Abstract
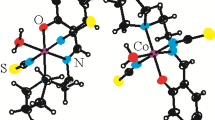
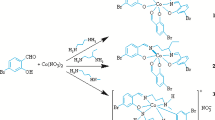
New complexes of cobalt(III) with the tridentate and tetradentate Schiff base ligands: 3-methoxy-2-{(Z)[(2-hydroxyphenyl)imino]methyl}phenol (H2L1), 4-[(2-hydroxyphenyl)imino]-2-pentanone (H2L2); and 2-((E)-1-(2-((E)-1-(2-hydroxy-4,5-dimethylphenyl)ethylideneamino)ethylimino)ethyl)-4,5 dimethylphenol (H2L3), namely [CoIII(L1)(N-MeIm)3]PF6 (1), [CoIII(L1)(py)3]ClO4 (2), [Co(L1)(py)3][Co(L1)2] (3) and [CoIII(L2)(N-MeIm)3]PF6 (4) and [Co(L3)(N-MeIm)2]PF6 (5), were synthesized and characterized by physico-chemical and spectroscopic methods. The crystal structures of the complexes were determined by X-ray crystallography. In each of these complexes, the cobalt(III) centre has a slightly distorted octahedral environment, utilizing all available coordination centres of the ligands. The complexes were also screened for in vitro antibacterial activities against four human pathogenic bacteria, and their minimum inhibitory concentrations indicated good antibacterial activities.








Similar content being viewed by others
References
Saghatforoush LA, Chalabian F, Aminkhani A, Karimnezhad G, Ershad S (2009) Eur J Med Chem 44:4490–4495
Munisamy T, Gipson SL, Franken A (2010) Inorg Chim Acta 363:20–24
Jouad EM, Larcher G, Allain M, Riou A, Bouet GM, Khan MA, Thanh XD (2001) J Inog Biochem 86:565–571
Chen D, Martell AE (1987) Inorg Chem 26:1026–1030
Nayab S, Park W, Woo HY, Sung IK, Hwang WS, Lee H (2012) Polyhedron 42:102–109
Bottche A, Takeuchi T, Hardcastle KI, Meade TJ, Gray HB (1997) Inorg Chem 36:2498–2504
Akitsu T, Itoh T (2010) Polyhedron 29:477–487
Akitsu T (2007) Polyhedron 26:2527–2535
Romanowski G, Kira J, Wera M (2014) Polyhedron 67:529–539
Hirota S, Kosugi E, Marzilli LG, Yamauchi O (1998) Inorg Chim Acta 275–276:90–97
Rajasekar M, Sreedaran S, Prabu R, Narayanan V, Jegadeesh R, Raaman N, Rahiman AK (2010) J Coord Chem 63:136–146
Abdallah SM, Mohamed GG, Zayed MA, Abou El-Ela MS (2009) Spectrochim Acta A 73:833–840
Dhanaraj CJ, Nair MS (2009) J Coord Chem 62:4018–4028
Bottcher A, Takeuchi T, Hardcastle KI, Meade TJ, Gray HB (1997) Inorg Chem 36:2498–2504
Zhu Y, Li WH (2010) Transition Met Chem 35:745–749
Fleck M, Karmakar D, Ghosh M, Ghosh A, Saha R, Bandyopadhyay D (2012) Polyhedron 34:157–162
Salehi M, Dutkiewicz G, Kubicki M (2010) Acta Crystallogr Sect E 66:1590–1591
Amirnasr M, Langer V, Rasouli N, Salehi M, Meghdadi S (2005) Can J Chem 83:2073–2081
Schenk KJ, Meghdadi S, Amirnasr M, Habibi MH, Amiri A, Salehi M, Kashi A (2007) Polyhedron 26:5448–5457
Salehi M, Amirnasr M, Mereiter K (2010) J Iran Chem Soc 7:740–751
Zhang QL, Zhu BX (2008) J Coord Chem 61:2340–2346
Oxford Diffraction, CRYSALIS PRO, Version 1.171.33.36d, Oxford Diffraction Ltd., Yarn ton, Oxford shire (2009)
Altomare A, Cascarano G, Giacovazzo C, Gualardi A (1993) J Appl Crystallogr 26:343–350
Sheldrick GM (2008) Acta Crystallogr Sect A 64:112–122
Bauer AW, Kirby WM, Csheris J, Turck M (1966) Am J Clin Pathol 45:493–496
E. Goldman, L. H. Green. Practical handbook of microbiology, 2nd edn. CRC Press, Taylor & Francis Group, New York, (2009) pp 37–39
EUCAST Definitive Document E.DEF 3.1, June 2000: Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by agar dilution, (2000) Clin Microbiol Infect 6:509-515
Yu G-M, Yang X-Y, Wang Y, Xiao Y-J, Li Y-H (2011) Acta Crystallogr Sect E 67:m329
Nejo AA, Kolawole GA, Nejo AO (2010) J Coord Chem 63:4398–4410
Mishra A, Kaushik NK, Verma AK, Gupta R (2008) Eur J Med Chem 43:2189–2196
Manjunatha M, Naik VH, Kulkarnib AD, Patilb SA (2011) J Coord Chem 64:4264–4275
Hasnain S, Zulfequar M, Nishat N (2011) J Coord Chem 64:952–964
Raman N, Pothiraj K, Baskaran T (2011) J Coord Chem 64:4286–4300
Acknowledgments
We thank Semnan University for supporting this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hasanzadeh, M., Salehi, M., Kubicki, M. et al. Synthesis, crystal structures, spectroscopic studies and antibacterial properties of a series of mononuclear cobalt(III) Schiff base complexes. Transition Met Chem 39, 623–632 (2014). https://doi.org/10.1007/s11243-014-9841-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-014-9841-x