Abstract
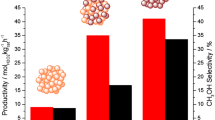
It is shown that Au–zinc oxide–alumina catalysts are suitable for the water–gas shift reaction and for methanol (MeOH) and DME synthesis, indicating their use in a direct single-stage process for converting syngas to a DME + methanol mixture. Temperatures above 340 °C were required in order to obtain reasonable catalytic activity. A 67 % DME selectivity was achieved at 380 °C with a low space velocity 0.75 dm3 h−1 g−1 and 50 bar. The lower CO conversions at the higher temperature of 460 °C was probably due to the MeOH equilibrium limitation in the range of temperatures 340 to 460 °C, but deactivation is observed as well, above 460 °C. Au/ZnO/γ-Al2O3 is more stable than traditional copper-based catalysts, which are stable below about 300 °C, and then only in the absence of water. The gold composite catalyst was mainly selective toward DME, MeOH and CH4, and to C2 to C5 hydrocarbons. An analysis of the main reactions involved indicates that only the methanol synthesis reaction reaches a near-equilibrium situation, with the other reactions being under kinetic control.
















Similar content being viewed by others
References
Keshav TR, Basu S (2007) Fuel Proc Technol 88:493
Marchionna M, Patrini R, Sanfilippo D, Migliavacca G (2008) Fuel Proc Technol 89:1255
Olah G, Goeppert A, Prakash Surya (2009) Beyond oil and gas: the methanol economy. Wiley-VCH, Weinheim
Fleisch TH, Sills RA, Briscoe MD (2002) J Nat Gas Chem 11:1
Varisli D, Dogu T (2008) J Sci 21:37
Semelesberger TA, Borup RL, Greene HL (2006) J Power Sources 156:497
Lu W-Z, Teng L-H, Xioa WD (2004) Chem Eng Sci 59:5455
Tanaka Y, Kikuchi R, Takeguchi T, Eguchi K (2007) Appl Catal B 57:211
Moradi GR, Nostrati S, Yaripour F (2007) Catal Commun 8:598
Seo Y, Jo JH, Ryu H-J, Yi C-K, Jin GT (2008) J Chem Eng Jpn 41:585
Cocco D, Pettinau A, Cau GJ (2006) J Power Energy 220:95
Aguayo AT, Erena J, Siera I, Olazar M, Bilbao J (2005) Catal Today 106:265
Moradi GR, Ghanei R, Yaripour F (2007) Int J Chem Reactor Eng 5:A14
Zhang Q, Li Q, Asami K, Asaoka S, Fujimoto K (2004) Fuel Proc Technol 85:1139
Pyatnitskii YI, Strizhak PE, Lunev NK (2009) Theor Exper Chem 45:312
Sakurai H, Haruta M (1995) Appl Catal A 127:93
Mpela A, Hildebrandt D, Glasser D, Scurrell MS (2007) Gold Bull 40:219
Wang L, Fang D, Huang X, Zhang S, Qi Y, Liu Z (2006) J Nat Gas Chem 15:38
Moradi G, Ahmadpour J, Nazari M, Yaripour F (2008) Ind Eng Chem Res 47:7672
Ramos FS, de Farias AMD, Borges LEP, Monteiro JL, Fraga MA, Sousa Aguiar EF (2005) Catal Today 101:39
Zhao Y, Mpela A, Enache DI, Taylor SH, Hildebrandt D, Glasser D, Hutchings GJ, Atkins MP, Scurrell MS (2007) Stud Surf Sci Catal 163:141
Kozlov AI, Kozlova AP, Liu H, Iwasawa Y (1999) Appl Catal A 182:9
Svelle S, Ronning PO, Olsbye U, Kolboe S (2005) J Catal 234:385
Zhu Y, Wang S, Ge X, Liu Q, Luo Z, Cen K (2010) Fuel Proc Technol 91:424
Dutta P, Roy SC, Nandi LN, Samuel P, Pillai SM, Bhat BD, Ravindranathan M (2004) J Mol Catal A 223:231
Wu X, Abraha MG, Anthony RG (2004) Appl Catal A 260:63
Campbell CT, Daube KA, White JM (1987) Surf Sci 182:458
Leon y Leon CA, Vannice MA (1991) Appl Catal 69:291
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mbuyi, K.G., Scurrell, M.S., Hildebrandt, D. et al. Conversion of Synthesis Gas to Dimethylether Over Gold-based Catalysts. Top Catal 55, 771–781 (2012). https://doi.org/10.1007/s11244-012-9865-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-012-9865-4