Abstract
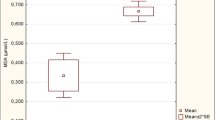
The present study aimed to assess hyperglycaemia with special reference to diabetes mellitus in cattle by clinico-biochemical estimation and evaluation of oxidative stress indices. A total of 256 cattle exhibiting weakness, poor body condition and reduced milk yield in lactating cattle were included in the study. These animals were screened with blood glucose level, urine glucose and ketone bodies. Out of these, 32 (12.5 %) cattle showed hyperglycaemia and glycosuria, of which 25 % exhibited ketonuria. Diabetes was confirmed in five cattle by estimation of fasting blood glucose, glycated haemoglobin, serum fructosamine, intravenous glucose tolerance test and insulin level. This reports first confirmation of diabetes in cattle in India. All these five animals revealed low level of serum insulin suggestive of insulin-dependent diabetes mellitus in cattle. The level of aspartate aminotransferase (AST) and gamma glutamyl transferase (GGT) was found to be increased in diabetic cattle. Oxidant/antioxidant balance was assessed in hyperglycaemic cattle and five age-matched Holstein Friesian (HF) cross-bred healthy control animals. Diabetic cattle revealed significantly higher (P ≤ 0.01) levels of erythrocytic lipid peroxides in comparison with other hyperglycaemic cattle and healthy controls whereas the level of superoxide dismutase (SOD) and catalase was found to be significantly lower in diabetes-affected animals in comparison to healthy controls. Reduced glutathione did not show a significant difference between hyperglycaemic and control groups. It is concluded from the present study that oxidative stress associated with diabetes in cattle is obvious compared with other hyperglycaemic cattle.
Similar content being viewed by others
References
Anderi, S., Matei, S., Zinveliu, D., Pintea, A., Bunea, A., Ciupe, S and Groza, I. 2010. Correlations between antioxidant enzyme activity and lipid peroxidation level in blood and milk from cows with subclinical mastitis. Vet. Med. 67 (1):6–10.
Benedict, S.R. 1908. A reagent for detection of reducing sugars. J. Biol. Chem. 5 (6):485–487.
Bergmeyer, H.U. 1983. UV method of catalase assay. In: Methods of Enzymatic Analysis, Vol 3rd, Weinheim. Deer Field Beach, Florida, pp: 273.
Clark, Z. 2003. Diabetes mellitus in a 6 month charolais heifer calf. Can. Vet. J. 44 (11):921–922.
Dennison, A.C., VanMetre, D. C., callan, R.J., Dinsmore, P., Mason, G.L and Ellis, R.P. 2002. Haemorrhagic bowel syndrome in dairy cattle: 22 cases (1997–2000). J. Am. Vet. Med. Assoc. 221(5):686–689.
Dimri, U., Sharma, M.C., Yamdagni, A., Ranjan, R and Zama, M.M.S. 2010. Psoroptic mange infestation increases oxidative stress and decreases antioxidant status in sheep. Vet. Parasitol. 168:318–322.
Fang, Y.Z., Yang, S and Wu, G. 2002. Free radicals, antioxidants and nutrition. Nutrition. 18: 872–878.
Fridlyand, L.E and Philipson, L.H.2010. Glucose sensing in the pancreatic β cell: a computational systems analysis. Theoretical Biology and Medical Modelling. 7:15.
Ghanem, M.M and Abdel-Hamid, O.M. 2010. Clinical, haematological and biochemical alterations in heat intolerance (panting) syndrome in Egyptian cattle following natural foot-and- mouth disease (FMD). Trop. Anim. Health Prod. 42 (6): 1167–1173.
Hasegawa, T., Uchda, K., Yanase, J., Kitazaki, K., Uchino, Y., Nakamura, S and Sakimoto, H. 1999. A case of Diabetes mellitus in Japanese black cattle. J. Vet. Med. Sci. 61:965–966.
Holtenius, K., Sternbauer, K and Holtenius, P. 2000. The effects of the plasma glucose level on the abomasal function in dairy cows. J. Anim. Sci. 78:1930–1935.
Hsu, W.H and Hummel, S.K. 1981. Xylazine induced hyperglycemia in cattle: A possible involvement of a2-Adrenergic Receptors regulating insulin release. Endocrinology. 109 (3):825–829.
Indran, M., Rokiah, P., Chan, S.P and Kuppusamy, U.R. 2004. Alteration of lipid peroxidation and antioxidant enzymes in young Malaysian IDDM patients. Med. J. Malaysia. 59: 162–170.
Jabeen, R and saleemuddin, M. 2006. Polyclonal antibodies inhibit the glycation induced inactivation of bovine CU, Zn-superoxide dismutase. Biotechnol. Appl. Biochem. 43: 49–53.
Jain, S.K., Movie, R., Duett, J and Herbst, J.J.1989. Erythrocyte membrane lipid peroxidation and glycosylated haemoglobin in diabetes. Diabetes. 38: 1539–1543.
Jun, H.S and Yoon, J.W.2001.The role of viruses in type 1 diabetes: two distinct cellular and molecular pathogenic mechanisms of virus induced diabetes in animals. Diabetaologia. 44:271–285.
Kahn, S.E. 2003. The relative contributions of insulin resistance and beta cell dysfunction to the pathophysiology of Type 2 diabetes. Diabetologia. 46: 3–19.
Kaneko, J.J., Harvey, J.W and Bruss, M.L. 2008. Carbohydrate metabolism and its diseases, Clinical Biochemistry of domestic animals, 6th edn., Elsevier Science. pp:49.
Kaplan, L.A. 1984. Carbohydrates and metabolites, In: Clinical Chemistry: Theory, Analysis and correlation. Kaplan, L.A and Pesce A.J. (Eds) C.V. Mosby, Toronto. pp :1032–1040.
Kuneko, I.J and Rhode, E.A. 1964. Diabetes mellitus in a cow. Journal of the American Veterinary Medical Association. 144 (4): 367–373.
Madesh, M and Balasubramanian, K.A.1998. Microtitre plate assay for superoxide dismutase using MTT reduction by superoxide. Indian J Biochem Biophy. 35:184–188.
Matsuda, K and Taniyama, H. 2013. Virus related diabetes in cattle. Diabetes and Viruses. pp: 87–97.
Nahed, S.T. 2010. Investigation of serum insulin and cortisol concentrations in Foot and Mouth disease – infected cattle in relation to changes in serum biochemical variables and protein electrophoretic Fractionation profile. Global veterinaria. 4 (5): 450–455.
Nazifi, S., Karimi, T and Ghasrodahti, A.R. 2004. Diabetes mellitus and fatty liver in a cow: case report. Comp. Clin. Path. 13:82–85.
Placer, Z.A., Cushman, L.L and Johnson, B.1966. Estimation of product of lipid peroxidation (malonaldehyde) in biochemical system. Anal Biochem. 16:359–364.
Prins, H.K and Loos, J.A. 1969. Biochemical Methods in Red Cell Genetics. Academic Press London. UK. pp: 530.
Sahu, A and Sarkar, P.D. 2008. Comparative study of NBT reduction method for estimation of glycated protein (serum fructosamine) with glycated HbA1c estimated on DCA 2000 analyzer. Ind. J. Physiol. Pharmacol. 52 (4):408–412.
Saunders, G.K., Blodgett, D.J., Hutchins, T.A., Prater, R.M.,Robertson, J.L., Friday, P.A and Scarratt, W.K. 2000. Suspected citrus pulp toxicosis in dairy cattle. J. Vet. Diagn. Invest. 12: 269–271.
Senthil Kumar, V., Safiullah, M.A., Kathiravan, G., Subramanian, M and Mani, K. 2013. Economic Analysis of metabolic diseases in bovines: A review. Int. J. Adv. Vet Sci. Tech.2 (1):64–71.
Sordillo, L.M and Aitken S.L. 2009. Impact of oxidative stress on the health and immune function of dairy cattle, Veterinary Immunol. Immunopathol. 128: 104–109.
Stanley,C.C. 2005. Regulation of glucose metabolism in dairy cattle. Ph.D thesis Submitted to the Graduate Faculty of the Louisiana State University and Agricultural and Mechanical College.pp: 1–4.
Tajima, M., Yuasa, M., Kawanabe, M.,Taniyama, H., Yamato, O and Maede, Y. 1999. Possible causes of diabetes mellitus in cattle infected with bovine viral diarrhoea virus. Zentralbl Veterinarmed B. 46 (3): 207–15
Tajima, M., Yazawa, T., Hagiwara, K., Kursawa, T and Takahashi, K. 2010. Diabetes mellitus in cattle infected with Bovine viral Diarrhea /Mucosal Disease Virus. J. Vet. Med. Series A. 39:616–620.
Takemoto, K., Tanaka, M., Iwata, H., Nishihara, R., Ishihara, K., wang, D.H., Ogino, K., Taniuchi, K and Masuoka, N. 2009. Low catalase activity in the blood is associated with the diabetes caused by alloxan. Clinica. Chimica. Acta. 407: 43–46.
Taniyama, H., Ushiki, T., Tajima, M., Kurosawa, T., Kitamura, N., Takahashi, K., Matukawa, K and Itakura, C. 1995. Spontaneous diabetes mellitus associated with persistent bovine viral diarrhea virus infection in young cattle. Vet. Pathol. 32:221–229.
Taniyama, H., Hirayama, K., Kagawa, Y., Kurosawa, T., Tajima, M., Yoshino, T and Furuoka, H., 1999. Histopathological and immunohistochemical analysis of the endocrine and exocrine pancreas in twelve cattle with insulin-dependent diabetes mellitus (IDDM). J. Vet. Med. Sci. 61:803–810.
Trivelli, L. A., Ranney, H.A and Lai, H.T. 1971. Haemoglobin components in patients with diabetes mellitus. N. Engl. J. Med. 284: 353–357.
Varashree, B.S and Bhat, G.P. 2011. Correlation of lipid peroxidation with glycated haemoglobin levels in diabetes mellitus. Online J. Health Allied Scs. 10:11.
Wilcox, G. 2005. Insulin and Insulin resistance. Clin. Biochem. Rev. 26: 19.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deepa, P.M., Dimri, U., Jhambh, R. et al. Alteration in clinico-biochemical profile and oxidative stress indices associated with hyperglycaemia with special reference to diabetes in cattle—a pilot study. Trop Anim Health Prod 47, 103–109 (2015). https://doi.org/10.1007/s11250-014-0691-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-014-0691-5