Abstract
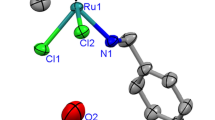
Four half-sandwich Ru(II) complexes (1)–(4) with the general formulae [Ru(η6-p-cymene)(L)Cl2] were synthesized by the reaction of one equivalent of the Ru(II) p-cymene dimer with two equivalents of a p-substituted aniline derivative L (where L is p-methyl, p-isopropyl, p-methoxy, or p-hydroxy aniline). The structures of complexes (2)–(4) were determined by single-crystal X-ray diffraction studies. The structural analysis revealed piano-stool geometry at the Ru(II) ions which are coordinated to the η6-p-cymene, two chloride anions and the amine group of the aniline ligand. In the structure of (2)–(4), the coordinated chloride ions make intermolecular hydrogen bonding with the –NH2 group of an adjacent molecules (NH–Cl) resulting in hydrogen bond networks. The catalytic activities of the complexes in transfer hydrogenation of acetophenone were studied. Complex [Ru(η6-p-cymene)(p-methylaniline)Cl2] (1) showed the best catalytic performance in the transfer hydrogenation of acetophenone. The presence and positions of methyl and bromide groups on the acetophenone have an impact on the catalytic activity in transfer hydrogenation properties of the complex (1). Moreover, catalytic activity of the complex (1) is significantly higher in the transfers hydrogenation of cyclohexanone than 2-hexanone.



Similar content being viewed by others
References
Magano J, Dunetz JR (2012) Org Process Res Dev 16:1156
Ikemoto T, Ito T, Hashimoto H, Kawarasaki T, Nishiguchi A, Mitsudera H (2000) Org Process Res Dev 4:520
Fuenfschilling PC, Hoehn P, Mutz J (2007) Org Process Res Dev 11:13
Xia H, Xu S, Hu H, An J, Li C (2018) RSC Adv 8:30875
Foubelo F, Nájera C, Yus M (2015) Tetrahedron: Asymmetry 26:769
Breuer M, Ditrich K, Habicher T, Hauer B, Keßeler M, Stürmer R, Zelinski T (2004) Angew Chem Int Ed 43:788
Stefane B, Pozgan F (2016) Top Curr Chem 374:1
Palmer MJ, Wills M (1999) Tetrahedron: Asymmetry 10:2045
Ikarıya T, Blacker AJ (2007) Acc Chem Res 40:1300
Gladiali S, Alberico E (2006) Chem Soc Rev 35:226
Wang D, Astruc D (2015) Chem Rev 115:6621
Ohkuma T, Utsumi N, Tsutsumi K, Murata K, Sandoval C, Noyori R (2006) J Am Chem Soc 1288:724
Noyori R, Hashiguchi S (1997) Acc Chem Res 30:97
Díaz-álvarez AE, Cadierno V (2013) Appl Sci 3:55
Baráth E (2018) Catalysts 8:671
Peris E, Crabtree RH (2004) Coord Chem Rev 248:2239
Hillier AC, Lee HM, Stevens ED, Nolan SP (2001) Organometallics 20:4246
Gierz V, Urbanaite A, Seyboldt A, Kunz D (2012) Organometallics 31:7532
Cheung FK, Lin C, Minissi F, Criville AL, Graham MA, Fox DJ, Wills M, Park A (2007) Org Lett 9:4659
Pelagatti P, Carcelli M, Calbiani F, Cassi C, Elviri L, Pelizzi C, Rizzotti U, Rogolino D, Generale C, Analitica C, Fisica C (2005) Organometallics 24:5836
Fern FE, Puerta MC, Valerga P (2011) Organometallics 30:5793
Dayan O, Demirmen S, Özdemir N (2015) Polyhedron 85:926
Li K, Niu J, Yang M, Li Z, Wu L, Hao X, Song M (2015) Organometallics 34:1170
Singh P, Singh AK (2010) Organometallics 29:6433
Alonso DA, Brandt P, Nordin SJM, Andersson PG, Uni V, Copenhagen D.-, March RV (1999) J Am Chem Soc 121:9580
Soriano ML, Jalón FA, Manzano BR, Maestro M (2009) Inorganica Chim Acta 362:4486
Sandoval CA, Ohkuma T, Muñiz K, Noyori R (2003) J Am Chem Soc 125(44):13490
Ohkuma T, Takeno H, Honda Y, Noyori R (2001) Adv Synth Catal 343(4):369
Ohkuma T, Ooka H, Yamakawa M, Ikariya T, Noyori R (1996) J Org Chem 61(15):4872
Kitamura M, Ohkuma T, Inoue S, Sayo N, Kumobayashi H, Akutagawa S, Ohta T, Takaya H, Noyori R (1988) J Am Chem Soc 110(2):629
Bruker (1998). APEX2 and SAINT. Bruker AXS Inc., Madison, Wisconsin, USA.
Bruker (2009). SADABS. Bruker AXS Inc., Madison, Wisconsin, USA
Sheldrick GM (2008) Acta Crystallogr Sect A Found Crystallogr 64:112
Sheldrick GM (2015) Acta Crystallogr C 71:3
Bacchi A, Loffi C, Pagano P, Pelagatti P, Scè F (2015) J Organomet Chem 778:1
Tyagi D, Binnani C, Rai RK, Dwivedi AD, Gupta K, Li PZ, Zhao Y, Singh SK (2016) Inorg Chem 55(12):6332
Otsuka T, Ishii A, Dub PA, Ikariya T (2013) J Am Chem Soc 135:9600
Dub PA, Ikariya T (2013) J Am Chem Soc 135:2604
Dub PA, Gordon JC (2018) Nat Rev Chem 2:396
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sadık, M., Karabork, M., Sahin, I. et al. Half-sandwich ruthenium(II) complexes containing 4-substituted aniline derivatives: structural characterizations and catalytic properties in transfer hydrogenation of ketones. Transit Met Chem 46, 457–464 (2021). https://doi.org/10.1007/s11243-021-00461-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-021-00461-9