Abstract
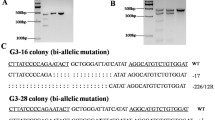
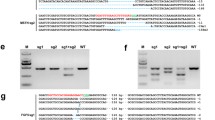
Homozygous mice devoid of functional Prnp are resistant to scrapie and prion propagation, but heterozygous mice for Prnp disruption still suffer from prion disease and prion deposition. We have previously generated heterozygous cloned goats with one allele of Prnp functional disruption. To obtain goats with both alleles of Prnp be disrupted which would be resistant to scrapie completely, a second-round gene targeting was applied to disrupt the wild type allele of Prnp in the heterozygous goats. By second-round gene targeting, we successfully disrupted the wild type allele of Prnp in primary Prnp +/− goat skin fibroblasts and obtained a Prnp −/− cell line without Prnp expression. This is the first report on successful targeting modification in primary adult somatic cells of animals. These cells were used as nuclear donors for somatic cell cloning to produce Prnp −/− goats. A total of 57 morulae or blastocytes developed from the reconstructed embryos were transferred to 31 recipients, which produced 7 pregnancies at day 35. At 73 days of gestation, we obtained one cloned fetus with Prnp −/− genotype. Our research not only indicated that multiple genetic modifications could be accomplished by multi-round gene targeting in primary somatic cells, but also provided strong evidence that gene targeting in adult cells other than fetal cells could be applied to introduce precise genetic modifications in animals without destroying the embryos.






Similar content being viewed by others
Abbreviations
- DMSO:
-
Dimethyl sulfoxide
- Dpl:
-
Doppel protein
- FBS:
-
Fetal bovine serum
- GMEM:
-
Glasgow minimal essential medium
- neo :
-
Neomycin phosphotransferase gene
- Prnp :
-
Prion protein gene
- PrP:
-
Prion protein
- puro :
-
Puromycin N-acetyl-transferase gene
References
Aguzzi A, Sigurdson C, Heikenwaelder M (2008) Molecular mechanisms of prion pathogenesis. Annu Rev Pathol 3:11–40. doi:10.1146/annurev.pathmechdis.3.121806.154326
Büeler H, Fischer M, Lang Y et al (1992) Normal development and behaviour of mice lacking the neuronal cell-surface PrP protein. Nature 356(6370):577–582. doi:10.1038/356577a0
Büeler H, Aguzzi A, Sailer A et al (1993) Mice devoid of PrP are resistant to scrapie. Cell 73(7):1339–1347. doi:10.1016/0092-8674(93)90360-3
Büeler H, Raeber A, Sailer A et al (1994) High prion and PrPSc levels but delayed onset of disease in scrapie-inoculated mice heterozygous for a disrupted PrP gene. Mol Med 1(1):19–30
Dai Y, Vaught TD, Boone J et al (2002) Targeted disruption of the a1,3-galactosyltransferase gene in cloned pigs. Nat Biotechnol 20(3):251–255. doi:10.1038/nbt0302-251
Denning C, Burl S, Ainslie A et al (2001) Deletion of the a(1,3)galactosyl transferase (GGTA1) gene and the prion protein (PrP) gene in sheep. Nat Biotechnol 19(6):559–562. doi:10.1038/89313
Kubota C, Yamakuchi H, Todoroki J et al (2000) Six cloned calves produced from adult fibroblast cells after long-term culture. Proc Natl Acad Sci USA 97(3):990–995. doi:10.1073/pnas.97.3.990
Kuroiwa Y, Kasinathan P, Matsushita H et al (2004) Sequential targeting of the genes encoding immunoglobulin-mu and prion protein in cattle. Nat Genet 36(7):775–780. doi:10.1038/ng1373
Lai L, Kolber-Simonds D, Park KW et al (2002) Production of a-1,3-galactosyltransferase knockout pigs by nuclear transfer cloning. Science 295(5557):1089–1092. doi:10.1126/science.1068228
Mallucci GR, Ratté S, Asante EA et al (2002) Post-natal knockout of prion protein alters hippocampal CA1 properties, but does not result in neurodegeneration. EMBO J 21(3):202–210. doi:10.1093/emboj/21.3.202
Manson JC, Clarke AR, McBride PA et al (1994) PrP gene dosage determines the timing but not the final intensity or distribution of lesions in scrapie pathology. Neurodegeneration 3(4):331–340
McCreath KJ, Howcroft J, Campbell KH et al (2000) Production of gene-targeted sheep by nuclear transfer from cultured somatic cells. Nature 405(6790):1066–1069. doi:10.1038/35016604
Meehan DT, Zink MA, Mahlen M et al (2008) Gene targeting in adult rhesus macaque fibroblasts. BMC Biotechnol 8:31. doi:10.1186/1472-6750-8-31
Moore RC, Lee IY, Silverman GL et al (1999) Ataxia in prion protein (PrP)-deficient mice is associated with upregulation of the novel PrP-like protein doppel. J Mol Biol 292(4):797–817. doi:10.1006/jmbi.1999.3108
Prusiner SB (1998) Prions. Proc Natl Acad Sci USA 95(23):13363–13383. doi:10.1073/pnas.95.23.13363
Richt JA, Kasinathan P, Hamir AN et al (2007) Production of cattle lacking prion protein. Nat Biotechnol 25(1):132–138. doi:10.1038/nbt1271
Rogers CS, Hao Y, Rokhlina T et al (2008) Production of CFTR-null and CFTR-DeltaF508 heterozygous pigs by adeno-associated virus-mediated gene targeting and somatic cell nuclear transfer. J Clin Invest 118(4):1571–1577. doi:10.1172/JCI34773
Rossi D, Cozzio A, Flechsig E et al (2001) Onset of ataxia and Purkinje cell loss in PrP null mice inversely correlated with Dpl level in brain. EMBO J 20(4):694–702. doi:10.1093/emboj/20.4.694
Sedivy JM, Vogelstein B, Liber HL et al (1999) Gene targeting in human cells without isogenic DNA. Science 283(1):9. doi:10.1126/science.283.5398.9a
Stewart LA, Rydzewska LH, Keogh GF et al (2008) Systematic review of therapeutic interventions in human prion disease. Neurology 70(15):1272–1281. doi:10.1212/01.wnl.0000308955.25760.c2
Tremblay P, Meiner Z, Galou M et al (1998) Doxycycline control of prion protein transgene expression modulates prion disease in mice. Proc Natl Acad Sci USA 95(21):12580–12585. doi:10.1073/pnas.95.21.12580
Wilmut I, Schnieke AE, McWhir J et al (1997) Viable offspring derived from fetal and adult mammalian cells. Nature 385(6619):810–813. doi:10.1038/385810a0
Yu G, Chen J, Yu H et al (2006) Functional disruption of the prion protein gene in cloned goats. J Gen Virol 87(Pt4):1019–1027. doi:10.1099/vir.0.81384-0
Zou X, Wang Y, Cheng Y et al (2002) Generation of cloned goats (Capra hircus) from transfected foetal fibroblast cells, the effect of donor cell cycle. Mol Reprod Dev 61(2):164–172. doi:10.1002/mrd.1143
Acknowledgments
We gratefully thank Hongying sha, Laixiang Ge, Dawei Hu, Fazhan Xie and Lijun Ding for their technical assistance to this work. This work was supported by grants from Shanghai Rising-Star Program (07QB14022) and China Postdoctoral Science Foundation (20060400174).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, C., Li, B., Yu, G. et al. Production of Prnp −/− goats by gene targeting in adult fibroblasts. Transgenic Res 18, 163–171 (2009). https://doi.org/10.1007/s11248-008-9220-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-008-9220-5