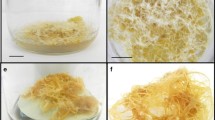
Abstract
Artemisia alba Turra is an essential oil bearing plant, with Euro-Mediterranean and Southeastern European distribution. One of the distinctive characteristics of its essential oil is the variability of its terpenoid profile due to environmental, genetic and other factors. In the present work, tissue culture experiment has provided a model system of in vitro morphogenesis alteration. Auxin and cytokinin treatments were applied alone or in different combinations, leading to the development of directly rooting and root suppressed in vitro plantlets. Direct in vitro rooting was definitely related to obtaining biomass richest in flavonoid compounds (irrespectively of the PGR combination leading to obtaining this morphotype). The directly rooting morphotype was also distinguished by the highest flavones/flavonoles ratio as compared with the in situ and the rest of the in vitro samples. Underground parts of in vitro samples were shown to be significantly richer in caffeoylquinic acids as compared with aerial parts. An elevation of DCQA/CQA ratios in PGR treated plants as compared with the non-treated control could be estimated as an indication of stimulation of the esterification process with the purpose of coping with the stress of the impairment of the physiological state of normal shoot-to-root tissue formation. It was shown that in vitro morphogenesis moderation could be applied as a simple and reproducible protocol to alter polyphenolics production in tissue cultures of this plant species.
Key message
in vitro culture system of Artemisia alba Turra allowed for its morphogenesis modification. Changes in root system development and callusogenesis were related to targeted alterations of flavones/flavonols ratios and esterification degree of caffeoylquinic acids.
AbstractSection Graphical Abstract









Similar content being viewed by others
Data availability
Datasets generated during and/or analyzed during the current study are available in Supplementary material. Any additional material is available from the corresponding author on reasonable request.
Abbreviations
- BAP:
-
Benzyl adenine
- QA:
-
Quinic acids
- CQA:
-
Caffeoylquinic acid
- 5-O-CQA:
-
5-O- Caffeoylquinic acid
- 3,5-di-O-CQA:
-
3,5-O-Dicaffeoylquinic acid
- DCQA:
-
Dicaffeoylquinic acid
- DW:
-
Dry weight
- FQA:
-
Feruloylquinic acid
- FW:
-
Fresh weight
- G5:
-
Gamborg
- IBA:
-
Indole-3-butyric acid
- MS:
-
Murashige and Skoog
- PGR:
-
Plant growth regulators
- TCQA:
-
Tricaffeoylquinic acid
- UHPLC-PDA-MS:
-
Ultrahigh performance liquid chromatography coupled to photodiode array detection and mass spectrometry
References
Abbaspour J, Ehsanpour AA, Aghaei M, Ghanadian M (2019) Sesquiterpene lactones from shoot culture of Artemisia aucheri with cytotoxicity against prostate and breast cancer cells. Res Pharm Sci 14:329–334. https://doi.org/10.4103/1735-5362.263557
Appendino G, Gariboldi P, Menichini F (1985) Oxygenated nerolidol derivatives from Artemisia alba. Phytochem 24:1729–1733. https://doi.org/10.1016/S0031-9422(00)82542-X
Ben Rejeb F, Chograni H, Ben Romdhane M, Riahi L (2023) Effects of two abiotic elicitors on secondary metabolites accumulation and bioactivity in tree wormwood in vitro shoot cultures. Nat Res Hum Health 3:189–195
Bora KS, Sharma A (2011) The Genus Artemisia: A Comprehensive Review. Pharm Biol 49:101–109. https://doi.org/10.3109/13880209.2010.497815
Coassini Lokar L, Maurich V, Mellerio G, Moneghini M, Poldini L (1987) Variation in terpene composition of Artemisia alba in relation to environmental conditions. Biochem Syst Ecol 15:327–333. https://doi.org/10.1016/0305-1978(87)90007-X
Danova K (2020) Artemisia alba Turra as a flexible model to study interrelations between developmental patterns and secondary metabolites productivity in plant cell tissue and organ culture conditions. In: Roe J (ed) Artemisia: classification, cultivation and uses. Nova Science Publishers, New York, pp 81–118
Danova K, Todorova M, Trendafilova A, Evstatieva L (2012) Cytokinin and auxin effect on the terpenoid profile of the essential oil and morphological characteristics of shoot cultures of Artemisia alba. Nat Prod Commun 7:1075–1076
Danova K, Motyka V, Todorova M, Trendafilova A, Krumova S, Dobrev P, Andreeva T, Oreshkova T, Taneva S, Evstatieva L (2018) Effect of cytokinin and auxin treatments on morphogenesis, terpenoid biosynthesis, photosystem structural organization, and endogenous isoprenoid cytokinin profile in Artemisia alba Turra in vitro. J Plant Growth Reg 37:403–418. https://doi.org/10.1007/s00344-017-9738-y
Danova K, Trendafilova A, Motyka V, Dobrev P, Ivanova V, Todorova M (2020) Therapeutic Potential and Biotechnological Utilization of the Indigenous Biosynthetic Capacity of Artemisia alba Turra: A Review. Ecol Balk SE3:257–273
Dias MI, Sousa MJ, Alves RC, Ferreira ICFR (2016) Exploring plant tissue culture to improve the production of phenolic compounds: A review. Ind Crops Prod 82:9–22. https://doi.org/10.1016/j.indcrop.2015.12.016
Dordevic S, Stanisavljevic D, Ristic M, Milenkovic M, Velickovic D, Stojicevic S, Zlatkovic B (2013) Chemical, antioxidant and antimicrobial analysis of the essential oil and extract of Artemisia alba Tura. Digest J Nanomat Biostruct 8:1377–1388
Gamborg OL, Miller RA, Ojima K (1968) Nutreint requerments of suspension culture of soybean root cells. Exp Cell Res 50:151–158. https://doi.org/10.1016/0014-4827(68)90403-5
Han JL, Wang H, Ye HC, Liu Y, Li ZQ, Zhang Y, Zhang YS, Yan F, Li GF (2005) High efficiency of genetic transformation and regeneration of Artemisia annua L. via Agrobacterium tumefaciens-mediated procedure. Plant Sci 168:73–80. https://doi.org/10.1016/j.plantsci.2004.07.020
Holobiuc I, Blindu R (2007) In Vitro Culture Introduction for Ex situ Conservation of Some Rare Plant Species. Rom J Biol - Plant Biol 51:13–23
Ivanescu B, Miron A, Corciova A (2015) Sesquiterpene Lactones from Artemisia Genus: Biological Activities and Methods of Analysis. J Anal Methods Chem 2015:247685. https://doi.org/10.1155/2015/247685
Jakovljević MR, Grujičić D, Živanović M, Stanković M, Ćirić A, Djurdjević P, Todorović Ž, Živančević-Simonović S, Mihaljević O, Milošević-Djordjević O (2019) Ethyl acetate extracts of two Artemisia species: analyses of phenolic profile and anticancer activities against SW-480 colon cancer cells. Nat Prod Commun 14:1–6. https://doi.org/10.1177/1934578X19843011
Jakovljević MR, Grujičić D, Vukajlović JT, Marković A, Milutinović M, Stanković M, Vuković N, Vukić M, Milošević-Djordjević O (2020) In vitro study of genotoxic and cytotoxic activities of methanol extracts of Artemisia vulgaris L. and Artemisia alba Turra. S Afr J Bot 132:117–126. https://doi.org/10.1016/j.sajb.2020.04.016
Janaćković P, Rajčević N, Gavrilović M, Novaković J, Giweli A, Stešević D, Marin PD (2019) Essential oil composition of five Artemisia (Compositae) species in regards to chemophenetics. Biochem Syst and Ecol 87:103960. https://doi.org/10.1016/j.bse.2019.103960
Kane NF, Kyama MC, Nganga JK, Hassanali A, Diallo M, Kimani FT (2019) Comparison of phytochemical profiles and antimalarial activities of Artemisia afra plant collected from five countries in Africa. South Afr J Bot 125:126–133. https://doi.org/10.1016/j.sajb.2019.07.001
Kapustina LA, Torrell M, Valles J (2001) Artemisia communities in arid zones of Uzbekistan (Central Asia). In: Durant McA E, Fairbanks DJ (Eds) Shrubland ecosystem genetics and biodiversity: proceedings; 2000 June 13-15; Provo, UT. Proc. RMRS-P-21. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Ogden, pp 104–112
Koleva P, Wolfram E, Pedrussio S, Raynova Y, Evstatieva L, Danova K (2015) In vitro culture development and polyphenolics production of Artemisia alba Turra. J Biosci Biotech SE/Online 131–136
Liang N, Dupuis JH, Yada RY, Kitts DD (2019) Chlorogenic acid isomers directly interact with Keap 1-Nrf2 signaling in Caco-2 cells. Mol Cell Biochem 457:105–118. https://doi.org/10.1007/s11010-019-03516-9
Maggio A, Rosselli S, Brancazio CL, Safder M, Spadaro V, Bruno M (2011) Artalbic acid, a sesquiterpene with an unusual skeleton from Artemisia alba (Asteraceae) from Sicily. Tetrahedr Lett 52:4543–4545. https://doi.org/10.1016/j.tetlet.2011.06.088
Maggio A, Rosselli S, Bruno M, Spadaro V, Raimondo FM, Senatore F (2012) Chemical composition of essential oil from Italian populations of Artemisia alba Turra (Asteraceae). Molecules 17:10232–10241. https://doi.org/10.3390/molecules170910232
Maggio A, Rosselli S, Brancazio CL, Spadaro V, Raimondo FM, Bruno M (2013) Metabolites from the aerial parts of the Sicilian population of Artemisia alba. Nat Prod Commun 8:283–286
Matkowski A (2008) Plant in vitro culture for the production of antioxidants—a review. Biotechnol Adv 26:548–560. https://doi.org/10.1016/j.biotechadv.2008.07.001
Moglia A, Lanteri S, Comino C, Hill L, Knevitt D, Cagliero C, Rubiolo P, Bornemann S, Martin C (2014) Dual catalytic activity of hydroxycinnamoyl-coenzyme A quinate transferase from tomato allows it to moonlight in the synthesis of both mono- and dicaffeoylquinic acids. Plant Physiol 166:1777–1787. https://doi.org/10.1104/pp.114.251371
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plantarum 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Olennikov DN, Chirikova NK, Kashchenko NI, Nikolaev VM, Kim SW, Vennos C (2018) Bioactive Phenolics of the Genus Artemisia (Asteraceae): HPLC-DAD-ESI-TQ-MS/MS Profile of the Siberian Species and Their Inhibitory Potential Against ɑ-Amylase and ɑ-Glucosidase. Front Pharmacol 9:756. https://doi.org/10.3389/fphar.2018.00756
Pecheva D, Danova K (2022) Light and auxin treatments affect morphogenesis and polyphenolics productivity in Artemisia alba Turra cell aggregates in vitro. BioRisk 17:213–225. https://doi.org/10.3897/biorisk.17.77452
Petrova N, Koleva P, Velikova V, Tsonev T, Andreeva T, Taneva S, Krumova S, Danova K (2018) Relations between photosynthetic performance and polyphenolics productivity of Artemisia alba Turra in in vitro tissue cultures. Int J Bioautom 22:73. https://doi.org/10.7546/ijba.2018.22.1.73-82
Pieracci Y, Vento M, Pistelli L, Lombardi T, Pistelli L (2022) Halophyte Artemisia caerulescens L.: Metabolites from In Vitro Shoots and Wild Plants. Plants 11:1081. https://doi.org/10.3390/plants11081081
Radulović N, Blagojević P (2010) Volatile profiles of Artemisia alba from contrasting serpentine and calcareous habitats. Nat Prod Commun 5:1117–1122
Ronse AC, De Pooter HL (1990) Essential Oil Production by Belgian Artemisia alba (Turra) before and after Micropropagation. J Ess Oil Res 2:237–242. https://doi.org/10.1080/10412905.1990.9697873
Stojanovic G, Palic R, Mitrovic J, Djokovic D (2000) Chemical composition and antimicrobial activity of the essential oil of Artemisia lobelii All. J Ess Oil Res 12:621–624. https://doi.org/10.1080/10412905.2000.9712172
Todorova M, Trendafilova A, Danova K, Simmons L, Wolfram E, Meier B, Riedl R, Evstatieva L (2015) Highly oxygenated sesquiterpenes in Artemisia alba Turra. Phytochem 110:140–149. https://doi.org/10.1016/j.phytochem.2014.12.008
Trendafilova A, Todorova M, Genova V, Peter S, Wolfram E, Danova K, Evstatieva L (2018) Phenolic profile of Artemisia alba Turra. Chem Biodiv 15:e1800109. https://doi.org/10.1002/cbdv.201800109
Tu Y (2016) Artemisinin - a gift from traditional Chinese medicine to the world (Nobel lecture). Angew Chem Int Ed 55:10210–10226. https://doi.org/10.1002/anie.201601967
Uleberg E, Rohloff J, Jaakola L, Trost K, Junttila O, Haggman H, Martinussen I (2012) Effects of temperature and photoperiod on yield and chemical composition of northern and southern clones of bilberry (Vaccinium myrtillus L.). J Agric Food Chem 60:10406–10414. https://doi.org/10.1021/jf302924m
Vallès J, McArthur ED (2001) Artemisia systematics and phylogeny: cytogenetic and molecular insights. In: McArthur ED, Fairbanks DJ (eds) Proceedings: shrubland ecosystem genetics and biodiversity 2000 June 13-15; Provo, UT. Proc. RMRS-P-21. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Ogden, pp 67–74
Winkel BSJ (2006) The Biosynthesis of Flavonoids. In: Grotewold E (ed) The Science of Flavonoids. Springer, New York, pp 71–95. https://doi.org/10.1007/978-0-387-28822-2_3
Zeb S, Ali A, Zaman W, Zeb S, Ali S, Ullah F, Shakoor A (2018) Pharmacology, taxonomy and phytochemistry of the genus Artemisia specifically from Pakistan: a comprehensive review. Pharm Biomed Res 4:1–12. https://doi.org/10.18502/pbr.v4i4.543
Zhen L, Chen S, Chen F, Fang W, Li J, Wang H (2010) Karyotype and meiotic analysis of five species in the genus Artemisia. Caryologia 63:382–390. https://doi.org/10.1080/00087114.2010.10589750
Acknowledgements
This research was funded by a grant of National Scientific Fund, Bulgaria, grant КП-06-H39/6.
Funding
This research was supported by the National Scientific Fund, Bulgaria (КП-06-H39-6).
Author information
Authors and Affiliations
Contributions
AT has performed methodology development for phytochemical analyses, software utilization for phytochemical data analysis and review and editing of the manuscript; MT has performed methodology for phytochemical analyses; EW has developed and performed methodology for phytochemical analyses, supervision of analytical work; SP has performed methodology for phytochemical analyses, software processing of data; VI has performed sample preparation for phytochemical analyses; KD has performed conceptualization of the experimental design and manuscript tailoring, tissue culture development, project administration. All co-authors have consented to publish our experimental results in the Plant Cell Tissue and Organ Culture Journal and have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Mohammad Faisal.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Trendafilova, A., Todorova, M., Wolfram, E. et al. Unfolding phenolics biosynthetic plasticity of Artemisia alba Turra through plant tissue culture techniques. Plant Cell Tiss Organ Cult 157, 29 (2024). https://doi.org/10.1007/s11240-024-02756-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11240-024-02756-y