Abstract
There has been great interest in metal–borane complexes following the discovery of metallaboratrane complexes in 1999. These complexes contain unusual Z-type (σ-acceptor) coordination of borane functional groups and have been shown to provide unexpected coordination geometries and properties for a transition metal in a particular oxidation state. Additionally, such ligands exhibit unusually high trans influences only in certain geometries. Herein, a simple qualitative description, based on crystal field theory arguments, is presented for the explanation of the geometries and atypical trans labilisations observed.
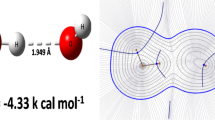
Graphical Abstract
Trans labilisation: This discussion highlights the geometries observed within metal–borane complexes and discusses the origin of the labilisation of ligands in the site trans to boron.














Similar content being viewed by others
References
Hill AF, Owen GR, White AJP, Williams DJ (1999) Angew Chem Int Ed 38:2759
St. Foreman MRJ, Hill AF, Owen GR, White AJP, Williams DJ (2003) Organometallics 22:4446
St. Foreman MRJ, Hill AF, White AJP, Williams DJ (2004) Organometallics 23:913
Crossley IR, Hill AF (2004) Organometallics 23:5656
Crossley IR, St. Foreman MRJ, Hill AF, White AJP, Williams DJ (2005) Chem Commun 221
Crossley IR, Hill AF, Willis AC (2005) Organometallics 24:1062
Crossley IR, Hill AF, Humphrey ER, Willis AC (2005) Organometallics 24:4083
Crossley IR, Hill AF, Willis AC (2006) Organometallics 25:289
Crossley IR, Hill AF, Willis AC (2007) Organometallics 26:3891
Crossley IR, Hill AF, Willis AC (2008) Dalton Trans 201
Crossley IR, Hill AF, Willis AC (2008) Organometallics 27:312
Crossley R, St. Foreman MRJ, Hill AF, Owen GR, White AJP, Williams DJ, Willis AC (2008) Organometallics 27:381
Figueroa JS, Melnick JG, Parkin G (2006) Inorg Chem 45:7056
Pang K, Quan SM, Parkin G (2006) Chem Commun 5015
Mihalcik DJ, White JL, Tanski JM, Zakharov LN, Yap GPA, Incarvito CD, Rheingold AL, Rabinovich D (2004) Dalton Trans 1626
Blagg RJ, Charmant JPH, Connelly NG, Haddow MF, Orpen AG (2006) Chem Commun 2350
Senda S, Ohki Y, Hirayama T, Toda D, Chen J-L, Matsumoto T, Kawaguchi H, Tatsumi K (2006) Inorg Chem 45:9914
Pang K, Tanski JM, Parkin G (2008) Chem Commun 1008
Landry VK, Melnick JG, Buccella D, Pang K, Ulichny JC, Parkin G (2006) Inorg Chem 45:2588
Rudolf GC, Hamilton A, Orpen AG, Owen GR (2009) Chem Commun 553
Owen GR, Gould PH, Charmant JPH, Hamilton A, Saithong S (2010) Dalton Trans B911651F
Bontemps S, Gornitzka H, Bouhadir G, Miqueu K, Bourissou D (2006) Angew Chem Int Ed 45:1611
Bontemps S, Bouhadik G, Miqueu K, Bourissou D (2006) J Am Chem Soc 128:12056
Sircoglou M, Bontemps S, Mercy M, Saffon N, Takahashi M, Bouhadir G, Maron L, Bourissou D (2007) Angew Chem Int Ed 46:8583
Bontemps S, Sircoglou M, Bouhadir G, Puschmann H, Howard JAK, Dyer PW, Miqueu K, Bourissou D (2008) Chem Eur J 14:731
Sircoglou M, Bouhadir G, Saffon N, Miqueu K, Bourissou D (2008) Organometallics 27:1675
Tsoureas N, Haddow MF, Hamilton A, Owen GR (2009) Chem Commun 2538
Tsoureas N, Bevis T, Butts CP, Hamilton A, Owen GR (2009) Organometallics 28:5222
Green MLH (1995) J Organomet Chem 500:127
Hill AF (2006) Organometallics 25:4741
Parkin G (2006) Organometallics 25:4744
Shriver DF (1963) J Am Chem Soc 85:3509
Parshall GW (1964) J Am Chem Soc 86:361
Johnson MP, Shriver DF (1966) J Am Chem Soc 88:301
Braunschweig H, Wagner T (1994) Chem Ber 127:1613
Braunschweig H, Wagner T (1996) Z Naturforsch B 51:1618
Braunschweig H, Kollann C (1999) Z Naturforsch B 54:839
Burlitch JM, Leonowicz ME, Peterson RB, Hughes RE (1979) Inorg Chem 18:1097
Cook KS, Piers WE, McDonald R (1999) Organometallics 18:1575
Cook KS, Piers WE, McDonald R (2001) Organometallics 20:3927
Cook KS, Piers WE, McDonald R (2002) J Am Chem Soc 124:5411
Braunschweig H, Kollann C, Rais D (2006) Angew Chem Int Ed 45:5254
Westcott SA, Marder TB, Baker RT, Harlow RL, Calabrese JC, Lam KC, Lin Z (2004) Polyhedron 23:2665
Curtis D, Lesley MJG, Norman NC, Orpen AG, Starbuck J (1999) J Chem Soc Dalton Trans 1687
Braunschweig H, Radacki K, Rais D, Whittell GR (2005) Angew Chem Int Ed 44:1192
Fehlner TP (2005) Angew Chem Int Ed 44:2056
Zhu J, Lin Z (2005) Inorg Chem 44:9384
Braunschweig H (1998) Angew Chem Int Ed 37:1786
Irvine GI, Lesley MJG, Marder TB, Norman NC, Rice CR, Robins EG, Roper WR, Whittell GR, Wright LJ (1998) Chem Rev 98:2685
Smith MR III (1999) Prog Inorg Chem 48:505
Braunschweig H, Colling M (2001) Coord Chem Rev 223:1
Aldrich S, Coombs DL (2004) Coord Chem Rev 248:535
Cambridge Structural Database (November 2009) The longest Rh-PMe3 bond distance with the exception of compounds containing a bridging coordination mode
Kuzu I, Krummenacher I, Armbruster F, Breher F (2008) Dalton Trans 5836
Fontaine F-G, Boudreau J, Thibault M-H (2008) Eur J Inorg Chem 5439
Jörgensen CK (1962) Absorption spectra and chemical bonding in complexes. Pergamon, Oxford
Jörgensen CK (1963) Orbitals in atoms and molecules. Academic Press, London
Pauling L (1939) The nature of the chemical bond. Cornell University Press, Ithaca
Pritchard HO, Skinner HA (1955) Chem Rev 55:745
Tsoureas N, Owen GR, Hamilton A, Orpen AG (2008) Dalton Trans 6039
Acknowledgements
The author acknowledges the support from the Ramsay Trust and Royal Society for the provision of British Centenary Ramsay Memorial and Dorothy Hodgkin Research Fellowships.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Owen, G.R. Crystal field arguments to explain the trans labilisation within transition metal–borane complexes. Transition Met Chem 35, 221–228 (2010). https://doi.org/10.1007/s11243-009-9317-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-009-9317-6