Abstract
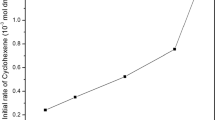
2-(2′-Pyridyl)benzimidazole (PBIMH) was functionalized onto chloromethylated polystyrene beads crosslinked with 6.5 % divinylbenzene, and this solid support was then reacted with Na2PdCl4 in methanol. The functionalized beads were then activated using sodium borohydride. The resultant polymer-supported [2-(2′-pyridyl)benzimidazole]palladium complex (PSDVB–PBIM–PdCl2) and its activated form were characterized by various physicochemical techniques. XPS studies confirmed the +2 oxidation state of palladium in the supported complex. The activated complex was found to catalyse the hydrogenation of various organic substrates including olefins, nitro and Schiff base compounds. Kinetic measurements for the hydrogenation of cyclopentene, cyclohexene and cyclooctene were carried out by varying temperature, catalyst and substrate concentration. The energy and entropy of activation were evaluated from the kinetic data. The catalyst showed an excellent recycling efficiency over six cycles without leaching of metal from the polymer support, whereas the unsupported complex was unstable as metal leached out into the solution during the first run.










Similar content being viewed by others
References
Chepaikin EG, Khidekel ML (1978) J Mol Catal A Chem 4:103–112
Zofia MM, Ostaszewski B, Zientarska J, Sobczak JW (1998) J Mol Catal A Chem 129:207–218
Cornils B, Herrmann WA (1996) Applied homogeneous catalysis with organometallic compounds. Wiley-VCH, Weinheim
Hartley FR (1985) Supported metal complexes–a new generation of catalysts, I edn. Academic Press, Dordercht Holland
Alexander S, UdayKumar V, Nagaraju N, Gayathri V (2010) Trans Met Chem 35:247–251
Drelinkiewicz A, Knapik A, Waksmundzka GA, Bukowska A, Bukowski W, Noworol J (2008) React Funct Polym 68:1059–1071
Hsiao MC, Liu ST (2010) Catal Lett 139:61–66
McGrath MP, Erik DS, Samuel JT (1995) Chem Rev 95:381–398
Xi Xiangli, Liu Yingliang, Shi Jun, Cao Shaokui (2003) J Mol Catal A Chem 192:1–7
Selvaraj PC, Mahadevan V (1997) J Polym Sci A Polym Chem 35:105–122
Figueras Francois, Coq Bernard (2001) J Mol Catal A Chem 173:223–230
Dilip RP, Ram RN (1998) J Mol Catal A Chem 130:57–64
Wan BS, Liao SJ, Xu Y, Yu DR (1998) React Kinet Catal Lett 63:397–401
McNamara CA, Dixon MJ, Bradley M (2002) Chem Rev 102:3275–3300
Alexander S, Udayakumar V, Gayathri V (2009) J Mol Catal A Chem 314:21–27
Ghosh S, Teillout A-L, Floresyona D, de Oliveira P, Hagege A, Remita H (2015) Int J Hydrogen Energy 40:4951–4959
Zhou Y, Xiang Z, Cao D, Liu CJ (2013) Chem Commun 49:5633–5635
Anna MMD, Intini S, Romanazzi G, Rizzuti A, Leonelli C, Piccinni F, Mastrorilli P (2014) J Mol Catal A Chem 395:307–314
Sun Q, Zhu LF, Sun ZH, Meng XJ, Shou XF (2012) Sci China 55:2095–2103
Shilpa ML, Gayathri V (2013) Trans Met Chem 38:705–713
Perrin DD, Armanego WCF, Perrin DR (1966) Purification of laboratory chemicals, chap 3, I edn. Pergamon Press, New York
Roe A, Montgonery JA (1953) J Am Chem Soc 75:910–912
Jose SC, Castineiras A, Martinez EG, Parajo Y, Perez Paralle ML, Gonzalez AS, Sordo J (2005) Z Anorg Allg Chem 631:2258–2264
Shilpa ML, Gayathri V (2013) Trans Met Chem 38:53–62
Belyaev SV, Vainshtein EF, Klyuev MV (2002) Kinet Catal 43:245–248
Pomogailo AD (2004) Kinet Catal 45:61–103
Purwanto P, Deshpande RM, Chaudhari RV, Delmas H (1996) J Chem Eng Data 41:1414–1417
Alexander S, Udayakumar V, Nararaju N, Gayathri V (2010) Trans Met Chem 35:427–435
Udayakumar V, Alexander S, Gayathri V, Shivakumaraiah, Patil KR, Vishwanathan B (2010) J Mol Catal A Chem 317:111–117
Udayakumar V, Alexander S, Gayathri V, Shivakumaraiah, Vishwathan B (2011) Chin J Catal 32:280–285
Welton T (1999) Chem Rev 99:2071–2083
Jainamma M, Mahadevan V (1990) J Mol Catal 60:189–207
Acknowledgments
Authors wish to thank UGC, New Delhi, India, for Major Research Project [F No. 39-741/2010(SR)]. Department of Chemistry, Bangalore University, Bangalore, for instrumentation facilities and Thermax Ltd. for providing PSDVB are gratefully acknowledged. Authors gratefully thank Dr. M. S. Hedge for XPS studies. Authors also thank Prof. Puttaswamy, Dept. of Chemistry, Bangalore University, Bangalore, for his kind help in deriving reaction mechanism and rate law equation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shilpa, M.L., Gayathri, V. Reduction of olefins, nitroarenes and Schiff base compounds by a polymer-supported [2-(2′-pyridyl)benzimidazole]palladium complex. Transition Met Chem 41, 393–401 (2016). https://doi.org/10.1007/s11243-016-0035-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-016-0035-6