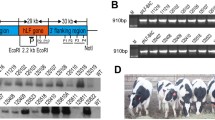
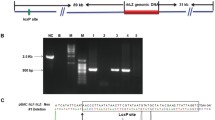
Abstract
Purified plasma derived human albumin has been available as a therapeutic product since World War II. However, cost effective recombinant production of albumin has been challenging due to the amount needed and the complex folding pattern of the protein. In an effort to provide an abundant source of recombinant albumin, a herd of transgenic cows expressing high levels of rhA in their milk was generated. Expression cassettes efficiently targeting the secretion of human albumin to the lactating mammary gland were obtained and tested in transgenic mice. A high expressing transgene was transfected in primary bovine cell lines to produce karyoplasts for use in a somatic cell nuclear transfer program. Founder transgenic cows were produced from four independent cell lines. Expression levels varying from 1–2 g/l to more than 40 g/l of correctly folded albumin were observed. The animals expressing the highest levels of rhA exhibited shortened lactation whereas cows yielding 1–2 g/l had normal milk production. This herd of transgenic cattle is an easily scalable and well characterized source of rhA for biomedical uses.








Similar content being viewed by others
References
Alexander MR, Alexander B, Mustion AL, Spector R, Wright CB (1982) Therapeutic use of albumin. JAMA 247:831–833. doi:10.1001/jama.247.6.831
Baguisi A, Behboodi E, Melican DT, Pollock JS, Detrempes MM, Cammuso C, Williams JL, Nims SD, Porter CA, Midura P, Palacios MJ, Ayers SL, Denniston RS, Hayes ML, Ziomek CA, Meade HM, Godke RA, Gavin WG, Overström EW, Echelard Y (1999) Production of goats by somatic cell nuclear transfer. Nat Biotechnol 17:456–461. doi:10.1038/8632
Behboodi E, Groen W, Destrempes MM, Williams JL, Ohlrichs C, Gavin WG, Broek DM, Ziomek CA, Faber DC, Meade HM, Echelard Y (2001) Transgenic production from in vivo-derived embryos: effect on calf birth weight and sex ratio. Mol Reprod Dev 60:27–37. doi:10.1002/mrd.1058
Brophy B, Smolenski G, Wheeler T, Wells D, L’Huillier P, Laible G (2003) Cloned transgenic cattle produce milk with higher levels of beta-casein and kappa-casein. Nat Biotechnol 21:157–162. doi:10.1038/nbt783
Chen RF (1967) Removal of fatty acids from serum albumin by charcoal treament. J Biol Chem 242:173–181
Chuang VT, Kragh-Hansen U, Otagiri M (2002) Pharmaceutical strategies utilizing recombinant human serum albumin. Pharm Res 19:569–577. doi:10.1023/A:1015396825274
Chung JH, Whiteley M, Felsenfeld G (1993) A 5′ element of the chicken beta-globin domain serves as an insulator in human erythroid cells and protects. Cell 74:505–514. doi:10.1016/0092-8674(93)80052-G
Church GM, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci USA 81:1991–1995. doi:10.1073/pnas.81.7.1991
Cibelli JB, Stice SL, Golueke PJ, Kane JJ, Jerry J, Blackwell C, Ponce de Leon A, Robl JM (1998) Cloned transgenic calves produced from nonquiescent fetal fibroblasts. Science 280:1256–1258. doi:10.1126/science.280.5367.1256
Clark AJ (1998) The mammary gland as a bioreactor: Expression, processing, and production of recombinant proteins. J Mammary Gland Biol 3:337–350. doi:10.1023/A:1018723712996
Cox H, Mead D, Sudbery P, Eland RM, Mannazzu I, Evans L (2000) Constitutive expression of recombinant proteins in the methylotrophic yeast Hansenula polymorpha using the PMA1 promoter. Yeast 30:1191–1203. doi:10.1002/1097-0061(20000930)16:13<1191::AID-YEA589>3.0.CO;2-2
Damiani P, Fissore RA, Cibelli JB, Long CR, Balise JJ, Robl JM, Duby RT (1996) Evaluation of developmental competence, nuclear and ooplasmic maturation of calf oocytes. Mol Reprod Dev 45:521–534. doi:10.1002/(SICI)1098-2795(199612)45:4<521::AID-MRD15>3.0.CO;2-Z
DiTullio P, Cheng SH, Marshall J, Gregory RJ, Ebert KM, Meade HM, Smith AE (1992) Production of cystic fibrosis transmembrane conductance regulator in the milk of transgenic mice. Biotechnology (NY) 10:74–77. doi:10.1038/nbt0192-74
Drouin R, Harvey P, Richer CL, Rémy-Prince S, Montplaisir S (1987) Human serum albumin as a substitute for foetal calf serum in blast transformation assays and cytogenetic analyses. Cytobios 53:193–207
Ebert KM, DiTullio P, Barry CA, Schindler JE, Ayres SL, Smith TE, Pellerin LJ, Meade HM, Denman J, Roberts B (1994) Induction of human tissue plasminogen activator in the mammary gland of transgenic goats. Biotechnology (NY) 12:699–702. doi:10.1038/nbt0794-699
Echelard Y, Destrempes MM, Koster JA, Blackwell C, Groen W, Pollock D, Williams JL, Behboodi E, Pommer J, Meade HM (2002) Production of recombinant human serum albumin in the milk of transgenic cows. Theriogenology 57:779
Echelard Y, Ziomek CA, Meade HM (2006) Production of recombinant therapeutic proteins in the milk of transgenic animals. BioPharm Int 19:36–46
Erstad BL, Gales BJ, Rappaport WD (1991) The use of albumin in clinical practice. Arch Intern Med 151:901–911. doi:10.1001/archinte.151.5.901
Farran I, Sanchez-Serrano JJ, Medina JF, Prieto J, Mingo-Castel AM (2002) Targeted expression of human serum albumin to potato tubers. Transgenic Res 11:337–346. doi:10.1023/A:1016356510770
Finlayson JS (1980) Albumin products. Semin Thromb Hemost 6:85–120
Fleer R, Yeh P, Amelllal N, Maury I, Fournier A, Bacchetta F, Baduel P, Jung G, L’Hote H, Becquart J, Fukuhara H, Mayaux JF (1991) Stable multicopy vectors for high-level secretion of recombinant human serum albumin by Kluyveromyces yeasts. Biotechnology (NY) 9:968–975. doi:10.1038/nbt1091-968
Gutierrez A, Meade H, Ditullio P, Pollock D, Harvey M, Jimenez-Flores R, Anderson G, Murray J, Medrano J (1996) Expression of a bovine kappa-CN cDNA in the mammary gland of transgenic mice utilizing a genomic milk protein gene as an expression cassette. Transgenic Res 5:271–279. doi:10.1007/BF01972881
Haselbeck A (2003) Epoetins: differences and their relevance to immunogenicity. Curr Med Res Opin 19:430–432
Hennessen W (1980) The clinical uses of albumin: report of an IABS study. Dev Biol Stand 48:49–52
Hewick RM, Hunkapiller MW, Hood LE, Dreyer WJ (1981) A gas-liquid solid phase peptide and protein sequenator. J Biol Chem 256:7990–7997
Hogan F, Constantini F, Lacy E (1986) Manipulating the mouse embryo: a laboratory manual. Cold Spring Harbor Lab. Press, Plainview
Holst N, Bertheussen K, Forsdahl F, Hakonsen MB, Hansen LJ, Nielsen HI (1990) Optimization and simplification of culture conditions in human in vitro fertilization (IVF) and preembryo replacement by serum-free media. J In Vitro Fertil Em 7:47–53. doi:10.1007/BF01133884
Houdebine LM (1994) The production of pharmaceutical proteins from the milk of transgenic animals. J Biotechnol 34:269–287. doi:10.1016/0168-1656(94)90062-0
Kobayashi K, Nakamura N, Sumi A, Ohmura T, Yokoyama K (1998) The development of recombinant human serum albumin. Ther Apher 2:257–262. doi:10.1111/j.1744-9987.1998.tb00118.x
Laird PW, Zijderveld A, Linders K, Rudnicki MA, Jaenisch R, Berns A (1991) Simplified mammalian DNA isolation procedure. Nucleic Acids Res 19:4293. doi:10.1093/nar/19.15.4293
Lanza RP, Cibelli JB, Blackwell C, Cristofalo VJ, Francis MK, Baerlocker GM, Mak J, Schertzer M, Chavez EA, Sawyer N, Lansdorp PM, West MD (2000) Extension of cell life-span and telomere length in animals cloned from senescent somatic cells. Science 288:665–669. doi:10.1126/science.288.5466.665
Lawn RM, Adelman J, Bock SC, Franke AE, Houck CM, Najarian RC, Seeburg PH, Wion KL (1981) The sequence of human serum albumin cDNA and its expression in E. coli. Nucleic Acids Res 25:6103–6114. doi:10.1093/nar/9.22.6103
Ledford H (2006) The farmyard drug store. Nature 443:16–17. doi:10.1038/443733a
Maga EA, Murray JD (1995) Mammary gland expression of transgenes and the potential for altering the properties of milk. Biotechnology (NY) 13:1452–1457. doi:10.1038/nbt1295-1452
Marth E, Kleinhappl B (2001) Albumin is a necessary stabilizer of TBE-vaccine to avoid fever in children after vaccination. Vaccine 20:532–537. doi:10.1016/S0264-410X(01)00329-2
Morimoto Y, Fujimoto S (1985) Albumin microspheres as drug carriers. Crit Rev Ther Drug 2:19–63
Niemann H, Kues WA (2007) Transgenic farm animals: an update. Reprod Fertil Dev 19:762–770. doi:10.1071/RD07040
Ohtani W, Nawa Y, Takeshima K, Kamuro H, Kobayashi K, Ohmura T (1998) Physicochemical and immunochemical properties of recombinant human serum albumin from Pichia pastoris. Anal Biochem 256:56–62. doi:10.1006/abio.1997.2480
Peters T Jr (ed) (1996) All about albumin: biochemistry genetics and medical applications. Academic Press, San Diego
Pollock DP, Kutzko JP, Birck-Wilson E, Williams JL, Echelard Y, Meade HM (1999) Transgenic milk as a method for the production of recombinant antibodies. J Immunol Methods 231:147–157. doi:10.1016/S0022-1759(99)00151-9
Quirk AV, Geisow MJ, Woodrow JR, Burton SJ, Wood PC, Sutton AD, Johnson RA, Dodsworth N (1989) Production of recombinant human serum albumin from Saccharomyces cerevisiae. Biotechnol Appl Biochem 11:273–287
Roberts B, DiTullio P, Vitale J, Hehir K, Gordon K (1992) Cloning of the goat b-casein-encoding gene and expression in transgenic mice. Gene 121:225–262
Robertson I, Nelson RE (1998) Certification and identification of the embryo. In: Stringfellow DA, Seidel SM (eds) Manual of the international embryo transfer society, vol 3. International Embryo Transfer Society, Savoy
Rudolph NS (1995) Regulatory issues relating to protein production in transgenic animal milk. Genet Eng News 15:16–18
Saunders CW, Schmidt BJ, Mallonee RL, Guyer MS (1987) Secretion of human serum albumin from Bacillus subtilis. J Bacteriol 169:2917–2925
Shaklai N, Garlick RL, Bunn HF (1984) Nonenzymatic glycosylation of human serum albuman alters its confirmation and function. J Biol Chem 259:3812–3817
Shani M, Barash I, Nathan M, Ricca G, Searfoss GH, Dekel I, Faerman A, Givol D, Hurwitz DR (1992) Expression of human serum albumin in the milk of transgenic mice. Transgenic Res 1:195–208. doi:10.1007/BF02524750
Sijmons PC, Dekker BM, Schrammeijer B, Verwoerd TC, van den Elzen PJ, Hoekema A (1990) Production of correctly processed human serum albumin in transgenic plants. Biotechnology (NY) 8:217–221. doi:10.1038/nbt0390-217
Soriano P, Montgomery C, Geske R, Bradley A (1991) Targeted disruption of the c-src proto-oncogene leads to osteopetrosis in mice. Cell 64:693–702. doi:10.1016/0092-8674(91)90499-O
Stadalnik RC, Kudo M, Eckelman WC, Vera DR (1993) In vivo functional imaging using receptor-binding radiopharmaceuticals; technetium 99 m-galactosyl-neoglycoalbumin as a model. Invest Radiol 28:64–70. doi:10.1097/00004424-199301000-00018
Tarelli E, Mire-Sluis A, Tivnann HA, Bolgiano B, Crane DT, Gee C, Lemercinier X, Athayde ML, Sutcliffe N, Corran PH, Rafferty B (1998) Recombinant human albumin as a stabilizer for biological materials and for the preparation of international reference reagents. Biologicals 26:331–346. doi:10.1006/biol.1998.0163
Tullis JL (1977) Albumin 1 background and use. JAMA 237:355–360. doi:10.1001/jama.237.4.355
van Berkel PHC, Welling MM, Geerts M, van Veen HA, Ravensbergen B, Salaheddine M, Pauwels EKJ, Pierper F, Nuijens JH, Nibbering PH (2002) Large scale production of recombinant human lactoferrin in the milk of transgenic cows. Nat Biotechnol 20:484–487. doi:10.1038/nbt0502-484
Wilkes MM, Navickis RJ (2001) Patient survival after human albumin administration a meta-analysis of randomized, controlled trials. Ann Intern Med 135:149–164
World Health Organization (1996) Bovine spongiform encephalopathy. Wkly Epidemiol Rec 71:113–116
World Health Organization guidelines on transmissible spongiform encephalopathies in relation to biological and pharmaceutical products (2003) World Health Organization, Blood safety and clinical technology department, Health technology and pharmaceuticals cluster Geneva, Switzerland
Zawada WM, Cibelli JB, Choi PK, Clarkson ED, Golueke PJ, Witta SE, Bell KP, Kane J, Ponce de Leon FA, Jerry DJ, Robl JM, Freed CR, Stice SL (1998) Somatic cell cloned transgenic bovine neurons for transplantation in parkinsonian rats. Nat Med 4:569–574. doi:10.1038/nm0598-569
Acknowledgments
We thank our colleagues Cathy Blackwell, Dr. Carol Ziomek, Gregory Liposky, Scott Fulton, Christopher Hendry, Debra Trainor, Daniel Couto, France Dumas, Dr. Eszter Birck-Wilson, Dr. Steven Stice, Dr. James Robl, Dr. Volker Lang, and Dr. Wolfram Eichner for their expert advice, support and encouragement at numerous stages of this project. This work was supported in part by Grant 1R44HL078300-01 from the NIH National Heart, Lung and Blood Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Echelard, Y., Williams, J.L., Destrempes, M.M. et al. Production of recombinant albumin by a herd of cloned transgenic cattle. Transgenic Res 18, 361–376 (2009). https://doi.org/10.1007/s11248-008-9229-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-008-9229-9