Abstract
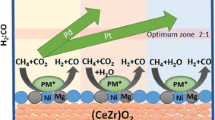
Low temperature dry reforming of methane was studied over (0.13–0.51 wt%) Pd and (0.16–0.64 wt%) Pt doped 1.4 wt%Ni–1.0 wt%Mg catalysts on a ceria–zirconia oxide support, as well as control catalysts containing Pt or Pd, but not Ni and Mg. Temperature-programmed reduction studies showed Pd catalysts having initial reduction peaks at lower temperatures compared to Pt catalysts. Reaction studies showed the lowest 10 % conversion of methane at 383 °C for the 0.13%Pd–1.39%Ni–1.0%Mg/(Ce0.6Zr0.4)O2 catalyst. The same catalyst also had the lowest 10 % conversion temperature (366 °C) for carbon dioxide. Syngas ratios (H2:CO) for Pd catalysts (0.24–0.41) were better than Pt catalysts (0.22–0.30) at 450 °C, however still not close to the stoichiometric ratio as a result of the RWGS reaction simultaneously occurring at the low operating temperatures studied. Turnover frequencies were calculated from steady-state reaction experiments done in the temperature range of 430–470 °C. Both catalysts’ TOFs increased with increasing temperature, however overall Pt catalysts had marginally higher TOFs (2.69–4.74 s−1) compared to Pd catalysts (2.40–4.58 s−1). Although both Pd and Pt catalysts had comparable activities and rates, the 0.13 %Pd–1.39 %Ni–1.0 %Mg/(Ce0.6Zr0.4)O2 catalyst was selected as the most promising one because of its higher H2:CO ratios, negligible changes during reaction as evidenced in post-reaction characterization, more basic sites that may decrease coking, and its lower cost.





Similar content being viewed by others
References
Methane emissions (2010) EPA, http://www.epa.gov/climatechange/ghgemissions/gases/ch4.html. Accessed 07 July 2014
Damyanova S, Pawelec B, Arishtirova K, Huerta MVM, Fierro JLG (2009) The effect of CeO2 on the surface and catalytic properties of Pt/CeO2–ZrO2 catalysts for methane dry reforming. Appl Catal B 89:149–159
Zhang S, Muratsugu S, Ishiguro N, Tada M (2013) Ceria-doped Ni/SBA-16 catalysts for dry reforming of methane. ACS Catal 3:1855–1864
Elsayed NH, Roberts NRM, Joseph B, Kuhn JN (2015) Low temperature dry reforming of methane over Pt–Ni–Mg/ceria–zirconia catalysts. Appl Catal B 179:213–219
Dry ME (2004) In: Fischer–Tropsch Technology: Studies in Surface Science and Catalysis. Elsevier, USA
Dry ME (2002) The Fischer–Tropsch process: 1950–2000. Catal Today 71:227–241
Rostrup-Nielsen JR (2000) New aspects of syngas production and use. Catal Today 63:159–164
Mortensen PM, Dybkjær I (2015) Industrial scale experience on steam reforming of CO 2-rich gas. Appl Catal A 495:141–151
Walker DM, Pettit SL, Wolan JT, Kuhn JN (2012) Synthesis gas production to desired hydrogen to carbon monoxide ratios by tri-reforming of methane using Ni–MgO–(Ce, Zr)O2 catalysts. Appl Catal A 445–446:61–68
Amin NAS, Yaw TC (2007) Thermodynamic equilibrium analysis of combined carbon dioxide reforming with partial oxidation of methane to syngas. Int J Hydrog Energy 32:1789–1798
Nakagawa K, Anzai K, Matsui N, Ikenaga N, Suzuki T, Teng Y, Kobayashi T, Haruta M (1998) Effect of support on the conversion of methane to synthesis gas over supported iridium catalysts. Catal Lett 51:163–167
Li Y, Wang Y, Zhang X, Mi Z (2008) Thermodynamic analysis of autothermal steam and CO2 reforming of methane. Int J Hydrog Energy 33:2507–2514
Bradford M, Vannice M (1999) CO2 reforming of CH4. Catal Rev 41:1–42
Simakov DS, Luo HY, Román-Leshkov Y (2015) Ultra-low loading Ru/γ-Al2 O3: a highly active and stable catalyst for low temperature solar thermal reforming of methane. Appl Catal B 168–169:540–549
Zhang Z, Verykios XE (1996) Carbon dioxide reforming of methane to synthesis gas over Ni/La2O3 catalysts. Appl Catal A 138:109–133
Valderrama G, Urbina de Navarro C, Goldwasser MR (2013) CO2 reforming of CH4 over Co–La-based perovskite-type catalyst precursors. J Power Sources 234:31–37
Kumar P, Sun Y, Idem RO (2007) Nickel-based ceria, zirconia, and ceria–zirconia catalytic systems for low-temperature carbon dioxide reforming of methane. Energy Fuels 21:3113–3123
Olah GA, Goeppert A, Czaun M, Prakash GKS (2013) Bi-reforming of methane from any source with steam and carbon dioxide exclusively to metgas (CO–2H2) for methanol and hydrocarbon synthesis. J Am Chem Soc 135:648–650
Rezaei M, Alavi SM, Sahebdelfar S, Yan Z-F (2006) Syngas production by methane reforming with carbon dioxide on noble metal catalysts. J Nat Gas Chem 15:327–334
Bradford MCJ, Vannice MA (1996) Catalytic reforming of methane with carbon dioxide over nickel catalysts I. Catalyst characterization and activity. Appl Catal A 142:73–96
Lemonidou AA, Vasalos IA (2002) Carbon dioxide reforming of methane over 5 wt% Ni/CaO–Al2O3 catalyst. Appl Catal A 228:227–235
Courson C, Udron L, Świerczyński D, Petit C, Kiennemann A (2002) Hydrogen production from biomass gasification on nickel catalysts: tests for dry reforming of methane. Catal Today 76:75–86
Courson C, Makaga E, Petit C, Kiennemann A (2000) Development of Ni catalysts for gas production from biomass gasification. Reactivity in steam- and dry-reforming. Catal Today 63:427–437
Guo J, Lou H, Zhao H, Chai D, Zheng X (2004) Dry reforming of methane over nickel catalysts supported on magnesium aluminate spinels. Appl Catal A 273:75–82
Dantas SC, Escritori JC, Soares RR, Hori CE (2010) Effect of different promoters on Ni/CeZrO2 catalyst for autothermal reforming and partial oxidation of methane. Chem Eng J 156:380–387
Crisafulli C, Scirè S, Maggiore R, Minicò S, Galvagno S (1999) CO2 reforming of methane over Ni–Ru and Ni–Pd bimetallic catalysts. Catal Lett 59:21–26
Kim G, Cho D-S, Kim K-H, Kim J-H (1994) The reaction of CO2 with CH4 to synthesize H2 and CO over nickel-loaded Y-zeolites. Catal Lett 28:41–52
Rossignol S, Gérard F, Duprez D (1999) Effect of the preparation method on the properties of zirconia-ceria materials. J Mater Chem 9:1615–1620
Goguet A, Meunier F, Breen JP, Burch R, Petch MI, Faur Ghenciu A (2004) Study of the origin of the deactivation of a Pt/CeO2 catalyst during reverse water gas shift (RWGS) reaction. J Catal 226:382–392
Dong W-S, Jun K-W, Roh H-S, Liu Z-W, Park S-E (2002) Comparative Study on Partial Oxidation of Methane over Ni/ZrO2, Ni/CeO2 and Ni/Ce–ZrO2 Catalysts. Catal Lett 78:215–222
Escritori JC, Dantas SC, Soares RR, Hori CE (2009) Methane autothermal reforming on nickel–ceria–zirconia based catalysts. Catal Commun 10:1090–1094
Passos FB, de Oliveira ER, Mattos LV, Noronha FB (2005) Partial oxidation of methane to synthesis gas on Pt/CexZr1 − xO2 catalysts: the effect of the support reducibility and of the metal dispersion on the stability of the catalysts. Catal Today 101:23–30
Boaro M, Vicario M, de Leitenburg C, Dolcetti G, Trovarelli A (2003) The use of temperature-programmed and dynamic/transient methods in catalysis: characterization of ceria-based, model three-way catalysts. Catal Today 77:407–417
Dong W-S, Roh H-S, Jun K-W, Park S-E, Oh Y-S (2002) Methane reforming over Ni/Ce-ZrO2 catalysts: effect of nickel content. Appl Catal A 226:63–72
Giordano F, Trovarelli A, de Leitenburg C, Giona M (2000) A Model for the Temperature-Programmed Reduction of Low and High Surface Area Ceria. J Catal 193:273–282
Yu Q, Chen W, Li Y, Jin M, Suo Z (2010) The action of Pt in bimetallic Au–Pt/CeO2 catalyst for water–gas shift reaction. Catal Today 158:324–328
Roland U, Braunschweig T, Roessner F (1997) On the nature of spilt-over hydrogen. J Mol Catal A 127:61–84
Rogemond E, Fréty R, Perrichon V, Primet M, Salasc S, Chevrier M, Gauthier C, Mathis F (1997) Preparation of alumina-supported ceria. II. Measurement of ceria surface area after impregnation with platinum or rhodium. J Catal 169:120–131
Kamiuchi N, Haneda M, Ozawa M (2013) CO oxidation over Pt/Ce–Zr oxide catalysts with low content of platinum and cerium components. Catal Today 201:79–84
Devi KS, Jayashree S (2013) Modified ceria as a substitute for sulfuric acid in the liquid phase nitration of toluene. React Kinet Mech Catal 108:183–192
Roh H-S, Jun K-W, Dong W-S, Chang J-S, Park S-E, Joe Y-I (2002) Highly active and stable Ni/Ce–ZrO2 catalyst for H2 production from methane. J Mol Catal A 181:137–142
Liotta LF, Macaluso A, Longo A, Pantaleo G, Martorana A, Deganello G (2003) Effects of redox treatments on the structural composition of a ceria–zirconia oxide for application in the three-way catalysis. Appl Catal A 240:295–307
Baudouin D, Candy J-P, Rodemerck U, Krumeich F, Veyre L, Webb PB, Thieuleux C, Copéret C (2014) Preparation of Sn-doped 2–3 nm Ni nanoparticles supported on SiO2 via surface organometallic chemistry for low temperature dry reforming catalyst: the effect of tin doping on activity, selectivity and stability. Catal Today 235:237–244
Chen J, Yao C, Zhao Y, Jia P (2010) Synthesis gas production from dry reforming of methane over Ce0.75Zr0.25O2-supported Ru catalysts. Int J Hydrog Energy 35:1630–1642
Qin D, Lapszewicz J (1994) Study of mixed steam and CO2 reforming of CH4 to syngas on MgO-supported metals. Catal Today 21:551–560
Kambolis A, Matralis H, Trovarelli A, Papadopoulou C (2010) Ni/CeO2-ZrO2 catalysts for the dry reforming of methane. Appl Catal A 377:16–26
Özkara-Aydınoğlu Ş, Erhan Aksoylu A (2013) A comparative study on the kinetics of carbon dioxide reforming of methane over Pt–Ni/Al2O3 catalyst: effect of Pt/Ni ratio. Chem Eng J 215–216:542–549
Acknowledgments
The authors gratefully acknowledge funding from Hinkley Center for Solid and Hazardous Waste Management and the Graduate Students Success Fellowship (to NHE) that is administered by the USF School of Graduate Studies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elsayed, N.H., Roberts, N.R.M., Joseph, B. et al. Comparison of Pd–Ni–Mg/Ceria–Zirconia and Pt–Ni–Mg/Ceria–Zirconia Catalysts for Syngas Production via Low Temperature Reforming of Model Biogas. Top Catal 59, 138–146 (2016). https://doi.org/10.1007/s11244-015-0513-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-015-0513-7