Abstract
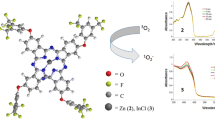
In this study, new 4-chloro-5-(2-isopropyl-5-methylphenoxy)phthalonitrile compound, containing bioactive thymoxy group, and its metal-free phthalocyanine and metallo-phthalocyanine derivatives were synthesized for the first time. Their structures were determined by spectroscopic methods such as FTIR, UV–Vis, 1H-, and 13C-NMR (for phthalonitrile derivative), MALDI-TOF mass spectrometry (for phthalocyanine derivatives) and elemental analysis as well. The phthalocyanines showed excellent solubility in polar and nonpolar solvents without aggregation and absorb at long wavelengths with their high molar coefficient. In N,N-dimethylformamide, the effects of the type of central metal ions [metal-free, indium(III) acetate, lutetium(III) acetate, magnesium(II) or zinc(II)] in the phthalocyanine, containing bioactive thymoxy group, cavity on the spectroscopic, photophysical, and photochemical properties of the phthalocyanines were determined. These features are compared with each other. Lutetium(III) acetate phthalocyanine did not show any fluorescence, while metal-free phthalocyanine and indium(III) acetate phthalocyanine showed low fluorescence. It was determined that magnesium phthalocyanine significantly enriched the fluorescence, and zinc phthalocyanine had appropriate and sufficient fluorescence. Lutetium(III) acetate and zinc(II), especially indium(III) acetate phthalocyanines, could produce large amounts of singlet oxygen. Metal-free and magnesium phthalocyanines had the capacity to produce sufficient singlet oxygen (it means production of enough amount of singlet oxygen by a photosensitizer candidate during PDT applications). All phthalocyanines have sufficient and suitable photostability (it means an ideal photosensitizer should be stable under light irradiation until complete its PDT activation, and it should be decomposed after its PDT activation so that it does not accumulate in the body). With these determined properties, magnesium(II), especially indium(III) acetate and zinc(II) phthalocyanines, may be suitable candidates as type II photosensitizers for photodynamic therapy applications. Lutetium(III) acetate phthalocyanine may be a photosensitizer candidate in photocatalytic applications.









Similar content being viewed by others
Data and material availability
Not applicable.
References
Crucius G (2013) Synthesen nichtperipher glykokonjugierter Zink (II) phthalocyanine. Universitätsbibliothek Tübingen
Josefsen LB, Boyle RW (2012) Unique diagnostic and therapeutic roles of porphyrins and phthalocyanines in photodynamic therapy, imaging and theranostics. Theranostics 2:916
Hamblin MR, Hasan T (2004) Photodynamic therapy: a new antimicrobial approach to infectious disease? Photochem Photobiol Sci 3:436–450
Patrice T (2003) Photodynamic therapy. Royal Society of Chemistry, London
Hirth A, Michelsen U, Wöhrle D (1999) Photodynamische tumortherapie. Chem unserer Zeit 33:84–94
Whitacre CM, Feyes DK, Satoh T, Grossmann J, Mulvihill JW et al (2000) Photodynamic therapy with the phthalocyanine photosensitizer Pc 4 of SW480 human colon cancer xenografts in athymic mice. Clin Cancer Res 6:2021–2027
Allen CM, Sharman WM, Van Lier JE (2001) Current status of phthalocyanines in the photodynamic therapy of cancer. J Porphyr Phthalocyanines 5:161–169
Dolmans DE, Fukumura D, Jain RK (2003) Photodynamic therapy for cancer. Nat Rev Cancer 3:380–387
Brasseur N (2003) Sensitizers for PDT: phthalocyanines. In: Patrice T (ed) Photodynamic therapy, vol 2. The Royal Society of Chemistry, London, pp 105–118
Flom SR (2003) Nonlinear optical properties of phthalocyanines. In: Kadish KM, Smith KM, Guilard R (eds) The porphyrin handbook. Application of phthalocyanines. Elsevier Academic Press, California, pp 179–190
Tanaka M (2009) Phthalocyanines – high performance pigments and their applications. In: Faulkner EB, Schwartz RJ (eds) High performance pigments. John Wiley and Sons, Weinheim, pp 275–291
Wöhrle D, Schnurpfeil G (2003) Porphyrins and Phthalocyanines in Macromolecules-110. In: The Porphyrin Handbook. Academic Press, Amsterdam
Mckeown N, Weinreb S (2004) Science of Synthesis. Houben-Weyl Methods of Molecular Transformations–Hetarenes and Related Ring Systems. 17: 1237
Seikel E (2012) Axial funktionalisierte Metallophthalocyanine und-porphyrazine als Funktionsmoleküle für optoelektronische Anwendungen. Philipps-Universität Marburg
Wöhrle D, Eskes M, Shigehara K, Yamada A (1993) A Simple synthesis of 4, 5-disubstituted 1, 2-dicyanobenzenes and 2, 3, 9, 10, 16, 17, 23, 24-octasubstituted phthalocyanines. Synthesis 1993:194–196
Atalay Ş, Çoruh U, Akdemir N, Ağar E (2004) C–H⋯ π interactions in 4, 5-bis (2-isopropyl-5-methylphenoxy) phthalonitrile. Acta Crystallogr Sect E: Struct Rep Online 60:o303–o305
Nyokong T (2007) Effects of substituents on the photochemical and photophysical properties of main group metal phthalocyanines. Coord Chem Rev 251:1707–1722
Becker HG, Böttcher H, Dietz F, Rehorek D, Roewer G et al. (1983) Einführung in die Photochemie. Thieme Stuttgart
Klessinger M (1989) Physikalische organische Chemie. Verlag Chemie
Grofcsik A, Baranyai P, Bitter I, Csokai V, Kubinyi M et al (2004) Triple state properties of tetrasubstituted zinc phthalocyanine derivatives. J Mol Struct 704:11–15
Kumbhar PP, Dewang PM (2001) Eco-friendly pest management using monoterpenoids. I. Antifungal efficacy of thymol derivatives. J Sci Ind Res 60:645–648
Li Y, Wen J-m, Du C-j, Hu S-m, Chen J-x et al (2017) Thymol inhibits bladder cancer cell proliferation via inducing cell cycle arrest and apoptosis. Biochem Biophys Res Commun 491:530–536
Botelho M, Nogueira N, Bastos G, Fonseca S, Lemos T et al (2007) Antimicrobial activity of the essential oil from Lippia sidoides, carvacrol and thymol against oral pathogens. Braz J Med Biol Res 40:349–356
Braga PC, Dal Sasso M, Culici M, Bianchi T, Bordoni L, Marabini L (2006) Anti-inflammatory activity of thymol: inhibitory effect on the release of human neutrophil elastase. Pharmacology 77:130–136
Aeschbach R, Löliger J, Scott B, Murcia A, Butler J et al (1994) Antioxidant actions of thymol, carvacrol, 6-gingerol, zingerone and hydroxytyrosol. Food Chem Toxicol 32:31–36
Ma C, Tian D, Hou X, Chang Y, Cong F et al (2005) Synthesis and characterization of several soluble tetraphenoxy-substituted copper and zinc phthalocyanines. Synthesis 2005:741–748
Sobotta L, Lijewski S, Dlugaszewska J, Nowicka J, Mielcarek J, Goslinski T (2019) Photodynamic inactivation of Enterococcus faecalis by conjugates of zinc(II) phthalocyanines with thymol and carvacrol loaded into lipid vesicles. Inorg Chim Acta 489:180–190
Görlach B, Dachtler M, Glaser T, Albert K, Hanack M (2001) Synthesis and separation of structural isomers of 2(3),9(10),16(17),23(24)-tetrasubstituted phthalocyanines. Chem A Eur J 7:2459–2465
Atajanov R, Huraibat B, Odabaş Z, Özkaya AR (2023) Electrochemical, spectroelectrochemical, and electrocatalytic properties of novel soluble phthalocyanines containing peripheral thymoxy and chloride units. Inorg Chim Acta 547:121360
Fernandez JM, Bilgin MD, Grossweiner LI (1997) Singlet oxygen generation by photodynamic agents. J Photochem Photobiol B 37:131–140
Kubiak R, Janczak J, Śledź M, Bukowska E (2007) Comparative study of beryllium, magnesium and zinc phthalocyanine complexes with 4-picoline. Polyhedron 26:4179–4186
Acknowledgements
We are grateful to the Research Foundation of Marmara University, Commission of Scientific Research (BAPKO) for their support of this research as part of the project: FEN-C-YLP-120418-0164. In addition, we are very grateful to Assoc. Prof. Dr. Mehmet Pişkin for his skillful and fruitful collaboration.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
RA performed the synthesis, purification, and characterization of the starting compound and phthalocyanines in the article and write the manuscript. KK performed the photophysical and photochemical characterization of the phthalocyanines in the article. MD and ZO reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Atajanov, R., Khezami, K., Durmuş, M. et al. Synthesis, characterization, photophysical, and photochemical properties of novel phthalocyanines containing thymoxy groups as bioactive units. Transit Met Chem 48, 79–89 (2023). https://doi.org/10.1007/s11243-023-00525-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-023-00525-y