Abstract
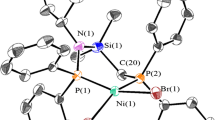
Two Schiff base macrocyclic ligands (L1 and L2) were synthesized using ethylenediamine and terephthalaldehyde (or isophthalaldehyde) as raw materials in acetonitrile solvent. Surprisingly, series of Schiff base macrocyclic ligands with different combination modes (L2-1, L2-2 and L2-3) with isophthalaldehyde as raw material were synthesized. Two Schiff base macrocyclic nickel complexes (Ni-1 and Ni-2) based on L1 and L2 for ethylene oligomerization were obtained with (DME)NiCl2. The synthesized ligands and their corresponding complexes were characterized by various analysis techniques to confirm their chemical structure and thermal stability. And the catalytic properties of the two nickel complexes were also investigated for ethylene oligomerization. When the ethylene pressure was 0.5 MPa, the Al/Ni ratio was 500:1, and the reaction time was 30 min in the presence of MAO, the catalytic activities of Ni-1 and Ni-2 were 4.53 × 104 g/(mol Ni·h) and 4.73 × 104 g/(mol Ni·h), respectively. Compared with PS-Ni complex based on 2,3-butanedione and p-phenylenediamine and three other nickel complexes with simpler ligand structures, Ni-1 and Ni-2 had lower catalytic activities and higher selectivity for C4 olefin because of the higher spatial resistance of the macrocycles.














Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available within the article.
References
Alferov KA, Belov GP, Meng Y (2017) Chromium catalysts for selective ethylene oligomerization to 1-hexene and 1-octene: recent results. Appl Catal A-Gen 542:71–124
Xu J, Wang R, Zheng L, Ma J, Yan W, Yang X, Wang J, Su X, Huang Y (2021) Unraveling the real active sites of an amorphous silica–alumina-supported nickel catalyst for highly efficient ethylene oligomerization. Catal Sci Technol 11:1510–1518
Moussa S, Concepción P, Arribas MA, Martínez A (2018) Nature of active nickel sites and initiation mechanism for ethylene oligomerization on heterogeneous Ni-beta catalysts. ACS Catal 8:3903–3912
Skupinska J (1991) Oligomerization of α-olefins to higher oligomers. Chem Rev 91:613–648
Arrozi USF, Bon V, Kutzscher C, Senkovska I, Kaskel S (2019) Towards highly active and stable nickel-based metal–organic frameworks as ethylene oligomerization catalysts. Dalton Trans 48:3415–3421
Finiels A, Fajula F, Hulea V (2014) Nickel-based solid catalysts for ethylene oligomerization—a review. Catal Sci Technol 4:2412–2426
Si G, Na Y, Chen C (2018) Ethylene (co)oligomerization by phosphine-pyridine based palladium and nickel catalysts. ChemCatChem 10:5135–5140
Chen L, Ma L, Jiang Y, Liu J, Li C, Zhang N, Wang J (2020) Synthesis and characterization of iron, cobalt and nickel complexes bearing para-phenylene-linked pyridine imine ligand and their catalytic properties for ethylene oligomerization. Polym Bull 78:415–432
Lu K, Jin F, Wu G, Ding Y (2019) The synergetic effect of acid and nickel sites on bifunctional MWW zeolite catalysts for ethylene oligomerization and aromatization. Sustain Energy Fuels 3:3569–3581
Gao R, Sun WH, Redshaw C (2013) Nickel complex pre-catalysts in ethylene polymerization: new approaches to elastomeric materials. Catal Sci Technol 3:1172
Koninckx E, Mendes PSF, Thybaut JW, Broadbelt LJ (2021) Ethylene oligomerization on nickel catalysts on a solid acid support: from new mechanistic insights to tunable bifunctionality. Appl Catal A-Gen 624:118296
de Oliveira LL, da Silva SM, Casagrande ACA, Stieler R, Casagrande OL (2018) Synthesis and characterization of Ni(II) complexes supported by phenoxy/naphthoxy-imine ligands with pendant N- and O-donor groups and their use in ethylene oligomerization. Appl Organomet Chem 32:e4414
Speiser F, Braunstein P, Saussine L (2005) Catalytic ethylene dimerization and oligomerization: recent developments with nickel complexes containing P, N-chelating ligands. Acc Chem Res 38:784–793
Song S, Xiao T, Liang T, Wang F, Redshaw C, Sun WH (2011) Synthesis, characterization and ethylene oligomerization behaviour of 8-(1-aryliminoethylidene)quinaldinylnickel dihalides. Catal Sci Technol 1:69–75
Suo H, Zhao T, Wang Y, Ban Q, Sun WH (2017) N-(2,2-dimethyl-1-(quinolin-2-yl)propylidene) arylaminonickel complexes and their ethylene oligomerization. Molecules 22:630
Zubkevich SV, Tuskaev VA, Gagieva SC (2020) Trapping the short-chain odd carbon number olefins using nickel(II)-catalyzed tandem ethylene oligomerization and Friedel-Crafts alkylation of toluene. ChemRxiv. https://doi.org/10.26434/chemrxiv.13359551.v1
Nyamato GS, Ojwach SO, Akerman MP (2016) Ethylene oligomerization studies by nickel(ii) complexes chelated by (amino)pyridine ligands: experimental and density functional theory studies. Dalton Trans 45:3407–3416
Gupta KC, Sutar AK (2008) Catalytic activities of Schiff base transition metal complexes. Coord Chem Rev 252:1420–1450
Abu-Dief AM, Mohamed IMA (2015) A review on versatile applications of transition metal complexes incorporating Schiff bases. Beni-Suef Univ J Basic Appl Sci 4:119–133
Utz D, Heinemann FW, Mukherjee J, Mukherjee R, Schindler S (2003) Synthesis and structural characterization of a new tetranuclear macrocyclic copper(I) complex. Z Anorg Allg Chem 629:2211–2215
Allmendinger M, Zell P, Amin A (2003) High yield synthesis, separation and structural characterization of new [N+N]-polyazamacrocycles. Heterocycles 60:1065–1081
Liu Y, Shen J, Sun C, Ren CL, Zeng HQ (2015) Intramolecularly hydrogen-bonded aromatic pentamers as modularly tunable macrocyclic receptors for selective recognition of metal ions. J Am Chem Soc 137:12055–12063
Gupta R, Mukherjee R (1997) A new tyrosinase model system: formation of a phenoxy- and hydroxy-bridged copper(II) complex with partial hydrolysis of a tetraaza macrocyclic Schiff base ligand. Inorg Chim Acta 263:133–137
Ding SY, Gao J, Wang Q, Zhang Y, Song WG, Su CY, Wang W (2011) Construction of covalent organic framework for catalysis: Pd/COF-LZU1 in Suzuki–Miyaura coupling reaction. J Am Chem Soc 133:19816–19822
Solórzano C, Méndez FJ, Brito JL, Silva P, Anacona JR, Bastardo-González E (2020) Heterogenization of Co(II)- and Cu(II)-complexes containing a terpyridine-based Schiff base macrocyclic ligand on thiol-functionalized mesostructured silica. J Organomet Chem 908:121073
Abudayyeh AM, Schott O, Feltham HLC, Hanan GS, Brooker S (2021) Copper catalysts for photo- and electro-catalytic hydrogen production. Inorg Chem Front 8:1015–1029
Li D, Ma F, Guo L, Huang J, Zhang Y, Li F, Li C (2021) Polynuclear (α-diimine) nickel(II) complex as catalyst for ethylene oligomerization. Appl Organomet Chem 36:e6509
Ceborska M, Tarnowska A, Ziach K, Jurczak J (2010) Dynamic combinatorial libraries of macrocycles derived from phthalic aldehydes and α, ω-diamines. Tetrahedron 66:9532–9537
Li G, Zhang B, Yan J, Wang Z (2014) Micro- and mesoporous poly(Schiff-base)s constructed from different building blocks and their adsorption behaviors towards organic vapors and CO2 gas. J Mater Chem A 2:18881–18888
Yang Y, Guan J, Qiu P, Kan Q (2009) Synthesis, characterization and catalytic properties of heterogeneous iron(III) tetradentate Schiff base complexes for the aerobic epoxidation of styrene. Transit Met Chem 35(3):263–270. https://doi.org/10.1007/s11243-009-9322-9
Ambrozini B, Dockal ER, Cavalheiro ÉTG (2013) Thermal behavior of tetradentate Schiff base chromium(III) complexes. J Therm Anal Calorim 115:979–986
Sun P, Yang J, Chen C (2020) Synthesis of a cellulosic Pd(salen)-type catalytic complex as a green and recyclable catalyst for cross-coupling reactions. Catal Lett 150:2900–2910
Alaghaz ANMA, Ammar YA, Bayoumi HA (2014) Synthesis, spectral characterization, thermal analysis, molecular modeling and antimicrobial activity of new potentially N2O2 azo-dye Schiff base complexes. J Mol Struct 1074:359–375
Kurokawa H, Miura K, Yamamoto K, Sakuragi T, Sugiyama T, Ohshima M, Miura H (2013) Oligomerization of ethylene to produce linear α-olefins using heterogeneous catalyst prepared by immobilization of α-diiminenickel(II) complex into fluorotetrasilicic mica interlayer. Catalysts 3:125–136
Li Y, Guo L, Li F, Huang J, Li D, Zhang N, Li C (2021) Influence of the ligand structure on the properties of bidentate salicylaldimine nickel(II) complexes in ethylene oligomerization. Transit Met Chem 46:307–314
Liu J, Wang J, Chen L, Zhang N, Lan T, Wang L (2019) Ethylene oligomerization study bearing nickel(II) and cobalt(II) complexes with N, O-donor Schiff bases as ligands. ChemistrySelect 4:13959–13963
Zhang N, Fu Z, Jiang Y, Guo D, Chen L, Wang J (2018) Synthesis of novel structure based low generation branched salicylaldimine nickel complexes and their catalysis of ethylene oligomerization. ChemistrySelect 3:10428–10433
Helldörfer M, Milius W, Alt HG (2003) The influence of halogen substituents at the ligand framework of (α-diimine)nickel(II) catalyst precursors on their behavior in ethylene oligomerization and polymerization. J Mol Catal A-Chem 197:1–13
Acknowledgements
This work was supported by the Heilongjiang Scientific Research Foundation for Postdoctoral Settlement of China (Grant No. 16190023) and the Heilongjiang Natural Science Foundation of China (Grant No. E2018012). For the characterization work, we are appreciative of the State Key Lab of Inorganic Synthesis and Preparative Chemistry at Jilin University and Analysis and Test Center of Northeast Petroleum University for the characterization work.
Author information
Authors and Affiliations
Contributions
JL wrote the main manuscript text and prepared all figures. CL helped to revise the text. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Guo, L., Huo, H. et al. Preparation of nickel catalysts bearing Schiff base macrocycles and their performance in ethylene oligomerization. Transit Met Chem 48, 99–111 (2023). https://doi.org/10.1007/s11243-023-00527-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-023-00527-w