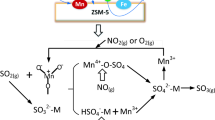
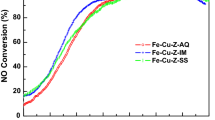
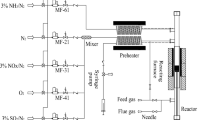
Abstract
Fe-ZSM-5 catalysts, prepared by different methods, have been characterized by BET, ICP-AES, XRD, XPS, NH3-TPD and NO-TPD and evaluated for NOx reduction according to standard NH3-SCR, NH3 oxidation and NO oxidation, in absence and presence of water. The presence of water has a significant influence on both the SCR and oxidation reactions. The most active catalyst for NH3-SCR is prepared by ion exchange using FeCl2 as iron precursor. The XPS results indicate that Fe2+ ions are the main active sites for the SCR reactions, while Fe3+ ions are the primarily active sites for oxidation of ammonia.







Similar content being viewed by others
References
Metkar PS, Salazar N, Muncrief R, Balakotaiah V, Harold MP (2011) Appl Catal B 104:110–126
Iwasaki M, Shinjoh H (2010) Appl Catal A 390:71–77
Twigg MV (2011) Catal Today 163:33–41
Auvray X, Partridge WP, Choi J-S, Pihl JA, Yezerets A, Kamasamudram K, Currier NW, Olsson L (2012) Appl Catal B 126:144–152
Colombo M, Nova I, Tronconi E, Schmeißer V, Bandl-Konrad B, Zimmermann L (2012) Appl Catal B 111–112:106–118
Forzatti P, Nova I, Tronconi E, Kustov A, Thøgersen JR (2012) Catal Today 184:153–159
Kröcher O (2007) In: Granger P, Pârvulescu VI (eds), Studies in surface science and catalysis. Elsevier, Amsterdam, pp 261–289
Brandenberger S, Krocher O, Tissler A, Althoff R (2008) Catal Rev 50:492–531
Rauscher M, Kesore K, Monnig R, Schwieger W, Tissler A, Turek T (1999) Appl Catal A 184:249–256
Pieterse JAZ, Booneveld S, van den Brink RW (2004) Appl Catal B 51:215–228
Long RQ, Yang RT (2001) Catal Lett 74:201–205
Kumar MS, Schwidder M, Grunert W, Bruckner A (2004) J Catal 227:384–397
Feng XB, Hall WK (1997) J Catal 166:368–376
Long RQ, Yang RT (1999) J Catal 188:332–339
Balle P, Geiger B, Kureti S (2009) Appl Catal B 85:109–119
Shwan S, Jansson J, Korsgren J, Olsson L, Skoglundh M (2012) Catal Today 197:24–37
Shi X, Liu F, Shan W, He H (2012) Chin J Catal 33:454–464
IZA Synthesis Commission. http://www.iza-online.org/synthesis/. Accessed 12 April 2013
Qi GS, Yang RT (2005) Appl Catal A 287:25–33
Iwasaki M, Yamazaki K, Banno K, Shinjoh H (2008) J Catal 260:205–216
Delahay G, Valade D, Guzman-Vargas A, Coq B (2005) Appl Catal B 55:149–155
Shi XY, Liu FD, Shan WP, He H (2012) Chin J Catal 33:454–464
Okpalugo TIT, Papakonstantinou P, Murphy H, McLaughlin J, Brown NMD (2005) Carbon 43:153–161
Sjövall H, Olsson L, Fridell E, Blint RJ (2006) Appl Catal B 64:180–188
Brandenberger S, Krocher O, Tissler A, Althoff R (2010) Appl Catal B 95:348–357
Brandenberger S, Krocher O, Tissler A, Althoff R (2010) Appl Catal A 373:168–175
Devadas M, Krocher O, Elsener M, Wokaun A, Mitrikas G, Soger N, Pfeifer M, Demel Y, Mussmann L (2007) Catal Today 119:137–144
Moulder JF, Stickle WF, Sobol PE, Bomben KD (1992) Handbook of X-ray photoelectron spectroscopy. Perkin-Elmer Corp., Eden Prairie, MN, pp 80–81
Shwan S, Nedyalkova R, Jansson J, Korsgren J, Olsson L, Skoglundh M (2012) Ind Eng Chem Res 51(39):12762–12772
Skarlis SA, Berthout D, Nicolle A, Dujardin C, Granger P (2012) J Phys Chem C 116:8437–8448
Lonyi F, Valyon J (2001) Microporous Mesoporous Mater 47:293–301
Sawa M, Niwa M, Murakami Y (1990) Zeolites 10:307–309
Sato H (1997) Catal Rev 39:395–424
Acknowledgments
This work has been performed within the Competence Centre for Catalysis (KCK) which is hosted by Chalmers University of Technology and financially supported by the Swedish Energy Agency and the member companies: AB Volvo, ECAPS AB, Haldor Topsøe A/S, Scania CV AB and Volvo Car Corporation AB. The authors would also like to thank ALS Scandinavia AB for help with the ICP-AES analysis, Lars Ilver for help with the XPS analysis and Soran Shwan for helpful hints.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shishkin, A., Carlsson, PA., Härelind, H. et al. Effect of Preparation Procedure on the Catalytic Properties of Fe-ZSM-5 as SCR Catalyst. Top Catal 56, 567–575 (2013). https://doi.org/10.1007/s11244-013-0016-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-013-0016-3