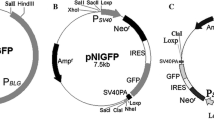
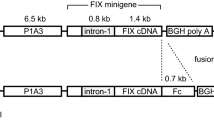
Abstract
There are growing numbers of recombinant proteins that have been expressed in milk. Thus one can consider the placement of any gene of interest under the control of the regulatory elements of a milk protein gene in a dairy farm animal. Among the transgene introducing techniques, only nuclear transfer (NT) allows 100 % efficiency and bypasses the mosaicism associated with counterpart techniques. In this study, in an attempt to produce a transgenic goat carrying the human coagulation factor IX (hFIX) transgene, goat fetal fibroblasts were electroporated with a linearized marker-free construct in which the transgene was juxtaposed to β-casein promoter designed to secret the recombinant protein in goat milk. Two different lines of transfected cells were used as donors for NT to enucleated oocytes. Two transgenic goats were liveborn. DNA sequencing of the corresponding transgene locus confirmed authenticity of the cloning procedure and the complementary experiments on the whey demonstrated expression of human factor IX in the milk of transgenic goats. In conclusion, our study has provided the groundwork for a prosperous and promising approach for large-scale production and therapeutic application of hFIX expressed in transgenic goats.








Similar content being viewed by others
References
Altschul SF, Madden TL, Schäffer AA, Zhang J, Miller W, Lipman DJ (1990) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acid Res 55:3389–3405
Arat S, Rzucidlo SJ, Gibbons J, Miyoshi K, Stice SL (2001) Production of transgenic bovine embryos by transfer of transfected granulosa cells into enucleated oocytes. Mol Reprod Dev 60:20–26
Arat S, Gibbons J, Rzucidlo SJ, Respess DS, Tumlin M, Stice SL (2002) In vitro development of bovine nuclear transfer embryos from transgenic clonal lines of adult and fetal fibroblast cells of the same genotype. Biol Reprod 66:1768–1774
Baguisi A, Behboodi E, Melican DT, Pollock JS, Destrempes MM, Cammuso C, Williams JL, Nims SD, Porter CA, Midura P, Palacios MJ, Ayres SL, Denniston RS, Hayes ML, Ziomek CA, Meade HM, Godke RA, Gavin WG, Overström EW, Echelard Y (1999) Production of goats by somatic cell nuclear transfer. Nat Biotechnol 17:456–461
Brownlee GG (1987) The molecular pathology of haemophilia B. Fourth Wellcome Trust lecture. Biochem Soc Trans 15:1–8
Campbell KH, McWhir J, Ritchie WA, Wilmut I (1996) Sheep cloned by nuclear transfer from a cultured cell line. Nature 380:64–66
Choo KH, Raphael K, McAdam W, Peterson MG (1987) Expression of active human blood clotting factor IX in transgenic mice: use of a cDNA with complete mRNA sequence. Nucleic Acids Res 15:871–884
Cibelli JB, Stice SL, Golueke PJ, Kane JJ, Jerry J, Blackwell C, Ponce de Leon FA, Robl JM (1998) Cloned transgenic calves produced from nonquiescent fetal fibroblasts. Science 281:1611
Coulibaly S, Besenfelder U, Fleischmann M, Zinovieva N, Grossmann A, Wozny M, Bartke I, Tögel M, Müller M, Brem G (1999) Human nerve growth factor beta (hNGF-beta): mammary gland specific expression and production in transgenic rabbits. FEBS Lett 444:111–116
Dalman A, Eftekhari-Yazdi P, Valojerdi MR, Shahverdi A, Gourabi H, Janzamin E, Fakheri R, Sadeghian F, Hasani F (2010) Synchronizing cell cycle of goat fibroblasts by serum starvation causes apoptosis. Reprod Domest Anim 45:e46–e53
Davie EW, Fujikawa K, Kisiel W (1991) The coagulation cascade: initiation, maintenance, and regulation. Biochemistry 30:10363–10370
Devinoy E, Thépot D, Stinnakre MG, Fontaine ML, Grabowski H, Puissant C, Pavirani A, Houdebine LM (1994) High level production of human growth hormone in the milk of transgenic mice: the upstream region of the rabbit whey acidic protein (WAP) gene targets transgene expression to the mammary gland. Transgenic Res 3:79–89
Gadbois DM, Crissman HA, Tobey RA, Bradbury EM (1992) Multiple kinase arrest points in the G1 phase of nontransformed mammalian cells are absent in transformed cells. Proc Natl Acad Sci USA 89:8626–8630
Houdebine LM (2000) Transgenic animal bioreactors. Transgenic Res 9:305–320
Keefer CL, Baldassarre H, Keyston R, Wang B, Bhatia B, Bilodeau AS, Zhou JF, Leduc M, Downey BR, Lazaris A, Karatzas CN (2001) Generation of dwarf goat (Capra hircus) clones following nuclear transfer with transfected and nontransfected fetal fibroblasts and in vitro-matured oocytes. Biol Reprod 64:849–856
Lagutina I, Lazzari G, Duchi R, Colleoni S, Ponderato N, Turini P, Crotti G, Galli C (2005) Somatic cell nuclear transfer in horses: effect of oocyte morphology, embryo reconstruction method and donor cell type. Reproduction 130:559–567
Maga EA, Murray JD (1995) Mammary gland expression of transgenes and the potential for altering the properties of milk. Biotechnology (NY) 13:1452–1457
Massoud M, Attal J, Thépot D, Pointu H, Stinnakre MG, Théron MC, Lopez C, Houdebine LM (1996) The deleterious effects of human erythropoietin gene driven by the rabbit whey acidic protein gene promoter in transgenic rabbits. Reprod Nutr Dev 36:555–563
Meade HM, Echelard Y, Ziomek CA et al (1998) Expression of recombinant proteins in the milk of transgenic animals. In: Fernandez JM, Hoeffler JP (eds) Gene expression systems: using nature for the art of expression. Academic Press, San Diego, pp 399–427
Montesino R, Toledo JR (2006) The mammary gland: bioreactor for the production of recombinant proteins. Biotecnol Apl S23:279–286
Onishi A, Iwamoto M, Akita T, Mikawa S, Takeda K, Awata T, Hanada H, Perry AC (2000) Pig cloning by microinjection of fetal fibroblast nuclei. Science 289:1188–1190
Park KW, Kuhholzer B, Lai L, Machaty Z, Sun QY, Day BN, Prather RS (2001) Development and expression of the green fluorescent protein in porcine embryos derived from nuclear transfer of transgenic granulosa-derived cells. Anim Reprod Sci 68:111–120
Platenburg GJ, Kootwijk EP, Kooiman PM, Woloshuk SL, Nuijens JH, Krimpenfort PJ, Pieper FR, de Boer HA, Strijker R (1994) Expression of human lactoferrin in milk of transgenic mice. Transgenic Res 3:99–108
Polejaeva IA, Campbell KH (2000) New advances in somatic cell nuclear transfer: applications in transgenesis. Theriogenology 53:117–126
Pollock DP, Kutzko JP, Birck-Wilson E, Williams JL, Echelard Y, Meade HM (1999) Transgenic milk as a method for the production of recombinant antibodies. J Immunol Methods 231:147–157
Prunkard D, Cottingham I, Garner I, Bruce S, Dalrymple M, Lasser G, Bishop P, Foster D (1996) High-level expression of recombinant human fibrinogen in the milk of transgenic mice. Nat Biotechnol 14:867–871
Reiner AP, Davie EW (1994) The physiology and biochemistry of factor IX. Haemost Thromb 3:309–332
Rosen JM, Li S, Raught B, Hadsell D (1996) The mammary gland as a bioreactor: factors regulating the efficient expression of milk protein-based transgenes. Am J Clin Nutr 63:627S–632S
Rudolph NS (1999) Biopharmaceutical production in transgenic livestock. Trends Biotech 17:367–374
Schnieke AE, Kind AJ, Ritchie WA, Mycock K, Scott AR, Ritchie M, Wilmut I, Colman A, Campbell KH (1997) Human factor IX transgenic sheep produced by transfer of nuclei from transfected fetal fibroblasts. Science 278:2130–2133
Sohn BH, Kim SJ, Park H, Park SK, Lee SC, Hong HJ, Park YS, Lee KK (1999) Expression and characterization of bioactive human thrombopoietin in the milk of transgenic mice. DNA Cell Biol 18:845–852
Stubbs MT, Bode W (1994) Coagulation factors and their inhibitors. Curr Opin Struct Biol 4:823–832
Wilmut I, Schnieke AE, McWhir J, Kind AJ, Campbell KHS (1997) Viable offspring derived from fetal and adult mammalian cells. Nature 385:810–813
Wright G, Carver A, Cottom D, Reeves D, Scott A, Simons P, Wilmut I, Garner I, Colman A (1991) High level expression of active human alpha-1-antitrypsin in the milk of transgenic sheep. Biotechnology (N Y) 9:830–834
Yu YS, Sun XS, Jiang HN, Han Y, Zhao CB, Tan JH (2003) Studies of the cell cycle of in vitro cultured skin fibroblasts in goats: work in progress. Theriogenology 59:1277–1289
Zinovieva N, Lassnig C, Schams D, Besenfelder U, Wolf E, Müller S, Frenyo L, Seregi J, Müller M, Brem G (1998) Stable production of human insulin-like growth factor 1 (IGF-1) in the milk of hemi- and homozygous transgenic rabbits over several generations. Transgenic Res 7:437–447
Acknowledgments
This work was supported by grant of Royan Institute for reproductive medicine, stem cell Biology and Biotechnology. The authors are thankful to laboratory Animal department of Royan institute, Dr. Seyed Mahdi Ghamsari and Dr. Taghi Taghipoor Bazargani for health care to transgenic goats and also Mr. Mehdi Totonchi for his technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Amir Amiri Yekta, Azam Dalman: Equally first authors.
Rights and permissions
About this article
Cite this article
Amiri Yekta, A., Dalman, A., Eftekhari-Yazdi, P. et al. Production of transgenic goats expressing human coagulation factor IX in the mammary glands after nuclear transfer using transfected fetal fibroblast cells. Transgenic Res 22, 131–142 (2013). https://doi.org/10.1007/s11248-012-9634-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-012-9634-y