers, C., Antao, E.M., Diehl, I., Philipp, H.C., Wieler, L.H., 2009. Intestine and environment of the chicken as reservoirs for extraintestinal pathogenic Escherichia coli strains with zoonotic potential. Appl Environ Microbiol. 75, 184–192.
Ewers, C., Bethe, A., Semmler, T., Guenther, S., Wieler, L.H., 2012. Extended-spectrum beta-lactamaseproducing and AmpC-producing Escherichia coli from livestock and companion animals, and their putative impact on public health: a global perspective. Clin Microbiol Infect. 18, 646–655.
Fischer, E.A., Dierikx, C.M., van Essen-Zandbergen, A., van Roermund, H.J., Mevius, D.J., Stegeman, A., Klinkenberg, D., 2014. The IncI1 plasmid carrying the blaCTX-M-1 gene persists in in vitro culture of a Escherichia coli strain from broilers. BMC Microbiol. 14, 77.
Giray, B., Uçar, F.B., Aydemir, S.Ş., 2016. Genotypic analysis of Escherichia coli strains that cause urosepsis in the Aegean region. Turk J Med Sci. 46, 1518–1527.
Hertz, F.B., Nielsen, J.B., Schønning, K., Littauer, P., Knudsen, J.D., Løbner-Olesen, A., Frimodt-Møller, N., 2016. Population structure of drug-susceptible,-resistant and ESBL-producing Escherichia coli from community-acquired urinary tract. BMC Microbiol. 16, 63.
Huijber, P.M.C., Graat, E.A.M., van Hoek, A.H.A.M., Veenman, C., de Jong, M.C.M., van Duijkeren, E., 2016. Transmission dynamics of extended-spectrum β-lactamase and AmpC β-lactamase-producing Escherichia coli in a broiler flock without antibiotic use. Prev Vet Med. 131, 12–19.
Huijbers, P.M.C., Graat, E.A.M., Haenen, A.P.J., van Santen, M.G., van Essen-Zandbergen, A., Mevius, D.J., van Duijkeren, E., van Hoek A.H.A.M., 2014. Extended-spectrum β-lactamase- and AmpC β-lactamase-producing Escherichia coli in broilers and people living and/or working on broiler farms: prevalence, risk factors, and molecular characteristics. J Antimicrob Chemother. 69, 2669–2675.
Ibrahim, D.R., Dodd, C.E., Stekel, D.J., Ramsden, S.J., Hobman, J.L., 2016. Multidrug resistant, extended spectrum β-lactamase (ESBL)-producing Escherichia coli isolated from a dairy farm. FEMS Microbiol Ecol. 92, fiw013
Jarlier, V., Nicolas, M.H., Fournier, G., Philippon, A., 1988. Extended broad-spectrum β- lactamases conferring transferable resistance to newer β-lactam agents in Enterobacteriaceae: hospital prevalence and susceptibility patterns. Rev Infect Dis. 10, 867–878.
Kameyama, M., Chuma, T., Yabata, J., Tominaga, K., Iwata, H., Okamoto, K., 2013. Prevalence and epidemiological relationship of CMY-2 AmpC β-lactamase and CTX-M extended-spectrum β-lactamase-producing Escherichia coli isolates from broiler farms in Japan. J Vet Med Sci. 75, 1009–1015.
Lambrecht, E., Van Meervenne, E., Boon, N., Van de Wiele, T., Wattiau, P., Herman, L., Heyndrickx, M., Van Coillie, E., 2018. Characterization of cefotaxime- and ciprofloxacin-resistant commensal Escherichia coli originating from Belgian farm animals indicates high antibiotic resistance transfer rates. Microb Drug Resist. 24, 707–717.
Liakopoulos, A., Olsen, B., Geurts, Y., Artursson, K., Berg, C., Mevius, D.J., Bonnedahl, J., 2016. Molecular Characterization of extended-spectrum-cephalosporin-resistant Enterobacteriaceae from wild kelp gulls in South America. Antimicrob Agents Chemother. 60, 6924–6927.
Lim, K.T., Yasin, R., Yeo, C.C., Puthucheary, S., Thong, K.L., 2009. Characterization of multidrug resistant ESBL-producing Escherichia coli isolates from hospitals in Malaysia. J Biomed Biotechnol. 2009, 165637.
Maamar, E., Hammami, S., Alonso, C.A., Dakhli, N., Abbassi, M.S., Ferjani, S., Hamzaoui, Z., Saidani, M., Torres, C., Boutiba-Ben Boubaker, I., 2016. High prevalence of extended-spectrum and plasmidic AmpC beta-lactamase-producing Escherichia coli from poultry in Tunisia. Int J Food Microbiol. 231, 69–75.
Maciuca, I.E., Williams, N.J., Tuchilus, C., Dorneanu, O., Guguianu, E., Carp-Carare, C., Rimbu, C., Timofte, D., 2015. High prevalence of Escherichia coli-producing CTX-M-15 extended-spectrum beta-lactamases in poultry and human clinical isolates in Romania. Microb Drug Resist. 21, 651–662.
Madoshi, B.P., Kudirkiene, E., Mtambo, M.M., Muhairwa, A.P., Lupindu, A.M., Olsen, J.E., 2016. Characterisation of commensal Escherichia coli isolated from apparently healthy cattle and their attendants in Tanzania. PLoS One, 11, e0168160.
Mesa, R.J., Blanc, V., Blanch, A.R., Cortés, P., González, J.J., Lavilla, S., Miró, E., Muniesa, M., Saco, M., Tórtola, M.T., Mirelis, B., Coll, P., Llagostera, M., Prats, G., Navarro, F., 2006. Extended-spectrum beta-lactamase-producing Enterobacteriaceae in different environments (humans, food, animal farms and sewage). J Antimicrob Chemother. 58, 211–215.
Mo, S.S., Slettemeås, J.S., Berg, E.S., Norström, M., Sunde, M., 2016. Plasmid and host strain characteristics of Escherichia coli resistant to extended-spectrum cephalosporins in the Norwegian broiler production. PLoS One. 11, e0154019.
Nilsson, O., Börjesson, S., Landén, A., Bengtsson, B., 2014. Vertical transmission of Escherichia coli carrying plasmid-mediated AmpC (pAmpC) through the broiler production pyramid. J Antimicrob Chemother. 69, 1497–500.
Olsen, R.H., Bisgaard, M., Löhren, U., Robineau, B., Christensen, H., 2014. Extended-spectrum β-lactamase-producing Escherichia coli isolated from poultry: a review of current problems, illustrated with some laboratory findings. Avian Pathol. 43, 199–208.
Pehlivanlar Önen, S., Aslantaş, Ö., Yılmaz, E.Ş., Kürekci, C., 2015. Prevalence of β-Lactamase Producing Escherichia coli from Retail Meat in Turkey. J Food Sci. 80, M2023–2029.
Pérez-Pérez, F.J., Hanson, N.D., 2002. Detection of plasmid-mediated AmpC beta-lactamase genes in clinical isolates by using multiplex PCR. J Clin Microbiol. 40, 2153–2162.
Philippon, A., Arlet, G., Jacoby, G.A., 2002. Plasmid-determined AmpC-type lactamases. Antimicrob Agents Chemother. 46, 1–11.
Picard, B., Garcia, J.S., Gouriou, S., Duriez, P., Brahimi, N., Bingen, E., Elion, J., Denamur, E. 1999. The link between phylogeny and virulence in Escherichia coli extraintestinal infection. Infect Immun. 67, 546–553.
Pires-dos-Santos, T., Bisgaard, M., Christensen, H. 2013. Genetic diversity and virulence profiles of Escherichia coli causing salpingitis and peritonitis in broiler breeders. Vet Microbiol. 162, 873–880.
Pitout, J.D. (2010): Infections with extended-spectrum beta-lactamase-producing enterobacteriaceae: Changing epidemiology and drug treatment choices. Drugs. 70, 313–33.
Pitout, J.D.D., Laupland, K.B. (2008): Extended-spectrum b-lactamase-producing Enterobacteriaceae. An emerging public-health concern. Lancet Infect Dis. 8, 159–166.
Quainoo, S., Coolen, J.P.M, van Hijum, S.A.F.T, Huynen, M.A., Melchers, W.J.G., van Schaik, W., Wertheim, H.F.L., 2017). Whole-genome sequencing of bacterial pathogens: the future of nosocomial outbreak analysis. Review. Clin Microbiol Rev. 30, 1015–1063.
Ranjbar, R., Pezeshknejad, P., Khamesipour, F., Amini, K., Kheiri, R., 2017. Genomic fingerprints of Escherichia coli strains isolated from surface water in Alborz province, Iran. BMC Res Notes. 10, 295.
Smet, A., Martel, A., Persoons, D., Dewulf, J., Heyndrickx, M., Catry, B., Herman, L., Haesebrouck, F., Butaye, P., 2008. Diversity of extended-spectrum beta-lactamases and class C beta-lactamases among cloacal Escherichia coli Isolates in Belgian broiler farms. Antimicrob Agents Chemother. 52, 1238–1243.
Smith, J.L., Drum, D.J., Dai, Y., Kim, J.M., Sanchez, S., Maurer, J.J., Hofacre, C.L., Lee, M.D., 2007. Impact of antimicrobial usage on antimicrobial resistance in commensal Escherichia coli strains colonizing broiler chickens. Appl Environ Microbiol. 73, 1404–1414.
Stephens, C.M., Skerker, J.M., Sekhon, M.S., Arkin, A.P., Riley, L.W., 2015. Complete genome sequences of four Escherichia coli ST95 isolates from bloodstream infections. Genome Announc. 3, e01241–15.
TMAF (Turkish Ministry of Agriculture and Forestry): Amendment pertaining Directive on production, importation, exportation, sale and use of feed additives and premixes Directive no:21.01.2006/26056. Available online: http://www.resmigazete.gov.tr/eskiler/2006/01/20060121.htm/ 28.08.2018
Toukdarian A (2004): Plasmid strategies for broad-host-range replication in Gram negative bacteria. In: Funnel B, Philips G, Eds. Plasmid Biology. Washington, DC: ASM Press,259–270.
Touzain, F., Le Devendec, L., de Boisséson, C., Baron, S., Jouy, E., Perrin-Guyomard, A., Blanchard, Y., Kempf, I., 2018. Characterization of plasmids harboring blaCTX-M and blaCMY genes in E. coli from French broilers. PLoS One. 13, e0188768.
Ünal N, Karagöz A, Aşkar Ş, Dilik Z, Yurteri Z (2017): Extended-spectrum β-lactamases among cloacal Escherichia coli isolates in healthy broilers in Turkey. Turk J Vet Anim Sci. 41: 72–76
Versalovic, J., Koeuth, T., Lupski, J.R., 1991. Distribution of repetitive DNA sequences in eubacteria and application to fingerprinting of bacterial genomes. Nucleic Acids Res. 19, 6823–31.
Villa, L., García-Fernández, A., Fortini, D., Carattoli, A., 2010. Replicon sequence typing of IncF plasmids carrying virulence and resistance determinants. J Antimicrob Chemother. 65, 2518–2529.
Vinueza-Burgos, C., Ortega-Paredes, D., Narváez, C., De Zutter, L., Zurita, J., 2019. Characterization of cefotaxime resistant Escherichia coli isolated from broiler farms in Ecuador. PLoS One. 14, e0207567.
Wang, G., Clark, C.G., Rodgers, F.G., 2002. Detection in Escherichia coli of the genes encoding the major virulence factors, the genes defining the O157:H7 serotype, and components of the type 2 Shiga toxin family by multiplex PCR. J Clin Microbiol. 40, 3613–3619.
WHO. 2015. World Health Organization: Worldwide country situation analysis: response to antimicrobial resistance. http://www.who.int/antimicrobial-resistance/publications/situationanalysis/en/. Accessed 05 September 2017.
Wirth, T., Falush, D., Lan, R., Colles, F., Mensa, P., Wieler, L.H., Karch, H., Reeves, P.R., Maiden, M.C., Ochman, H., Achtman, M., 2006. Sex and virulence in Escherichia coli: an evolutionary perspective. Mol Microbiol. 60, 1136–1151.
Zankari, E., Hasman, H., Kaas, R.S., Seyfarth, A.M., Agersø, Y., Lund, O., Larsen, M.V., Aarestrup, F.M., 2013. Genotyping using whole-genome sequencing is a realistic alternative to surveillance based on phenotypic antimicrobial susceptibility testing. J Antimicrob Chemother. 68, 771–777.
Funding
This work was supported by the Hatay Mustafa Kemal University Scientific Research Fund under Grant number of 18 M 014.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by Animal Ethic Committee of Hatay Mustafa Kemal University with decision number of 2017/4–5.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aslantaş, Ö. High occurrence of CMY-2-type beta-lactamase-producing Escherichia coli among broiler flocks in Turkey. Trop Anim Health Prod 52, 1681–1689 (2020). https://doi.org/10.1007/s11250-019-02167-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-019-02167-8