Abstract
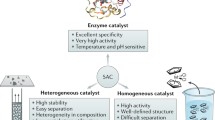
Whereas cooperative effect in catalysis, in which multiple chemical interactions participate cooperatively to achieve significant enhancement in catalytic activity and/or selectivity, is common in enzymatic reactions, it has been sparingly employed in heterogeneous catalytic systems. Here, some recent literature examples of abiotic catalysis, with emphasis on heterogeneous systems, that employ cooperation between acid and base and two metal centers are briefly described to demonstrate the principles involved. Since effective cooperation places strict demand on the positions of the different functional groups, new synthetic methods and strategies are needed to design and construct structures useful for cooperative catalysis. Recent progress in our laboratory in synthesizing new nanocage structures that possess molecular-size cavities, atomic layer thick, porous shells with internal functional groups is described. These recent developments suggest possibilities of new catalytic transformations that have not been attempted before. This is illustrated with two speculative examples utilizing cooperative catalysis: oxidative hydrolytic desulfurization and terminal carbon activation of hydrocarbon molecules.











Similar content being viewed by others
References
Voet D, Voet J (2004) Biochemistry, vol 1. John Wiley & Sons, New York
Huh S, Chen H-T, Wiench JW, Pruski M, Lin VS-Y (2005) Angew Chem Int Ed 44:1826
Huh S, Chen H-T, Wiench JW, Pruski M, Lin VS-Y (2005) Angew Chem Int Ed 44:1826
Zeidan RK, Hwang S-J, Davis ME (2006) Angew Chem Int Ed 45:6332–6335
Kwong C, Huang R, Zhang M, Shi M, Toy P (2007) Chem Eur J 13:2369–2376
Martin M, Manea F, Fiammengo R, Prins L, Pasquato L, Scrimin P (2007) J Am Chem Soc 129:6982–6983
Livieri M, Mancin F, Saielli G, Chin J, Tonellato U (2007) Chem Eur J 13:2246–2256
Li H, Marks TJ (2006) Proc Natl Acad Sci 103:15295–15302
Xue W, Kung MC, Kung HH (2005) Chem Comm 2164–2166
Zhang Y, Jiang M, Zhao J, Zhou J, Chen D (2004) Macromolecules 37(4):1537–1543
Chen J, Wiley B, Li Z-Y, Campbell D, Saeki F, Cang H, Au L, Lee J, Li X, Xia Y (2005) Adv Mater (Weinheim, Germany) 17:2255–2261
Huang H, Remsen EE, Kowalewski T, Wooley KL (1999) J Am Chem Soc 121(15):3805–3806
Sanji T, Nakatsuka Y, Ohnishi S, Sakurai H (2000) Macromolec 33(23):8524–8526
Cornelissen JJLM, Connor EF, Kim H-C, Lee VY, Rice MP, Volksen W, Sundberg LK, Miller DR (2003) Chem Commun 8:1010–1011
Suh Y-W, Kung MC, Wang Y, Kung HH (2006) J Am Chem Soc 128:2776
Lee J-K, Suh Y-W, Kung MC, Downing CM, Kung HH (2007) Tetrahedron Lett 48:4919
Lee J-K, Kung MC, Suh Y-W, Kung HH (2008) Chem Mater 20(2):373–375
Acknowledgment
Support of this work by the US Department of Energy, Basic Energy Sciences, Grant No. DE-FG02-01ER15184.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, JK., Kung, M.C. & Kung, H.H. Cooperative Catalysis: A New Development in Heterogeneous Catalysis. Top Catal 49, 136–144 (2008). https://doi.org/10.1007/s11244-008-9087-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11244-008-9087-y