Abstract
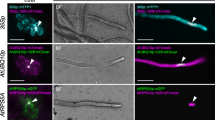
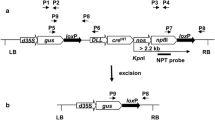
A genomic DNA fragment from wheat carrying the Glu-1Dx5 gene has been shown to exhibit reduced pollen transmission in transgenic maize. To localize the region of the DNA fragment responsible for this reduced pollen transmission, we produced transgenic maize plants in which the wheat genomic DNA proximal to the 1Dx5 coding sequence was replaced with the maize 27 kDa γ-zein promoter. Like the wheat promoter-driven Glu-1Dx5 transgene, this zein promoter-driven transgene functioned to produce 1Dx5 in maize endosperm. However, with the zein promoter-driven transgene, pollen transmission of the transgene loci was normal in most self- and cross-pollinations. We concluded that the wheat genomic DNA proximal to the wheat 1Dx5 coding sequence was required for reduced pollen transmission of the transgene in maize. In two of four transformation events of the wheat promoter-driven construct examined, pollen exhibited two morphological classes. In one class, pollen was normal in morphology and displayed average viability, and in the second, pollen was reduced in size and did not germinate on artificial media. DNA from the transgene was detectable in mature pollen from plants with reduced pollen transmission of transgene loci. To explain these observations, we hypothesize that elements within the transgene construct interfere with pollen development. We demonstrated that the wheat genomic DNA fragment can be used to control pollen transmission of an herbicide resistance transgene genetically linked to it. The wheat genomic DNA fragment may contain elements that are useful for controlling pollen transmission of transgene loci in commercial maize grain and seed production.






Similar content being viewed by others
References
Anderson OD, Greene FC, Yip RE, Halford NG, Shewry PR, Malpica-Romero JM (1989) Nucleotide sequences of the two high-molecular-weight glutenin genes from the D-genome of a hexaploid bread wheat, Triticum aestivum L. cv Cheyenne. Nucleic Acids Res 17:461–462
Aylor DE, Schultes NP, Shields EJ (2003) An aerobiological framework for assessing cross-pollination in maize. Agr Forest Meteorol 119:111–129
Brettschneider R, Becker D, Lorz H (1997) Efficient transformation of scutellar tissue of immature maize embryos. Theor Appl Genet 94:737–748
Burnham CR (1950) Chromosome segregation in translocations involving chromosome 6 in maize. Genetics 35:446–481
Chang MT, Neuffer GM (1989) Maize microsporogenesis. Genome 32:232–244
Daniell H (2002) Molecular strategies for gene containment in transgenic crops. Nat Biotechnol 20:581–586
Evans MMS, Kermicle JL (2001) Teosinte crossing barrier1, a locus governing hybridization of teosinte with maize. Theor Appl Genet 103:259–265
Frame BR, Zhang H, Cocciolone SM, Sidorenko LV, Dietrich CR, Pegg SE, Zhen S, Schnable PS, Wang K (2000) Production of transgenic maize from bombarded Type II callus: effect of gold particle size and callus morphology on transformation efficiency. In Vitro Cell Dev-Pl 36:21–29
Halsey ME, Remund KM, Davis CA, Qualls M, Eppard PJ, Berberich SA (2005) Isolation of maize from pollen-mediated gene flow by time and distance. Crop Sci 45:2172–2185
Harlow E, Lane D (1988) Antibodies: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Ma BL, Subedi KD, Reid LM (2004) Extent of cross-fertilization in maize by pollen from neighboring transgenic hybrids. Crop Sci 44:1273–1282
Maliga P (2004) Plastid transformation in higher plants. Annu Rev Plant Biol 55:289–313
Mariani C, De Beuckeleer M, Truettner J, Leemans J, Goldberg RB (1990) Induction of male sterility in plants by a chimaeric ribonuclease gene. Nature 347:737–741
McCormick S (2004) Control of male gametophyte development. Plant Cell 16:S142–S153
Muschietti J, Dircks L, Vancanneyt G, McCormick S (1994) LAT52 protein is essential for tomato pollen development: pollen expressing antisense LAT52 RNA hydrates and germinates abnormally and cannot achieve fertilization. Plant J 6:321–338
Pline WA, Viator R, Wilcut JW, Edmisten KL, Thomas J, Wells R (2002) Reproductive abnormalities in glyphosate-resistant cotton caused by lower CP4-EPSPS levels in the male reproductive tissue. Weed Sci 50:438–447
Saghai-Maroof MA, Soliman KM, Jorgensenn RA, Allard RW (1984) Ribosomal DNA spacer-length polymorphisms in barley: Mendelian inheritance, chromosomal location and population dynamics. Proc Natl Acad Sci USA 81:8014–8019
Sangtong V, Moran DL, Chikwamba R, Wang K, Woodman-Clikeman W, Long MJ, Lee M, Scott MP (2002) Expression and inheritance of the wheat Glu-1Dx5 gene in transgenic maize. Theor Appl Genet 105:937–945
Sari-Gorla M, Gatti E, Villa M, Pè ME (1997) A multi-nucleate male-sterile mutant of maize with gametophytic expression. Sex Plant Reprod 10:22–26
Schreiber DN, Dresselhaus T (2003) In vitro pollen germination and transient transformation of Zea mays and other plant species. Plant Mol Biol Reporter 21:31–41
Stadler LJ, Roman H (1948) The effect of x-rays upon mutation of the gene A in maize. Genetics 33:273–303
Stevens WE, Berberich SA, Sheckell PA, Wiltse CC, Halsey ME, Horak MJ, Dunn DJ (2004) Optimizing pollen confinement in maize grown for regulated products. Crop Sci 44:2146–2153
Vickers JE, Graham GC, Henry RJ (1996) A protocol for the efficient screening of putatively transformed plants for bar, the selectable marker gene, using the polymerase chain reaction. Plant Mol Biol Reporter 14:363–368
Yang SH, Moran DL, Jia HW, Bicar EH, Lee M, Scott MP (2002) Expression of a synthetic porcine alpha-lactalbumin gene in the kernels of transgenic maize. Transgenic Res 11:11–20
Acknowledgements
The authors wish to thank Merinda Struthers, Erik Mottl, and staff at the Bessey Microscopy Facility as well as the Pat Schnable laboratory for technical assistance. Names are necessary to report factually on the available data; however, the USDA neither guarantees nor warrants the standard of the product, and the use of the name by the USDA implies no approval of the product to the exclusion of others that may be suitable.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scott, M.P., Peterson, J.M., Moran, D.L. et al. A wheat genomic DNA fragment reduces pollen transmission of maize transgenes by reducing pollen viability. Transgenic Res 16, 629–643 (2007). https://doi.org/10.1007/s11248-006-9055-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-006-9055-x