Abstract
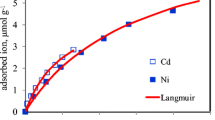
We study the mobility and interaction under competing conditions observed for copper (\(\text{ Cu}^{2+}\)) and zinc (\(\text{ Zn}^{2+}\)) ions in the context of laboratory-scale experiments performed in natural soil columns. The experiments focus on the analysis of solute breakthrough curves (BTCs) obtained after injection of an aqueous solution containing similar concentrations of the two metal ions into a soil column fully saturated with double deionized water. Transport of the competing ions is tested for the same soil under aerobic and anaerobic conditions. Measurements show that the species with lower affinity for the soil, \(\text{ Zn}^{2+}\), migrates occupying all available adsorption sites, and is then progressively replaced by the ion with higher affinity, \(\text{ Cu}^{2+}\). The two ions are displaced in the system with different effective retardation. The slowest species replaces the sorbed ions, resulting in observed \(\text{ Zn}^{2+}\) concentrations that display a non-monotonic behavior in time and which, for a certain period, are larger than the concentration supplied continuously at the inlet. In the absence of a complete geochemical characterization of the system, we show that the measured concentrations of both metals can be interpreted through simple models based on a set of coupled partial differential and algebraic equations, involving a small subset of aqueous and adsorbed species that are present in the system. Depending on the model considered, the relationship between aqueous and adsorbed ion concentrations is described at equilibrium by a Gaines–Thomas (GT) formulation, a competitive Sheindorf–Rebhun–Sheintuch (SRS) isotherm, or an Extended Langmuir (EL) isotherm, respectively. The GT formulation provides the best interpretation of the observed behavior among the models tested. We find that employing these simple models, which account only for the main governing reactive processes, allows reasonable estimation of the observed BTCs in experiments where only partial geochemical datasets are available.






Similar content being viewed by others
Abbreviations
- GT:
-
Gaines–Thomas
- SRS:
-
Sheindorf–Rebhun–Sheintuch
- EL:
-
Extended Langmuir
- ICP-MS:
-
inductively coupled plasma mass spectrometry
- BTC:
-
breakthrough curve
- ADE:
-
advection–dispersion equation
References
Altmann, R.S., Bourg, A.C.M.: Cadmium mobilization under conditions simulating anaerobic to aerobic transition in landfill leachate-polluted aquifer. Water Air Soil Pollut. 94, 385–392 (1997)
Antoniadis, V.A., McKinley, J.D., Zuhairi, W.Y.W.: Single-element and competitive metal mobility measured with column infiltration and batch tests. J. Environ. Qual. 36, 53–60 (2007)
Appelo, C.A.J., Postma, D.: A consistent model for surface complexation on birnessite (\(\text{ delta-MnO}_{2}\)) and its application to a column experiment. Geochim. Cosmochim. Acta 64(22), 3931–3931 (1999)
Atia, A.A., Donia, A.M., Elwakeel, K.Z.: Adsorption behaviour of non-transition metal ions on a synthetic chelating resin bearing iminoacetate functions. Sep. Purif. Technol. 43, 43–48 (2005)
Barry, D.A., Starr, J.L., Parlange, J.-Y., Braddock, R.D.: Numerical analysis of the snow-plow effect. Soil Sci. Soc. Am. J. 47(5), 862–868 (1983)
Barrow, N.J., Cartes, P., Mora, M.L.: Modification to the Freundlich equation to describe anion sorption over a large range and to describe competition between pairs of ions. Eur. J. Soil Sci. 56, 601–606 (2005)
Barrow, N.J.: The description of sorption curves. Eur. J. Soil Sci. 59, 900–910 (2008). doi:10.1111/j.1365-2389.2008.01041.x
Bianchi, A., Petrangeli Papini, M., Corsi, A., Behra, P., Beccari, M.: Competitive transport of cadmium and lead through a natural porous medium: influence of the solid/liquid interface processes. Water Sci. Technol. 48, 9–16 (2003)
Bianchi Janetti, E., Dror, I., Riva, M., Guadagnini, A., Berkowitz, B.: Estimation of single-metal and competitive sorption isotherms through maximum likelihood and model quality criteria. Soil Sci. Soc. Am. J. 76(4), 1229–1245 (2012)
Brooks, S.C., Taylor, D.L., Jardine, P.M.: Reactive transport of EDTA-complexed cobalt in the presence of ferrihydrite. Geochim. Cosmochim. Acta 60(11), 1899–1908 (1996)
Cornu, J.Y., Denaix, L., Schneider, A., Pellerin, S.: Temporal evolution of redox processes and free Cd dynamics in a metal-contaminated soil after rewetting. Chemosphere 70, 306–314 (2007)
Edery, Y., Guadagnini, A., Scher, H., Berkowitz, B.: Reactive transport in disordered media: Role of fluctuations in interpretation of laboratory experiments. Adv. Water Resour. (2012). doi:10.1016/j.advwatres.2011.12.008 (in press)
Freundlich, H.M.F.: Over the sorption in solution. J. Phys. Chem. 57, 385–470 (1906)
Figueira, M.M., Volesky, B., Azarian, K., Ciminelli, V.S.T.: Biosorption column performance with a metal mixture. Environ. Sci. Technol. 34, 4320–4326 (2000)
Fontes, M.P.F., Gomes, P.C.: Simultaneous competitive adsorption of heavy metals by the mineral matrix of tropical soils. Appl. Geochem. 18, 795–804 (2003)
Gaines, G., Thomas, H.: Adsorption studies on clay minerals, ii, a formulation of the thermodynamics of exchange adsorption. J. Chem. Phys. 21, 714–718 (1953)
Gu, B.H., Wu, W.M., Ginder-Vogel, M.A., Yan, H., Fields, M.W., Zhou, J., Fendorf, S., Criddle, C.S., Jardine, P.M.: Bioreduction of uranium in a contaminated soil column. Environ. Sci. Technol. 39(13), 4841–4847 (2005)
Harter, D., Naidu, R.: An assessment of environmental and solution parameter impact on trace-metal sorption by soils. Soil Sci. Soc. Am. J. 65, 597–612 (2001)
Hawari, A.H., Mulligan, C.N.: Effect of the presence of lead on the biosorption of copper, cadmium and nickel by anaerobic biomass. Process Biochem. 42, 1546–1552 (2007)
Hinz, C., Selim, H.M.: Transport of Zn and Cd in soils—experimental evidence and modeling approaches. Soil Sci. Soc. Am. J. 58(5), 1316–1327 (1994)
Inglett, P.W., Reddy, K.R., Corstanje, R.: Anaerobic soils. In: Hillel, D. (ed.) Encyclopedia of Soils in the Environment, pp. 71–78. Academic Press, New York (2005)
Jakob, A., Pfingsten, W., Van Loon, L.: Effects of sorption competition on caesium diffusion through compacted argillaceous rock. Geochim. Cosmochim. Acta 73(9), 2441–2456 (2009)
Kerner, M., Wallmann, K.: Remobilization events involving Cd and Zn from intertidal flat sediments in the Elbe estuary during the tidal cycle. Estuarine Coastal Shelf Sci. 35, 371–393 (1992)
Kittrick, J.A., Lee, F.Y.: Electron microprobe analysis of elements associated with zinc and copper in an oxidizing and an anaerobic soil environment. Soil Sci. Soc. Am. J. 48, 548–554 (1984)
Kratochvil, D., Volesky, B.: Advances in the biosorption of heavy metals. Trends Biotechnol. 16, 291–300 (1998)
Lack, J.G., Chaunduri, S.K., Kelly, S.D., Kemner, K.M., O’Connor, S.M., Coates, J.D.: Immobilization of radionuclides and heavy metals through anaerobic bio-oxidation of Fe(II). App. Environ. Microbiol. 68, 2704–2710 (2002)
Langmuir, I.: The sorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 40, 1361–1403 (1918)
Liao, L., Selim, H.M.: Competitive sorption of nickel and cadmium in different soils. Soil Sci. 174, 549–555 (2009)
Malandrino, M., Abollino, O., Giacomino, A., Aceto, M., Maentasti, E.: Adsorption of heavy metals on vermiculite: influence of pH and organic ligands. J. Colloid Interface Sci. 299, 537–546 (2006)
MATLAB 2011: MATLAB Version 2011a. The MathWorks Inc., Natick (2011)
Mao, X., Prommer, H., Barry, D.A., Langevin, C.D., Panteleit, B., Li, L.: Three-dimensional model for multi-component reactive transport with variable density groundwater flow. Environ. Modell. Softw. 21(5), 615–628 (2006)
McBride, M.B.: Environmental Chemistry of Soils. Oxford University Press, New York (1994)
McCormick, P.V., Cairns, J.: Algae as indicators of environmental change. J. Appl. Phycol. 6(5–6), 509–526 (1994). doi:10.1007/BF02182405
Naja, G., Volesky, B.: Multi-metal biosorption in a fixed-bed flow-through column. Colloids Surf. A 281, 194–201 (2006)
Nitzsche, O., Meinrath, G., Merkel, B.: Database uncertainty as a limiting factor in reactive transport prognosis. J. Contam. Hydrol. 44, 223–237 (2000)
Patrick Jr, W.H., Khalid, R.D.: Phosphate release and sorption by soils and sediments: effect of aerobic and anaerobic conditions. Science 186, 53–55 (1974)
Porta, G., Riva, M., Guadagnini, A.: Upscaling solute transport in porous media in the presence of an irreversible bimolecular reaction. Adv. Water Resour. 35, 151–162 (2012). doi:10.1016/j.advwatres.2011.09.004
Rubin, S., Dror, I., Berkowitz, B.: Experimental and modeling analysis of coupled non-Fickian transport and sorption in natural soils. J. Contam. Hydrol. 132, 28–36 (2012). doi:10.1016/j.jconhyd.2012.02.005
Saha, U.K., Taniguchi, S., Sakurai, K.: Simultaneous adsorption of cadmium, zinc, and lead on hydroxyaluminum- and hydroxyaluminosilicate-montmorillonite complexes. Soil Sci. Soc. Am. J. 66, 117–128 (2002)
Sanchez-Vila, X., Bolster, D.: An analytical approach to transient homovalent cation exchange problems. J. Hydrol. 378, 281–289 (2009)
Sanchez-Vila, X., Fernandez, D., Guadagnini, A.: Interpretation of column experiments of transport of solutes undergoing an irreversible bimolecular reaction using a continuum approximation. Water Resour. Res. 46, W12510 (2010). doi:10.1029/2010WR009539
Selim, H.M., Buchter, B., Hinz, C., Ma, L.: Modeling the transport and retention of Cd in soils: multireaction and multicomponent approaches. Soil Sci. Soc. Am. J. 56(4), 1004–1015 (1992)
Selim, H.M., Sparks, D.L.: Heavy Metals Release in Soils. CRC Press, Boca Raton (2001)
Seo, D.C., Yu, K., DeLaune, R.D.: Comparison of monometal and multimetal adsorption in Mississippi River alluvial wetland sediment: batch and column experiments. Chemosphere 73, 1757–1764 (2008)
Seuntjens, P., Tirez, K., Simunek, J., van Genuchten, MTh, Cornelis, C., Geuzens, P.: Aging effects on cadmium transport in undisturbed contaminated sandy soil columns. J. Environ. Qual. 30, 1040–1050 (2001)
Sheindorf, C., Rebhun, M., Sheintuch, M.: A Freundlich-type multicomponent isotherm. J. Colloid Interface Sci. 79, 136–141 (1981)
Sparks, D.L.: Environmental Soil Chemistry. Academic Press, San Diego (1995)
Srivastava, P., Singh, B., Angove, M.: Competitive adsorption behavior of heavy metals on kaolinite. J. Colloid Interface Sci. 290, 28–38 (2005)
Starr, J.L., Parlange, J.-Y.: Dispersion in soil columns—snow plow effect. Soil Sci. Soc. Am. J. 43(3), 448–450 (1979)
Teuchies, J., Beauchard, O., Jacobs, S., Meire, P.: Evolution of sediment metal concentrations in a tidal marsh restoration project. Sci. Total Environ. 419, 187–195 (2012). doi:10.1016/j.scitotenv.2012.01.016
Tsang, D.C.W., Lo, I.M.C.: Competitive Cu and Cd sorption and transport in soils: a combined batch kinetics, column, and sequential extraction study. Environ. Sci. Technol. 40, 6655–6661 (2006)
Tyler, L.D., McBride, M.B.: Mobility and extractability of cadmium, copper, nickel, and zinc in organic and mineral soil columns. Soil Sci. 134, 198–205 (1982)
Valocchi, A., Street, R., Roberts, P.: Transport of ion-exchanging solutes in groundwater: chromatographic theory and field simulation. Water Resour. Res. 17(5), 1517–1527 (1981)
Voegelin, A., Vulava, V.M., Kretzschmar, R.: Reaction-based model describing competitive sorption and transport of Cd, Zn, and Ni in an acidic soil. Environ. Sci. Technol. 35, 1651–1657 (2001)
Voegelin, A., Vulava, V.M., Kuhnen, F., Kretzschmar, R.: Multicomponent transport of major cations predicted from binary adsorption experiments. J. Contam. Hydrol. 46, 319–338 (2000)
Vulava, V., Kretzschmar, R., Rusch, U., Grolimund, D., Westall, J., Borkovec, M.: Cation competition in a natural subsurface material: modelling of sorption equilibria. Environ. Sci. Technol. 34, 2149–2155 (2000)
Williams, J.D.H., Syers, J.K., Shulka, S.S., Harris, R.F.: Levels of inorganic and total phosphorus in lake sediments as related to other sediment parameters. Environ. Sci. Technol. 5, 1113–1120 (1971)
Yang, R.T.: Gas separation by sorption processes. Butterworths, Boston (1987)
Yaron, B., Dror, I., Berkowitz, B.: Soil-Subsurface Change: Chemical Pollutant Impacts. Springer, Heidelberg (2012)
Zhang, H., Selim, H.M.: Modeling competitive arsenate-phosphate retention and transport in soils: a multi-component multi-reaction approach. Soil Sci. Soc. Am. J. 71(4), 1267–1277 (2007)
Acknowledgments
X. S. acknowledges financial support by the Spanish Ministry of Science and Innovation through the projects Consolider-Ingenio 2010 (ref. CSD2009-00065) and RARA-AVIS (ref. GCL2009-1114). B. B. acknowledges financial support from the Israel Science Foundation (Grant No. 221/11). M. R., A. G., and E. B. J. acknowledge financial support from the Politecnico di Milano (Project GEMINO, Progetti di ricerca 5 per mille junior).
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1
Each experiment was performed in duplicate to ensure reproducibility. Each measurement was repeated six times to allow error quantification. Figure 7 shows the two replicates illustrating the degree of reproducibility of the experiment. The total experimental and analysis error is represented in terms of error bars computed on the basis of the performed replicates.
Time dependence of concentration of metal ions eluted from the soil column under aerobic condition. The constant inlet concentrations, \(C_{0,\mathrm{Cu}} = 1.55\, \text{ mmol} \text{ L}^{-1},\, C_{0,\mathrm{Zn}} = 1.77\, \text{ mmol} \text{ L}^{-1}\), are shown for reference (\(dotted\) \(lines\)). \(Error\) \(bars\) corresponding to \(\pm 2\sigma \) are reported, \(\sigma \) being the estimated standard deviation of the performed measurement replicates. The two replicates of the same experiment are indicated as I and II
Appendix 2
Note that, by way of (4), Eq. (1) for \(i=3\) can be written as
Equation (12) is a classical one-dimensional ADE with constant (in space and time) retardation and can be solved independently of the other equations. It provides the space-time dependence of \(C_3\), i.e., \(C_3 (x,t)\), once a value of \(z_3\) (e.g., \(z_3 = 2\)) is assumed. We then replace \(C_3 (x,t)\) into (4) to obtain \(\beta _3 (x,t)\). This allows rewriting (2) as
Considering (1), we can introduce
The quantity \(u^{*}\) is a conservative component and satisfies a one-dimensional conservative ADE. We then define \(u=u^{*}-C_3 =C_1 +C_2\). Considering (2), (3), and (14), and noting that \(z_1 =z_2 =2\), we can write \(\beta _1\) as a function of \(C_1\) according to (for simplicity, we drop the space-time dependence from quantities)
From the last of (15) it follows that
Introducing (16)–(18) into (1) allows writing the partial differential equation governing the evolution of \(C_1\)
Equation (19) is nonlinear in \(C_1\) and is solved numerically. Note that (19) has the format of a classical ADE with a retardation and decay term (the second term on the left hand side). The retardation factor is highly non-linear. The decay term can be either positive or negative, depending on the sign of \(\partial u/\partial t\). Once \(C_1\) is known, one can calculate \(C_2\) as
Appendix 3
Rights and permissions
About this article
Cite this article
Bianchi Janetti, E., Dror, I., Riva, M. et al. Mobility and Interaction of Heavy Metals in a Natural Soil. Transp Porous Med 97, 295–315 (2013). https://doi.org/10.1007/s11242-013-0125-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11242-013-0125-2