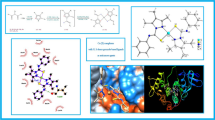
Abstract
The development of suitable compounds for the effective treatment of cancer is highly demanded. Inorganic complexes based on cobalt and copper centers have revealed a great potential for the treatment of various cancers. The current study aimed to develop effective pyrazole-based agents against breast cancer. A pyrazole-based ligand (L: Na[EtNCSPzMe2]) and its cobalt and copper complexes were synthesized, and the single crystals of the complexes were prepared for crystallographic analysis. The X-ray structure of the synthesized complexes indicated both of the complexes were mononuclear and monoclinic. The synthesized [Co(L)3] complex demonstrated a six-coordinated distorted octahedral geometry around the cobalt center, while the [Cu(L)2] complex occupied a four-coordinated seesaw geometry with N2S2 environment around the copper center. The compounds were evaluated for their anticancer activities against the human breast MDA-MB-231 cell lines with the MTT and flow cytometry assays to investigate the efficacy of these synthesized compounds in the induction of apoptosis and death in breast cancer cells. The investigated complexes significantly indicated more anticancer activity in compare with the free ligand and the among them Cu(II) complex showed considerable and higher activity against the tested cell lines. The molecular docking was carried out to explore the binding modes of these compounds on DNA and epidermal growth factor receptor precursor (EGFR) proteins. Altogether, in silico and in vitro investigations indicated that coordination of chelating organic ligand to the metal centers promoted the compounds anticancer activity and these complexes may be used the likely candidate for further development in cancer treatment after complementary preclinical evaluations.








Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Lukong KE (2017) Understanding breast cancer–the long and winding road. BBA Clin 7:64–77
Parkin DM, Fernández LM (2006) Use of statistics to assess the global burden of breast cancer. Breast J 12:S70–S80
Carlson RW, Allred DC, Anderson BO, Burstein HJ, Carter WB, Edge SB, Erban JK, Farrar WB, Goldstein LJ, Gradishar WJ (2009) Breast cancer. J Natl Compr Canc Netw 7(2):122–192
Hassan M, Ansari J, Spooner D, Hussain S (2010) Chemotherapy for breast cancer. Oncol Rep 24(5):1121–1131
Anampa J, Makower D, Sparano JA (2015) Progress in adjuvant chemotherapy for breast cancer: an overview. BMC Med 13(1):1–13
Jones LW, Haykowsky MJ, Swartz JJ, Douglas PS, Mackey JR (2007) Early breast cancer therapy and cardiovascular injury. J Am Coll Cardiol 50(15):1435–1441
Oun R, Moussa YE, Wheate NJ (2018) The side effects of platinum-based chemotherapy drugs: a review for chemists. Dalton Trans 47(19):6645–6653
Kelland L (2007) The resurgence of platinum-based cancer chemotherapy. Nat Rev Cancer 7(8):573–584
Mocniak KA, Kubajewska I, Spillane DE, Williams GR, Morris RE (2015) Incorporation of cisplatin into the metal–organic frameworks UiO66-NH 2 and UiO66–encapsulation vs conjugation. RSC Adv 5(102):83648–83656
Lokich J (2001) What is the “best” platinum: cisplatin, carboplatin, or oxaliplatin? Cancer Invest 19(7):756–760
Deng ZJ, Morton SW, Ben-Akiva E, Dreaden EC, Shopsowitz KE, Hammond PT (2013) Layer-by-layer nanoparticles for systemic codelivery of an anticancer drug and siRNA for potential triple-negative breast cancer treatment. ACS Nano 7(11):9571–9584
Biersack B, Ahmad A, Sarkar H, Schobert FR (2012) Coinage metal complexes against breast cancer. Curr Med Chem 19(23):3949–3956
Vojtek M, Marques MP, Ferreira IM, Mota-Filipe H, Diniz C (2019) Anticancer activity of palladium-based complexes against triple-negative breast cancer. Drug Discov Today 24(4):1044–1058
Bruijnincx PC, Sadler PJ (2008) New trends for metal complexes with anticancer activity. Curr Opin Chem Biol 12(2):197–206
Abdelrahman S, Alghrably M, Campagna M, Hauser CAE, Jaremko M, Lachowicz JI (2021) Metal complex formation and anticancer activity of Cu (I) and Cu (II) complexes with metformin. Molecules 26(16):4730
Banerjee S, Chakravarty AR (2015) Metal complexes of curcumin for cellular imaging, targeting, and photoinduced anticancer activity. Acc Chem Res 48(7):2075–2083
Palanimuthu D, Shinde SV, Somasundaram K, Samuelson AG (2013) In vitro and in vivo anticancer activity of copper bis (thiosemicarbazone) complexes. J Med Chem 56(3):722–734
Tardito S, Marchio L (2009) Copper compounds in anticancer strategies. Curr Med Chem 16(11):1325–1348
Adımcılar V, Çeşme M, Şenel P, Danış İ, Ünal D, Gölcü A (2021) Comparative study of cytotoxic activities, DNA binding and molecular docking interactions of anticancer agent epirubicin and its novel copper complex. J Mol Struct 1232:130072
Munteanu CR, Suntharalingam K (2015) Advances in cobalt complexes as anticancer agents. Dalton Trans 44(31):13796–13808
Ott I, Schmidt K, Kircher B, Schumacher P, Wiglenda T, Gust R (2005) Antitumor-active cobalt−alkyne complexes derived from acetylsalicylic acid: studies on the mode of drug action. J Med Chem 48(2):622–629
King AP, Gellineau HA, Ahn JE, MacMillan SN, Wilson JJ (2017) Bis (thiosemicarbazone) complexes of cobalt (III) synthesis, characterization, and anticancer potential. Inorg chem 56(11):6609–6623
Zou X, Shi P, Feng A, Mei M, Li Y (2021) Two metal complex derivatives of pyridine thiazole ligand: synthesis, characterization and biological activity. Transit Met Chem 46(3):263–272
Kirschner S, Wei Y-K, Francis D, Bergman JG (1966) Anticancer and potential antiviral activity of complex inorganic compounds. J Med Chem 9(3):369–372
Naim MJ, Alam O, Farah Nawaz M, Alam J, Alam P (2016) Current status of pyrazole and its biological activities. J Pharm Bioallied Sci 8(1):2
Patil SB (2020) Medicinal siganificance of pyrazole analogues: a review. J Pharm Sci Res 12(3):402–404
Khan MF, Alam MM, Verma G, Akhtar W, Akhter M, Shaquiquzzaman M (2016) The therapeutic voyage of pyrazole and its analogs: a review. Eur J Med Chem 120:170–201
Kumar V, Kaur K, Gupta GK, Sharma AK (2013) Pyrazole containing natural products: synthetic preview and biological significance. Eur J Med Chem 69:735–753
Ghorbanpour M, Soltani B, Ziegler CJ, Jamshidi-Ghaleh K (2021) Novel pyrazolate-bridged binuclear Ni (II), Cu (II) and Zn (II) complexes: synthesis, X-ray crystal structure and nonlinear optical studies. Inorg Chim Acta 514:119957
Keter FK, Kanyanda S, Lyantagaye SS, Darkwa J, Rees DJG, Meyer M (2008) In vitro evaluation of dichloro-bis (pyrazole) palladium (II) and dichloro-bis (pyrazole) platinum (II) complexes as anticancer agents. Cancer Chemother Pharmacol 63(1):127–138
David S, Perkins RS, Fronczek FR, Kasiri S, Mandal SS, Srivastava RS (2012) Synthesis, characterization, and anticancer activity of ruthenium-pyrazole complexes. J Inorg Biochem 111:33–39
Ghorbanpour M, Soltani B, Molavi O, Aghdam EM (2022) In vitro and In silico studies on the anticancer and antimicrobial activity of Cu (II), Ni (II) and Co (II) complexes with bis (pyrazolyl) borate derivative ligand. Pharm Sci 28:64–671
Farhadi S, Mahmoudi F, Dusek M, Eigner V, Kucerakova M (2017) A new inorganic–organic nanohybrid based on a copper (II) semicarbazone complex and the PMo12O403−polyanion: synthesis, characterization, crystal structure and photocatalytic activity for degradation of cationic dyes. Polyhedron 122:247–256
Mirtamizdoust B (2017) Sonochemical synthesis of nano lead (II) metal-organic coordination polymer; new precursor for the preparation of nano-materials. Ultrason Sonochem 35:263–269
Morsali A, Monfared HH, Bigdeli F, Morsali A, Mayer P (2018) Ultrasonic assisted synthesis of a new one-dimensional nanostructured Mn (II) coordination polymer derived from azide and new multi-topic nitrogen donor ligand. Ultrason Sonochem 42:376–380
Shaabani B, Khandar AA, Ramazani N, Fleck M, Mobaiyen H, Cunha-Silva L (2017) Chromium (III), manganese (II) and iron (III) complexes based on hydrazone Schiff-base and azide ligands: synthesis, crystal structure and antimicrobial activity. J Coord Chem 70(4):696–708
Sadr MH, Soltani B, Jalili A, Nejadghafar F, Kia R, Engle JT, Ziegler CJ (2012) Synthesis and characterization of copper (II) complexes incorporating pyrazolyl-derived N S-donor bidentate ligands. Transit Metal Chem 37(7):611–617
Abu Bakar A, Akhtar MN, Mohd Ali N, Yeap SK, Quah CK, Loh W-S, Alitheen NB, Zareen S, Ul-Haq Z, Shah SAA (2018) Design, synthesis and docking studies of flavokawain B type chalcones and their cytotoxic effects on MCF-7 and MDA-MB-231 cell lines. Molecules 23(3):616
Li G, Yang M, Zuo L, Wang MX (2018) MELK as a potential target to control cell proliferation in triple-negative breast cancer MDA-MB-231 cells. Oncol Lett 15(6):9934–9940
Zarei L, Asadi Z, Samolova E, Dusek M, Amirghofran Z (2020) Pyrazolate as bridging ligand in stabilization of self-assemble Cu (II) Schiff base complexes: synthesis, structural investigations, DNA/protein (BSA) binding and growth inhibitory effects on the MCF7, CT-26, MDA-MB-231 cell lines. Inorg Chim Acta 509:119674
Foley J, Nickerson NK, Nam S, Allen KT, Gilmore JL, Nephew KP, Riese II DJ (2010) EGFR signaling in breast cancer: bad to the bone. Semin Cell Dev Biol 21(9):951–960
Lo H-W, Hsu S-C, Hung M-C (2006) EGFR signaling pathway in breast cancers: from traditional signal transduction to direct nuclear translocalization. Breast Cancer Res Treat 95(3):211–218
Nakai K, Hung M-C, Yamaguchi H (2016) A perspective on anti-EGFR therapies targeting triple-negative breast cancer. Am J Cancer Res 6(8):1609
Aghdam EM, Barzegar A, Hejazi MS (2014) Evolutionary origin and conserved structural building blocks of riboswitches and ribosomal RNAs: riboswitches as probable target sites for aminoglycosides interaction. Adv Pharm Bull 4(3):225
Morris GM, Huey R, Olson AJ (2008) Using autodock for ligand-receptor docking. Curr Protoc Bioinform 24(1):8–14
Komeda S, Casini A (2012) Next-generation anticancer metallodrugs. Curr Top Med Chem 12(3):219–235
Arami S, Mahdavi M, Rashidi M-R, Yekta R, Rahnamay M, Molavi L, Hejazi M-S, Samadi N (2017) Apoptosis induction activity and molecular docking studies of survivin siRNA carried by Fe3O4-PEG-LAC-chitosan-PEI nanoparticles in MCF-7 human breast cancer cells. J Pharm Biomed Anal 142:145–154
Jafari-Gharabaghlou D, Pilehvar-Soltanahmadi Y, Dadashpour M, Mota A, Vafajouy-Jamshidi S, Faramarzi L, Rasouli S, Zarghami N (2018) Combination of metformin and phenformin synergistically inhibits proliferation and hTERT expression in human breast cancer cells. Iran J Basic Med Sci 21(11):1167
Bang H, Edwards J, Kim J, Lawler R, Reynolds K, Ryan W, Sweigart D (1992) Cobalt-59 NMR of six-coordinate cobalt (III) tetraphenylporphyrin complexes 4 the effect of phenyl ortho substituents on chemical shift, line width, and structure. J Am Chem Soc 114(8):2843–2852
Barik AK, Paul S, Butcher RJ, Kar SK (2000) Synthesis and characterization of two tris-chelate complexes of cobalt (III) with 3, 5-dimethyl-1-(N-methyl/ethyl) thiocarbamylpyrazole (HL1, HL2)—biologically important bidentate ligands with one ambidentate donor site. Polyhedron 19(28):2651–2655
Yang L, Powell DR, Houser RP (2007) Structural variation in copper (I) complexes with pyridylmethylamide ligands: structural analysis with a new four-coordinate geometry index, τ 4. Dalton Trans 9:955–964
Reineke MH, Sampson MD, Rheingold AL, Kubiak CP (2015) Synthesis and structural studies of nickel (0) tetracarbene complexes with the introduction of a new four-coordinate geometric index, τδ. Inorg Chem 54(7):3211–3217
Soltani B, Ghorbanpour M, Ziegler CJ, Ebadi-Nahari M, Mohammad-Rezaei R (2020) Nickel (II) and cobalt (II) complexes with bidentate nitrogen-sulfur donor pyrazole derivative ligands: syntheses, characterization, X-ray structure, electrochemical studies, and antibacterial activity. Polyhedron 180:114423
Acknowledgements
We are grateful to the University of Azarbaijan Shahid Madani for supporting of this research. The cytotoxicity and flow cytometry assays of the compounds were performed in the Tabriz University of Medical Sciences. Single crystal X-ray diffraction experiments were performed at the University of Akron. We thank them for their helpful collaboration.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Conceptualization was contributed by [BS, AS, MG, AM, EMA], Supervision was contributed by [BS, AS, AM, EMA],Writing—review and editing, was contributed by [BS, AS, MG, AM, EMA, APT], Formal analysis was contributed by [CJZ], Writing—original draft preparation, was contributed by [ BS, AS, MG, AM].
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Appendix
Appendix
CCDC 2,159,568 and 2,124,457 contain the supplementary crystallographic data for 1 and 2. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghorbanpour, M., Soltani, B., Shayanfar, A. et al. The anticancer impacts of N, S donor pyrazole based ligand and its Co(III) and Cu(II) complexes on breast cancer cells. Transit Met Chem 47, 311–320 (2022). https://doi.org/10.1007/s11243-022-00514-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-022-00514-7