Abstract
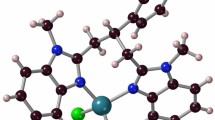
The d- and l-enantiomers of the ternary complex [Cu(phen)(ala)(H2O)]NO3 [phen = 1,10-phenanthroline; ala = alanine; l-enantiomer (1a); d-enantiomer (1b)] both undergo an aldol-type condensation with formaldehyde under mild acidic conditions to yield racemic rac-[Cu(phen)(4MeOCA)(H2O)(NO3)] (2) (4MeOCA = 4-methyl-oxazolidine-4-carboxylate). The precursors and the products were characterized by circular dichroism, FTIR, UV–Vis, fluorescence spectroscopy, molar conductivity and electrospray ionization mass spectrometry. The racemic nature of the product was established by X-ray crystallography . In the crystal structure, the copper atom has a tetragonally elongated octahedral geometry in which the water and an oxygen atom of a nitrate group are mutually trans. A possible mechanism for the racemization of the ligand is proposed.






Similar content being viewed by others
References
Teo SB, Ng CH, Horn E, Tiekink ERT (1991) Inorg Chim Acta 183:25
Teo SB, Teoh SG, Ng CH, Fun HK, Declercq JP (1995) Polyhedron 14:1447
Teo SB, Teoh SG, Ng CH, Fun HK, Declercq JP (2002) Inorg Chim Acta 340:81
Teo SB, Ng CH, Horn E, Tiekink ERT (1989) Inorg Chim Acta 163:129
Ng CH, Lin CM, Chang TM, Teoh SG, Yap SY, Ng SW (2006) Polyhedron 25:1287
Ng CH, Chong TS, Teoh SG, Adams F, Ng SW (2002) J Chem Soc, Dalton Trans 17:3361
Ng CH, Wang WS, Chong KV, Win YF, Neo KE, Lee HB, San SL, Rahman RNZRA, Leong WK (2013) Dalton Trans 42:10233
Wang WS (2016) MSc thesis, Universiti Tunku Abdul Rahman, unpublished data
Makoto C, Ng CH, Mallayan P (2015) Int J Mol Sci 16:22754 (review: DNA fibre + Cu(II) complexes, EPR)
Le X, Liao S, Liu X, Feng X (2006) J Coord Chem 59:985
Gu Q, Le X-Y, Lin Q-B, Liao S-R, Ma X-D, Feng X-L (2007) Chin J Chem 25:791
Kumar RS, Arunachalam (2007) Polyhedron 26:3255 (antimicrobial, DNA binding)
Ng CH, Kong SM, Tiong YL, Maah MJ, Sukram N, Ahmad M, Khoo SB (2014) Metallomics 6:892 (antitumor)
Ramakrishnan S, Rajendiran V, Palaniandavar M, Periasamy VS, Srinag BS, Krishnamurthy H, Akbarsha MA (2009) Inorg Chem 48:1309 (anticancer)
Alemón-Medina R, Breña-Valle M, Muñoz-Sánchez JL, Gracia-Mora MI, Ruiz-Azuara L (2007) Cancer Chemother Pharmacol 60:219 (anticancer)
Seng HL, Wang WS, Kong SM, Ong HKA, Win YF, Rahman RNZRA, Chikira M, Leong WK, Ahmad M, Khoo ASB, Ng CH (2012) Biometals 25:1061
Patra AK, Nethaji M, Chakravarty AR (2005) Dalton Trans 16:2798
Kemp S, Wheate JW, Buck DP, Nikac MJG, Collins JG, Aldrich-Wright JR (2007) J Inorg Biochem 101:1049
Arjmand F, Muddassir M, Khan RH (2010) Eur J Med Chem 45:3549
Montaňa AM, Bernal FK, Lorenzo J, Farnós JC, Batalla C, Prieto MJ, Moreno V, Avilés FX, Mesas JM, Alegre M-T (2008) Bioinorg Med Chem 16:172
Bartók M (2010) Chem Rev 110:1663
Noyori R (1990) Science 248:4960
Cao Z-Y, Brittain William DG, Fossey JS, Zhou F (2015) Catal. Sci Technol 5:3441
Bruker Advanced X-ray Solutios (2001) SMART (version 5.628). Bruker AXS Inc., Madison
Sheldrick GM (1996) SADABS. University of Göttingen, Göttingen
Bruker SHELXTL (version 2014/7) Bruker AXS Inc, Madison, Wisconsin, USA (2014)
Yousefi Y, Eshtiagh-Hosseini YH, Salimi AR, Soleimannejad J (2015) Acta Crystallogr C 71:386
Otieno T, Pinkston AT, Johnson NC, Parkin S (2002) Acta Crystallogr E58:m328
Mathews II, Manohar H (1991) Polyhedron 10:2163
Langer V, Gyepesová D, Kohútová M, Valent A (2009) Acta Crystallogr C65:m208
Langer V, Mach P, Gyepesová D, Andrezálová L, Kohútová M (2012) Acta Crystallogr C68:m326
Teo SB, Teoh SG, Snow MR (1985) Inorg Chim Acta 107:211
Creaser II, Geue RJ, Harrowfield JM, Herlt AJ, Sargeson AM, Snow MR (1982) J Am Chem Soc 104:6016
Geue RJ, McCarthy MG, Sargeson AM, Skelton BW, White AH (1985) Inorg Chem 24:1607
Solujic L, Prelesnik B, Herak R, Milosavljevic S, Nelson JH, Nikolic A (1990) Inorg Chem 29:4703
Angus PM, Elliot AJ, Sargeson AM, Willis AC (2000) J Chem Soc Dalton Trans 2931
Solujic L, Nelson JH, Fisher J (1999) Inorg Chim Acta 292:96
Phipps DA (1979) J Mol Catal 5:81
Masters PM, Friedman M (1979) J Agric Food Chem 27:507
Katsuki I, Motoda Y, Sunatsuki Y, Matsumoto N, Nakashima T, Kojima M (2002) J Am Chem Soc 124:629
Yamanari K, Hidaka J, Shimura Y (1973) Bull Chem Soc Jpn 46:3724
Armoroli N, De Cola L, Balzani V, Sauvage J-P, Dietrich-Buchecker CO, Kern JM (1992) J Chem Soc, Faraday Trans 88:553
Henry MS, Hoffman MZ (1979) J Phys Chem 83:618
Ng CH, Alan Ong HK, Kong CW, Su KK, Ng SW (2006) J Coord Chem 59:1089
Ikotun OF, Higbee EM, Quellete W, Doyle RP (2009) J Inorg Biochem 103:1254
Bolos C, Christidis PC, Will G, Wiehl L (1996) Inorg Chim Acta 248:209
Neuman NI, Franco VG, Ferroni FM, Baggio R, Passeggi MCG, Rizzi AC, Brondino CD (2012) J Phys Chem A 116:12314
Acknowledgments
This work is supported by Malaysian MOSTI eSc fund 02-02-09-SF0036 and the Malaysian Ministry of Higher Education grant FRGS/1/2012/SG01/UTAR/01/01.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ng, C.H., Wang, W.S., Win, Y.F. et al. Rac-aqua(4-methyl-oxazolidine-4-carboxylato)(nitrato)(1,10-phenanthroline)copper(II), the aldol condensation product of formaldehyde with the d- and l-(alaninato)aqua(1,10-phenanthroline)copper(II) nitrate. Transit Met Chem 41, 563–571 (2016). https://doi.org/10.1007/s11243-016-0053-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-016-0053-4