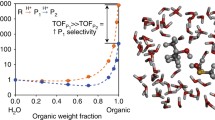
Abstract
Mixtures of water and organic cosolvents (mixed solvent systems) play an important role in mediating acid-catalyzed biomass conversion reactions. A minimum amount of water is typically required to dissolve biomass-derived materials, while adding an organic cosolvent can enhance the rates and selectivities of the desirable, catalytic reaction steps. Understanding the molecular-level bases underlying these solvent effects would provide a powerful measure of control over the reaction environment for biomass conversion processes, whereby the rates of desired reaction steps could be preferentially enhanced over the undesirable ones by modulating the composition of the solvent system. However, a quantitative basis to anticipate these solvent effects is currently lacking, and optimizing the composition of the liquid phase for new biomass conversion reactions typically requires laborious screening of the continuous space of possible mixed solvent systems. Herein, we summarize our efforts to estimate solvent effects on the rates and selectivities of liquid-phase, acid-catalyzed biomass conversions reactions using experiments, classical molecular dynamics simulations, and machine learning tools. We then synthesize these insights into a workflow that allows for the rational design of mixed solvent systems for acid-catalyzed biomass conversion processes using computationally efficient methods and minimal experiments. We demonstrate this design framework by analyzing two case studies: the acid-catalyzed dehydration of cyclohexanol to cyclohexene, and the partial dehydration of fructose to 5-hydroxymethylfurfural.
Similar content being viewed by others
References
Dumesic J, Topsøe H, Khammouma S, Boudart M (1975) Surface, catalytic and magnetic properties of small iron particles: II. Structure sensitivity of ammonia synthesis. J Catal 37:503–512
Ledesma C, Yang J, Chen D, Holmen A (2014) Recent approaches in mechanistic and kinetic studies of catalytic reactions using SSITKA technique. ACS Catal 4:4527–4547
Fan L, Ziegler T (1992) Nonlocal density functional theory as a practical tool in calculations on transition states and activation energies. Applications to elementary reaction steps in organic chemistry, J Am Chem Soc 114:10890–10897
Gokhale AA, Kandoi S, Greeley JP, Mavrikakis M, Dumesic JA (2004) Molecular-level descriptions of surface chemistry in kinetic models using density functional theory. Chem Eng Sci 59:4679–4691
Nørskov JK, Bligaard T, Rossmeisl J, Christensen CH (2009) Towards the computational design of solid catalysts. Nat Chem 1:37
Zhang L, Roling LT, Wang X, Vara M, Chi M, Liu J, Choi S-I, Park J, Herron JA, Xie Z (2015) Platinum-based nanocages with subnanometer-thick walls and well-defined, controllable facets. Science 349:412–416
Nørskov JK, Bligaard T, Logadottir A, Bahn S, Hansen LB, Bollinger M, Bengaard H, Hammer B, Sljivancanin Z, Mavrikakis M (2002) Universality in heterogeneous catalysis. J Catal 209:275–278
Centi G, Perathoner S (2009) Catalysis: role and challenges for a sustainable energy. Top Catal 52:948–961
Chheda JN, Huber GW, Dumesic JA (2007) Liquid-phase catalytic processing of biomass-derived oxygenated hydrocarbons to fuels and chemicals. Angew Chem Int Ed 46:7164–7183
Shuai L, Luterbacher J (2016) Organic solvent effects in biomass conversion reactions. Chemsuschem 9:133–155
Walker TW, Motagamwala AH, Dumesic JA, Huber GW (2018) Fundamental catalytic challenges to design improved biomass conversion technologies. J Catal 369:521–526
Serrano-Ruiz JC, Dumesic JA (2011) Catalytic routes for the conversion of biomass into liquid hydrocarbon transportation fuels. Energy Environ Sci 4:83–99
Mellmer MA, Sener C, Gallo JMR, Luterbacher JS, Alonso DM, Dumesic JA (2014) Solvent effects in acid-catalyzed biomass conversion reactions. Angew Chem Int Ed 53:11872–11875
Maugh TH (1983) Catalysis: no longer a black art. Science 219:474–477
Román-Leshkov Y, Chheda JN, Dumesic JA (2006) Phase modifiers promote efficient production of hydroxymethylfurfural from fructose. Science 312:1933–1937
Luterbacher JS, Rand JM, Alonso DM, Han J, Youngquist JT, Maravelias CT, Pfleger BF, Dumesic JA (2014) Nonenzymatic sugar production from biomass using biomass-derived γ-valerolactone. Science 343:277–280
Motagamwala AH, Won W, Maravelias CT, Dumesic JA (2016) An engineered solvent system for sugar production from lignocellulosic biomass using biomass derived γ-valerolactone. Green Chem 18:5756–5763
Shuai L, Questell-Santiago YM, Luterbacher JS (2016) A mild biomass pretreatment using γ-valerolactone for concentrated sugar production. Green Chem 18:937–943
Mushrif SH, Caratzoulas S, Vlachos DG (2012) Understanding solvent effects in the selective conversion of fructose to 5-hydroxymethyl-furfural: a molecular dynamics investigation. Phys Chem Chem Phys 14:2637–2644
Christianson JR, Caratzoulas S, Vlachos DG (2015) Computational insight into the effect of Sn-beta Na exchange and solvent on glucose isomerization and epimerization. ACS Catal 5:5256–5263
Assary RS, Redfern PC, Hammond JR, Greeley J, Curtiss LA (2010) Computational studies of the thermochemistry for conversion of glucose to levulinic acid. J Phys Chem B 114:9002–9009
Mellmer MA, Sanpitakseree C, Demir B, Bai P, Ma K, Neurock M, Dumesic JA (2018) Solvent-enabled control of reactivity for liquid-phase reactions of biomass-derived compounds. Nat Catal 1:199
Walker TW, Chew AK, Li H, Demir B, Zhang ZC, Huber GW, Van Lehn RC, Dumesic JA (2018) Universal kinetic solvent effects in acid-catalyzed reactions of biomass-derived oxygenates. Energy Environ Sci 11:617–628
Chew AK, Van Lehn RC (2019) Quantifying the stability of the hydronium ion in organic solvents with molecular dynamics simulations. Front Chem 7:439
Chew AK, Jiang S, Zhang W, Zavala VM, Van Lehn RC (2019) Fast predictions of liquid-phase acid-catalyzed reaction rates using molecular dynamics simulations and convolutional neural networks. ChemRxiv. https://doi.org/10.26434/chemrxiv.11299121.v1
Chew AK, Walker TW, Shen Z, Demir B, Witteman L, Euclide J, Huber GW, Dumesic JA, Van Lehn RC (2019) Effect of mixed-solvent environments on the selectivity of acid-catalyzed dehydration reactions. Acs Catal 10:1679–1691
Páll S, Abraham MJ, Kutzner C, Hess B, Lindahl E (2015) Tackling exascale software challenges in molecular dynamics simulations with GROMACS. In: Markidis S, Laure E (eds) Solving software challenges for exascale: international conference on exascale applications and software, EASC 2014, Stockholm, Sweden, April 2–3, 2014. Springer, Cham, pp 3–27
Best RB, Zhu X, Shim J, Lopes PEM, Mittal J, Feig M, MacKernell AD Jr (2013) Optimization of the additive CHARMM Allatom protein force field targeting improved sampling from the backbone and side chain and dihedral angles. J Chem Theory Comput 8:3257–3273
Vanommeslaeghe K, Hatcher E, Acharya C, Kundu S, Zhong S, Shim J, Darian E, Guvench O, Lopes P, Vorobyov I, MacKernell AD Jr (2009) CHARMM general force field: a force field for drug-like molecules compatible with the CHARMM all-atom additive biological force fields. J Comput Chem 31:671–690
Yu W, He X, Vanommeslaeghe K, MacKernell AD Jr (2012) Extension of the CHARMM general force field to sulfonyl- containing compounds and its utility in biomolecular simulations. J Comput Chem 33:2451–2468
Berendsen HJC, Grigera JR, Straatsma TP (1987) The missing term in effective pair potentials. J Phys Chem 91:6269–6271
Shivakumar D, Williams J, Wu YJ, Damm W, Shelley J, Sherman W (2010) Prediction of absolute solvation free energies using molecular dynamics free energy perturbation and the OPLS force field. J Chem Theory Comput 6:1509–1519
Shirts MR, Chodera JD (2008) Statistically optimal analysis of samples from multiple equilibrium states. J Chem Phys 129:124105
Klimovich PV, Shirts MR, Mobley DL (2015) Guidelines for the analysis of free energy calculations. J Comput Aided Mol Des 29:397–411
Daoutidis P, Marvin WA, Rangarajan S, Torres AI (2013) Engineering biomass conversion processes: a systems perspective. AIChE J 59:3–18
Madon RJ, Iglesia E (2000) Catalytic reaction rates in thermodynamically non-ideal systems. J Mol Catal A 163:189–204
Krishna SH, Walker TW, Dumesic JA, Huber GW (2017) Kinetics of levoglucosenone isomerization. Chemsuschem 10:129–138
Lindsay MJ, Walker TW, Dumesic JA, Rankin SA, Huber GW (2018) Production of monosaccharides and whey protein from acid whey waste streams in the dairy industry. Green Chem 20:1824–1834
Russakovsky O, Deng J, Su H, Krause J, Satheesh S, Ma S, Huang Z, Karpathy A, Khosla A, Bernstein M (2015) Imagenet large scale visual recognition challenge. Int J Comput Vision 115:211–252
Liu Y, Vjunov A, Shi H, Eckstein S, Camaioni DM, Mei D, Baráth E, Lercher JA (2017) Enhancing the catalytic activity of hydronium ions through constrained environments. Nat Commun 8:14113
Mei D, Lercher JA (2019) Effects of Local water concentrations on cyclohexanol dehydration in H-BEA zeolite. J Phys Chem C 123:25255–25266
He J, Zhao C, Lercher JA (2014) Impact of solvent for individual steps of phenol hydrodeoxygenation with Pd/C and HZSM-5 as catalysts. J Catal 309:362–375
Wang X, Rinaldi R (2013) A route for lignin and bio-oil conversion: dehydroxylation of phenols into arenes by catalytic tandem reactions. Angew Chem Int Ed 52:11499–11503
Smith MD, Mostofian B, Petridis L, Cheng XL, Smith JC (2016) Molecular driving forces behind the tetrahydrofuran-water miscibility gap. J Phys Chem B 120:740–747
Van Putten R-J, Van Der Waal JC, De Jong E, Rasrendra CB, Heeres HJ, de Vries JG (2013) Hydroxymethylfurfural, a versatile platform chemical made from renewable resources. Chem Rev 113:1499–1597
Patil SK, Lund CR (2011) Formation and growth of humins via aldol addition and condensation during acid-catalyzed conversion of 5-hydroxymethylfurfural. Energy Fuels 25:4745–4755
Asghari FS, Yoshida H (2007) Kinetics of the decomposition of fructose catalyzed by hydrochloric acid in subcritical water: formation of 5-hydroxymethylfurfural, levulinic, and formic acids. Ind Eng Chem Res 46:7703–7710
Teong SP, Yi G, Zhang Y (2014) Hydroxymethylfurfural production from bioresources: past, present and future. Green Chem 16:2015–2026
Motagamwala AH, Huang K, Maravelias CT, Dumesic JA (2019) Solvent system for effective near-term production of hydroxymethylfurfural (HMF) with potential for long-term process improvement. Energy Environ Sci 12:2212–2222
Qi X, Watanabe M, Aida TM, Smith RL Jr (2008) Selective conversion of D-fructose to 5-hydroxymethylfurfural by ion-exchange resin in acetone/dimethyl sulfoxide solvent mixtures. Ind Eng Chem Res 47:9234–9239
Tsilomelekis G, Josephson TR, Nikolakis V, Caratzoulas S (2014) Origin of 5-hydroxymethylfurfural stability in water/dimethyl sulfoxide mixtures. Chemsuschem 7:117–126
Weingarten R, Cho J, Xing R, Conner WC Jr, Huber GW (2012) Kinetics and reaction engineering of levulinic acid production from aqueous glucose solutions. Chemsuschem 5:1280–1290
Cai CM, Zhang T, Kumar R, Wyman CE (2013) THF co-solvent enhances hydrocarbon fuel precursor yields from lignocellulosic biomass. Green Chem 15:3140–3145
Smith MD, Mostofian B, Cheng X, Petridis L, Cai CM, Wyman CE, Smith JC (2016) Cosolvent pretreatment in cellulosic biofuel production: effect of tetrahydrofuran-water on lignin structure and dynamics. Green Chem 18:1268–1277
Mushrif SH, Varghese JJ, Krishnamurthy CB (2015) Solvation dynamics and energetics of intramolecular hydride transfer reactions in biomass conversion. Phys Chem Chem Phys 17:4961–4969
Nikbin N, Caratzoulas S, Vlachos DG (2012) A first principles-based microkinetic model for the conversion of fructose to 5-hydroxymethylfurfural. Chemcatchem 4:504–511
Varghese JJ, Mushrif SH (2019) Origins of complex solvent effects on chemical reactivity and computational tools to investigate them: a review. React Chem Eng 4:165–206
Vasudevan V, Mushrif SH (2015) Insights into the solvation of glucose in water, dimethyl sulfoxide (DMSO), tetrahydrofuran (THF) and N, N-dimethylformamide (DMF) and its possible implications on the conversion of glucose to platform chemicals. Rsc Adv 5:20756–20763
Hazra MK, Bagchi B (2018) Non-equilibrium solvation dynamics in water-DMSO binary mixture: composition dependence of non-linear relaxation. J Chem Phys 149:084501
Acknowledgements
This work was supported in part by the Great Lakes Bioenergy Research Center, U.S. Department of Energy, Office of Science, Office of Biological and Environmental Research under Award Numbers DE-SC0018409. This work used the Extreme Science and Engineering Discovery Environment (XSEDE), which is supported by National Science Foundation grant number ACI-1549562. This work also used the computing resources and assistance of the UW-Madison Center for High Throughput Computing (CHTC) in the Department of Computer Sciences. The CHTC is supported by UW-Madison, the Advanced Computing Initiative, the Wisconsin Alumni Research Foundation, the Wisconsin Institutes for Discovery, and the National Science Foundation, and is an active member of the Open Science Grid, which is supported by the National Science Foundation and the U.S. Department of Energy's Office of Science. The authors acknowledge support from the Department of Chemical and Biological Engineering at the University of Wisconsin-Madison and the Wisconsin Alumni Research Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest:
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Walker, T.W., Chew, A.K., Van Lehn, R.C. et al. Rational Design of Mixed Solvent Systems for Acid-Catalyzed Biomass Conversion Processes Using a Combined Experimental, Molecular Dynamics and Machine Learning Approach. Top Catal 63, 649–663 (2020). https://doi.org/10.1007/s11244-020-01260-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-020-01260-9