Abstract
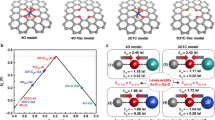
Formic acid and methanol are considered as liquid organic hydrogen carriers and could be produced sustainably from biomass or by CO2 hydrogenation using catalysts. The choice of a catalyst for decomposition of these substances is a challenge. We prepared N-free and N-doped mesoporous carbon materials containing less than 2 wt% of gold not stabilized by chlorine anions. High-angle annular dark field scanning transmission electron microscopy (STEM) data showed that the supported gold was present in the state of single Au atoms and Au nanoparticles after reduction. The use of the N-doped carbon support allowed reaching high dispersion of the Au nanoparticles with a mean size of ~ 2 nm, as compared to ~ 10 nm for the N-free support. Density functional theory studies indicated that a single Au atom may attach strongly (> 3.6 eV) to carbon atoms on the graphene edge or even more strongly to carbon atoms in a double vacancy; however, it may attach only weakly to N species of the N-doped carbon. Formic acid and methanol molecules interact relatively strongly (~ 0.75 eV and ~ 1.0 eV, respectively) via their oxygen atoms with the Au atom on the graphene edge, while they interact only weakly with the graphene fragments containing the Au atoms in double vacancies (~ 0.4 eV and ~ 0.3 eV, respectively). This demonstrates importance of coordinative unsaturation of Au atoms for adsorption and may influence further reactivity of the adsorbed species. The obtained results can be used for development of catalysts and electrocatalysts containing single Au atoms for hydrogen production from liquid organic hydrogen carriers.







Similar content being viewed by others
References
Li H, Zhang H-X, Yan X-L, Xu B-S, Guo J-J (2018) Carbon-supported metal single atom catalysts. New Carbon Mater 33(1):1–11. https://doi.org/10.1016/S1872-5805(18)60322-1
Mitchell S, Vorobyeva E, Pérez-Ramírez J (2018) Reactivity of single-atom heterogeneous catalysts: unique and multifaceted. Angew Chem Int Ed 57(47):15316–15329. https://doi.org/10.1002/anie.201806936
Wang A, Li J, Zhang T (2018) Heterogeneous single-atom catalysis. Nat Rev Chem 2(6):65–81. https://doi.org/10.1038/s41570-018-0010-1
Wang C, Yang M, Flytzani-Stephanopoulos M (2016) Single gold atoms stabilized on nanoscale metal oxide supports are catalytic active centers for various reactions. AIChE J 62(2):429–439. https://doi.org/10.1002/aic.15134
Rivera-Cárcamo C, Serp P (2018) Single atom catalysts on carbon-based materials. ChemCatChem 10:5059–5091. https://doi.org/10.1002/cctc.201801174
Bayatsarmadi B, Zheng Y, Vasileff A, Qiao S-Z (2017) Recent advances in atomic metal doping of carbon-based nanomaterials for energy conversion. Small 13(21):1700191. https://doi.org/10.1002/smll.201700191
Qiao B, Liang J-X, Wang A, Liu J, Zhang T (2016) Single atom gold catalysts for low-temperature CO oxidation. Chin J Catal 37(10):1580–1586. https://doi.org/10.1016/S1872-2067(16)62529-9
Wang Z, Gu L, Song L, Wang H, Yu R (2018) Facile one-pot synthesis of MOF supported gold pseudo-single-atom catalysts for hydrogenation reactions. Mater Chem Front 2(5):1024–1030. https://doi.org/10.1039/C8QM00081F
Malta G, Kondrat SA, Freakley SJ, Davies CJ, Lu L, Dawson S, Thetford A, Gibson EK, Morgan DJ, Jones W, Wells PP, Johnston P, Catlow CRA, Kiely CJ, Hutchings GJ (2017) Identification of single-site gold catalysis in acetylene hydrochlorination. Science 355(6332):1399–1403. https://doi.org/10.1126/science.aal3439
Liu X, Conte M, Elias D, Lu L, Morgan DJ, Freakley SJ, Johnston P, Kiely CJ, Hutchings GJ (2016) Investigation of the active species in the carbon-supported gold catalyst for acetylene hydrochlorination. Catal Sci Technol 6(13):5144–5153. https://doi.org/10.1039/C6CY00090H
Hardcastle TP, Seabourne CR, Zan R, Brydson RMD, Bangert U, Ramasse QM, Novoselov KS, Scott AJ (2013) Mobile metal adatoms on single layer, bilayer, and trilayer graphene: An ab invitio DFT study with van der Waals corrections correlated with electron microscopy data. Phys Rev B. https://doi.org/10.1103/PhysRevB.87.195430
Zan R, Bangert U, Ramasse Q, Novoselov KS (2011) Metal-graphene interaction studied via atomic resolution scanning transmission electron microscopy. Nano Lett 11(3):1087–1092. https://doi.org/10.1021/nl103980h
Wang H, Li K, Cheng Y, Wang Q, Yao Y, Schwingenschlögl U, Zhang X, Yang W (2012) Interaction between single gold atom and the graphene edge: a study via aberration-corrected transmission electron microscopy. Nanoscale 4(9):2920–2925. https://doi.org/10.1039/C2NR00059H
Krasheninnikov AV, Lehtinen PO, Foster AS, Pyykko P, Nieminen RM (2009) Embedding Transition-Metal Atoms in Graphene: Structure, Bonding, and Magnetism. Phys Rev Lett. https://doi.org/10.1103/PhysRevLett.102.126807
Kunaseth M, Poldorn P, Junkeaw A, Meeprasert J, Rungnim C, Namuangruk S, Kungwan N, Inntam C, Jungsuttiwong S (2017) A DFT study of volatile organic compounds adsorption on transition metal deposited graphene. Appl Surf Sci 396:1712–1718. https://doi.org/10.1016/j.apsusc.2016.11.238
Pulido A, Boronat M, Corma A (2011) Theoretical investigation of gold clusters supported on graphene sheets. New J Chem 35(10):2153–2161. https://doi.org/10.1039/c1nj20215d
Rangel E, Magana LF, Sansores LE (2014) A theoretical study of the interaction of hydrogen and oxygen with palladium or gold adsorbed on pyridine-like nitrogen-doped graphene. ChemPhysChem 15(18):4042–4048. https://doi.org/10.1002/cphc.201402532
Tang Y, Yang Z, Dai X (2011) Trapping of metal atoms in the defects on graphene. J Chem Phys 135(22):224704. https://doi.org/10.1063/1.3666849
Zhang W, Sun LT, Xu ZJ, Krasheninnikov AV, Huai P, Zhu ZY, Banhart F (2010) Migration of gold atoms in graphene ribbons: Role of the edges. Phys Rev B. https://doi.org/10.1103/PhysRevB.81.125425
Bulushev DA, Zacharska M, Shlyakhova EV, Chuvilin AL, Guo Y, Beloshapkin S, Okotrub AV, Bulusheva LG (2016) Single isolated Pd2+ cations supported on N-doped carbon as active sites for hydrogen production from formic acid decomposition. ACS Catal 6(2):681–691. https://doi.org/10.1021/acscatal.5b02381
Zacharska M, Bulusheva LG, Lisitsyn AS, Beloshapkin S, Guo Y, Chuvilin AL, Shlyakhova EV, Podyacheva OY, Leahy JJ, Okotrub AV, Bulushev DA (2017) Factors influencing the performance of Pd/C catalysts in the green production of hydrogen from formic acid. ChemSusChem 10(4):720–730. https://doi.org/10.1002/cssc.201601637
Podyacheva OY, Bulushev DA, Suboch AN, Svintsitskiy DA, Lisitsyn AS, Modin E, Chuvilin A, Gerasimov EY, Sobolev VI, Parmon VN (2018) Highly stable single-atom catalyst with Ionic Pd active sites supported on N-doped carbon nanotubes for formic acid decomposition. ChemSusChem 11(21):3724–3727. https://doi.org/10.1002/cssc.201801679
Bulushev DA, Zacharska M, Lisitsyn AS, Podyacheva OY, Hage FS, Ramasse QM, Bangert U, Bulusheva LG (2016) Single atoms of Pt-group metals stabilized by N-doped carbon nanofibers for efficient hydrogen production from formic acid. ACS Catal 6:3442–3451. https://doi.org/10.1021/acscatal.6b00476
Zacharska M, Podyacheva OY, Kibis LS, Boronin AI, Senkovskiy BV, Gerasimov EY, Taran OP, Ayusheev AB, Parmon VN, Leahy JJ, Bulushev DA (2015) Ruthenium clusters on carbon nanofibers for formic acid decomposition: effect of doping the support with nitrogen. ChemCatChem 7(18):2910–2917. https://doi.org/10.1002/cctc.201500216
Bulushev DA, Chuvilin AL, Sobolev VI, Stolyarova SG, Shubin YV, Asanov IP, Ishchenko AV, Magnani G, Ricco M, Okotrub AV, Bulusheva LG (2017) Copper on carbon materials: stabilization by nitrogen doping. J Mater Chem A 5(21):10574–10583. https://doi.org/10.1039/C7TA02282D
Zhao J, Xu J, Xu J, Zhang T, Di X, Ni J, Li X (2015) Enhancement of Au/AC acetylene hydrochlorination catalyst activity and stability via nitrogen-modified activated carbon support. Chem Eng J 262:1152–1160. https://doi.org/10.1016/j.cej.2014.10.071
Gil S, Lucas PJ, Nieto-Márquez A, Sánchez-Silva L, Giroir-Fendler A, Romero A, Valverde JL (2014) Synthesis and characterization of nitrogen-doped carbon nanospheres decorated with Au nanoparticles for the liquid-phase oxidation of glycerol. Ind Eng Chem Res 53(43):16696–16706. https://doi.org/10.1021/ie502873x
Lin R, Albani D, Fako E, Kaiser SK, Safonova OV, López N, Pérez-Ramírez J (2019) Design of single gold atoms on nitrogen-doped carbon for molecular recognition in alkyne semi-hydrogenation. Angew Chem Int Ed 58(2):504–509. https://doi.org/10.1002/anie.201805820
Bulushev DA, Ross JRH (2018) Towards sustainable production of formic acid. ChemSusChem 11:821–836. https://doi.org/10.1002/cssc.201702075
Preuster P, Albert J (2018) Biogenic formic acid as a green hydrogen carrier. Energy Technology 6(3):501–509. https://doi.org/10.1002/ente.201700572
Kamarudin SK, Shamsul NS, Ghani JA, Chia SK, Liew HS, Samsudin AS (2013) Production of methanol from biomass waste via pyrolysis. Biores Technol 129:463–468. https://doi.org/10.1016/j.biortech.2012.11.016
Hamelinck CN, Faaij APC (2002) Future prospects for production of methanol and hydrogen from biomass. J Power Sources 111(1):1–22. https://doi.org/10.1016/S0378-7753(02)00220-3
Bulushev DA, Ross JRH (2018) Heterogeneous catalysts for hydrogenation of CO2 and bicarbonates to formic acid and formates. Catal Rev 60(4):566–593. https://doi.org/10.1080/01614940.2018.1476806
Joo O-S, Jung K-D, Moon I, Rozovskii AY, Lin GI, Han S-H, Uhm S-J (1999) Carbon dioxide hydrogenation to form methanol via a reverse-water-gas-shift reaction (the CAMERE process). Ind Eng Chem Res 38(5):1808–1812. https://doi.org/10.1021/ie9806848
Álvarez A, Bansode A, Urakawa A, Bavykina AV, Wezendonk TA, Makkee M, Gascon J, Kapteijn F (2017) Challenges in the greener production of formates/formic acid, methanol, and DME by heterogeneously catalyzed CO2 hydrogenation processes. Chem Rev 117:9804–9838. https://doi.org/10.1021/acs.chemrev.6b00816
Shlyakhova EV, Bulusheva LG, Kanygin MA, Plyusnin PE, Kovalenko KA, Senkovskiy BV, Okotrub AV (2014) Synthesis of nitrogen-containing porous carbon using calcium oxide nanoparticles. Phys Status Solidi B 251:2607–2612. https://doi.org/10.1002/pssb.201451228
Perdew JP, Burke K, Ernzerhof M (1996) Generalized gradient approximation made simple. Phys Rev Lett 77:3865. https://doi.org/10.1103/PhysRevLett.77.3865
Bulushev DA, Yuranov I, Suvorova EI, Buffat PA, Kiwi-Minsker L (2004) Highly dispersed gold on activated carbon fibers for low-temperature CO oxidation. J Catal 224(1):8–17. https://doi.org/10.1016/j.jcat.2004.02.014
Wojnicki M, Rudnik E, Luty-Blocho M, Socha RP, Pedzich Z, Fitzner K, Mech K (2016) Kinetic studies of gold recovery from diluted chloride aqueous solutions using activated carbon organosorb 10 CO. Aust J Chem 69(3):254–261. https://doi.org/10.1071/ch15275
Kim SM, Kim KK, Jo YW, Park MH, Chae SJ, Duong DL, Yang CW, Kong J, Lee YH (2011) Role of anions in the AuCl3-doping of carbon nanotubes. ACS Nano 5(2):1236–1242. https://doi.org/10.1021/nn1028532
Bowden B, Davies M, Davies PR, Guan S, Morgan DJ, Roberts V, Wotton D (2018) The deposition of metal nanoparticles on carbon surfaces: the role of specific functional groups. Faraday Discuss 208:455–470. https://doi.org/10.1039/C7FD00210F
Podyacheva OY, Ismagilov ZR (2015) Nitrogen-doped carbon nanomaterials: to the mechanism of growth, electrical conductivity and application in catalysis. Catal Today 249:12–22. https://doi.org/10.1016/j.cattod.2014.10.033
Cao Y, Mao S, Li M, Chen Y, Wang Y (2017) Metal/porous carbon composites for heterogeneous catalysis: old catalysts with improved performance promoted by N-doping. ACS Catal 7(12):8090–8112. https://doi.org/10.1021/acscatal.7b02335
Fong Y-Y, Visser BR, Gascooke JR, Cowie BCC, Thomsen L, Metha GF, Buntine MA, Harris HH (2011) Photoreduction kinetics of sodium tetrachloroaurate under synchrotron soft X-ray exposure. Langmuir 27(13):8099–8104. https://doi.org/10.1021/la200463k
Mananghaya M, Rodulfo E, Santos GN, Villagracia AR, Ladines AN (2012) Theoretical investigation on single-wall carbon nanotubes doped with nitrogen, pyridine-like nitrogen defects, and transition metal atoms. J Nanomater. https://doi.org/10.1155/2012/104891
Singh S, Li S, Carrasquillo-Flores R, Alba-Rubio AC, Dumesic JA, Mavrikakis M (2014) Formic acid decomposition on Au catalysts: DFT, microkinetic modeling, and reaction kinetics experiments. AIChE J 60(4):1303–1319. https://doi.org/10.1002/aic.14401
Rousseau R, Marx D (2000) The interaction of gold clusters with methanol molecules: Ab initio molecular dynamics of Au +n CH3OH and AunCH3OH. J Chem Phys 112(2):761–769. https://doi.org/10.1063/1.480719
Li Y-C, Yang C-L, Sun M-Y, Li X-X, An Y-P, Wang M-S, Ma X-G, Wang D-H (2009) Density functional theory studies of Aun+(CH3OH)m (n = 3, 5, m = 1 − 5) complexes. J Phys Chem A 113(7):1353–1359. https://doi.org/10.1021/jp808040v
N’dollo M, Moussounda PS, Dintzer T, M’Passi-Mabiala B, Garin F (2013) Density functional theory (DFT) investigation of the adsorption of the CH3OH/Au(100) system. Surf Interface Anal 45(9):1410–1418. https://doi.org/10.1002/sia.5302
Gong J, Flaherty DW, Ojifinni RA, White JM, Mullins CB (2008) Surface chemistry of methanol on clean and atomic oxygen pre-covered Au(111). J Phys Chem C 112(14):5501–5509. https://doi.org/10.1021/jp0763735
Acknowledgements
This publication was conducted with the financial support of the Russian Science Foundation (Grant 16-13-00016).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bulushev, D.A., Chuvilin, A.L., Sobolev, V.I. et al. Single Au Atoms on the Surface of N-Free and N-Doped Carbon: Interaction with Formic Acid and Methanol Molecules. Top Catal 62, 508–517 (2019). https://doi.org/10.1007/s11244-019-01166-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-019-01166-1