Abstract
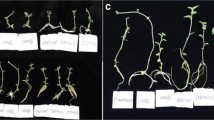
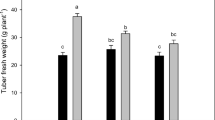
Potato is an important crop in terms of its nutritional value, cost-effective production, versatile utilization, and starch production. To produce disease-free potato starting material, the production of microtubers in vitro is a viable solution. The process of tuberization is a multifaceted physiological phenomenon that is regulated by a multitude of factors, including environmental conditions, nitrogen availability, plant growth regulators (PGRs), genetic makeup, nutrient availability, photoperiodic, temperature, explant source, potato cultivar, and sucrose. In this study, we evaluated the impact of diverse light qualities and photoperiods on in vitro microtuberzation of potato single node explant. Based on our finding, in comparison with white (W: control), the inclusion of the far-red wavelength in a red-blue light (RBF) in prolonged photoperiod significantly increased both tuberization percentage (18%) and tuberization degree (60%) in Sante® cultivar. Additionally, in comparison with W light at 16 h photoperiod, the application of RBF led to significant increment in various parameters, such as explant fresh weight (23%), microtuber diameter (40%), microtuber number (47%), and microtuber fresh weight (121%), microtuber dry weight (166%), and microtuber yield (FW and DW; 227%). Similarly, the utilization of the far-red spectrum in combination with the red-blue spectrum resulted in a 27% increase in protein content compared to W (control).
Key message
Potato in vitro microtuberzation was significantly improved when subjected to prolonged photoperiod and the application of far-red spectrum.




Similar content being viewed by others
Data Availability
The data will be available on a reasonable request.
References
Ai Y, Jing S, Cheng Z et al (2021) DNA methylation affects photoperiodic tuberization in potato (Solanum tuberosum L.) by mediating the expression of genes related to the photoperiod and GA pathways. Hortic Res 8. https://doi.org/10.1038/s41438-021-00619-7
Albalasmeh AA, Berhe AA, Ghezzehei TA (2013) A new method for rapid determination of carbohydrate and total carbon concentrations using UV spectrophotometry. Carbohydr Polym 97:253–261. https://doi.org/10.1016/j.carbpol.2013.04.072
Arregui LM, Veramendi J, Mingo-Castel AM (2003) Effect of gelling agents on in vitro tuberization of six potato cultivars. Am J Potato Res 80:141–144. https://doi.org/10.1007/BF02870213
Askari N, De Klerk GJ (2020) Elimination of epidermal wax from explants increases growth in tissue culture of lily. Sci Hortic (Amsterdam) 274:109637. https://doi.org/10.1016/j.scienta.2020.109637
Askari N, Visser RGF (2022) The role of scale explants in the growth of regenerating lily bulblets in vitro. Plant Cell Tissue Organ Cult 149:589–598. https://doi.org/10.1007/s11240-022-02328-y
Askari N, Aliniaeifard S, Visser RGF (2022) Low CO2 levels are detrimental for in vitro plantlets through disturbance of photosynthetic functionality and accumulation of reactive oxygen species. Horticulturae 8:44. https://doi.org/10.3390/horticulturae8010044
Askari N, Ghahramani R, Reisi A et al (2023) The role of thermal stress on in Vitro Potato Microtuber induction. J Veg Sci 6:73–84. https://doi.org/10.22034/IUVS.2022.562669.1236
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brini F, Mseddi K, Brestic M, Landi M (2022) Hormone-mediated plant responses to light quality and quantity. Environ Exp Bot 202
Chen L, Xue X, Yang Y et al (2018) Effects of red and blue LEDs on in vitro growth and microtuberization of potato single-node cuttings. Front Agric Sci Eng 5:197–205. https://doi.org/10.15302/J-FASE-2018224
Chen L, li ZHANGK, GONG X, chen et al (2020) Effects of different LEDs light spectrum on the growth, leaf anatomy, and chloroplast ultrastructure of potato plantlets in vitro and minituber production after transplanting in the greenhouse. J Integr Agric 19:108–119. https://doi.org/10.1016/S2095-3119(19)62633-X
Chincinska I, Gier K, Krügel U et al (2013) Photoperiodic regulation of the sucrose transporter StSUT4 affects the expression of circadian-regulated genes and ethylene production. Front Plant Sci 4. https://doi.org/10.3389/fpls.2013.00026
Fairbairn NJ (1953) A modified anthrone reagent. Chem Ind 4:86
Gong HL, Dusengemungu L, Igiraneza C, Rukundo P (2021) Molecular regulation of potato tuber dormancy and sprouting: a mini-review. Plant Biotechnol Rep 15:417–434
Herrera-Isidron L, Valencia-Lozano E, Rosiles-Loeza PY et al (2021) Gene expression analysis of microtubers of potato solanum tuberosum l. Induced in cytokinin containing medium and osmotic stress. Plants 10. https://doi.org/10.3390/plants10050876
Jiang L, Wang Z, Jin G et al (2019) Responses of Favorita Potato plantlets cultured in Vitro under fluorescent and light-emitting diode (LED) light sources. Am J Potato Res 96:396–402. https://doi.org/10.1007/s12230-019-09725-8
Kang JH, KrishnaKumar S, Atulba SLS et al (2013) Light intensity and photoperiod influence the growth and development of hydroponically grown leaf lettuce in a closed-type plant factory system. Hortic Environ Biotechnol 54:501–509. https://doi.org/10.1007/s13580-013-0109-8
Kondhare KR, Kumar A, Patil NS et al (2021) Development of aerial and belowground tubers in potato is governed by photoperiod and epigenetic mechanism. Plant Physiol 187:1071–1086
Li W, Liu SW, Ma JJ et al (2020) Gibberellin signaling is required for far-red light-induced shoot elongation in pinus tabuliformis seedlings1. Plant Physiol 182:658–668. https://doi.org/10.1104/pp.19.00954
Martínez-García JF, García-Martínez JL, Bou J, Prat S (2001) The interaction of gibberellins and photoperiod in the control of potato tuberization. J Plant Growth Regul 20:377–386. https://doi.org/10.1007/s003440010036
Melis R, van Staden J (1984) Tuberization and hormones. Z für Pflanzenphysiologie 113:271–283. https://doi.org/10.1016/s0044-328x(84)80008-2
Mohamed F, Omar G, Abd El-Hamed K, El-Safty B (2018) Influence of Plant Density and genotype on Potato Minituber Production from Microshoots and microtubers. Catrina Int J Environ Sci 17:77–84. https://doi.org/10.21608/cat.2018.14317
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with Tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/J.1399-3054.1962.TB08052.X
Plantenga FDM, Bergonzi S, Abelenda JA et al (2019) The tuberization signal StSP6A represses flower bud development in potato. J Exp Bot 70:925–936. https://doi.org/10.1093/jxb/ery420
Pundir RK, Pathak A, Upadhyaya DC et al (2021) Red and blue light-emitting diodes significantly improve in vitro tuberization of potato (Solanum Tuberosum L). J Hortic Res 29:95–108. https://doi.org/10.2478/johr-2021-0010
Rahman MZ, Shahinul Islam SM, Chowdhury AN, Subramaniam S (2015) Efficient microtuber production of potato in modified nutrient spray bioreactor system. Sci Hortic (Amsterdam) 192:369–374. https://doi.org/10.1016/j.scienta.2015.06.014
Raigond P, Buckseth T, Singh B et al (2019) Influence of photoperiod and EDTA salts on endogenous gibberellic acid concentration of tissue culture grown potato microplants. Agric Res 8:176–183. https://doi.org/10.1007/s40003-018-0364-0
Ramawat K, Merillon J (2013) Bulbous plants: biotechnology. CRC Press
Rehman M, Fahad S, Saleem MH et al (2020) Red light optimized physiological traits and enhanced the growth of ramie (Boehmeria nivea L). Photosynthetica 58:922–931. https://doi.org/10.32615/ps.2020.040
Salem J, Hassanein AM (2017) In vitro propagation, microtuberization, and molecular characterization of three potato cultivars. Biol Plant 61:427–437. https://doi.org/10.1007/s10535-017-0715-x
Sauer M, Robert S, Kleine-Vehn J (2013) Auxin: simply complicated. J Exp Bot 64:2565–2577
Sivakumar G, Heo JW, Kozai T, Paek KY (2006) Effect of continuous or intermittent radiation on sweet potato plantlets in vitro. J Hortic Sci Biotechnol 81:546–548. https://doi.org/10.1080/14620316.2006.11512101
Sun R, Okabe M, Miyazaki S et al (2023) Biosynthesis of gibberellin-related compounds modulates far-red light responses in the liverwort Marchantia polymorpha. bioRxiv
Teng Y, Zhang Y, Guo JT et al (2019) Acid pretreatment improves microtuberization of potato plantlets. Vitr Cell Dev Biol - Plant 55:36–43. https://doi.org/10.1007/s11627-018-09950-6
Uchendu EE, Shukla M, Saxena PK, Keller JER (2016) Cryopreservation of potato microtubers: the critical roles of sucrose and desiccation. Plant Cell Tissue Organ Cult 124:649–656. https://doi.org/10.1007/s11240-015-0916-y
Veramendi J, Sota V, Fernandez-San Millan A et al (2000) An in vitro tuberization bioassay to assess maturity class of new potato clones. J Hortic Sci Biotechnol 75:733–738. https://doi.org/10.1080/14620316.2000.11511315
Vreugdenhil D, Sergeeva LI (1999) Gibberellins and tuberization in potato. Potato Res 42:471–481. https://doi.org/10.1007/BF02358163
Wheeler RM, Fitzpatrick AH, Tibbitts TW (2019) Potatoes as a crop for Space Life Support: Effect of CO2, irradiance, and Photoperiod on Leaf Photosynthesis and Stomatal Conductance. Front Plant Sci 10. https://doi.org/10.3389/fpls.2019.01632
Xu X, Van Lammeren AAM, Vermeer E, Vreugdenhil D (1998) The role of gibberellin, abscisic acid, and sucrose in the regulation of potato tuber formation in vitro. Plant Physiol 117:575–584. https://doi.org/10.1104/pp.117.2.575
Xu JM, Liu Y, Liu MX, Xu ZG (2022) Proteomic, physiological, and anatomical analyses reveal the effects of Red, Blue, and White Light on the growth of Potato plantlets under in Vitro Culture. Russ J Plant Physiol 69. https://doi.org/10.1134/S1021443722060346
Yagiz AK, Yavuz C, Tarim C et al (2020) Effects of Growth regulators, media and explant types on microtuberization of Potato. Am J Potato Res 97:523–530. https://doi.org/10.1007/s12230-020-09801-4
Yang L, Fanourakis D, Tsaniklidis G et al (2021) Contrary to red, blue monochromatic light improves the bioactive compound content in broccoli sprouts. Agronomy 11. https://doi.org/10.3390/agronomy11112139
Acknowledgements
The authors acknowledge University of Jiroft for their supports, Mehdi Seif and Pegah Behzadi Rad are also appreciated for their scientific cooperation.
Funding
This work was supported by University of Jiroft (Iran).
Author information
Authors and Affiliations
Contributions
N.A. designed and directed the experiments. N.A. and A.R., M.S.H. and B.P. fulfilled the project. N.A analyzed the data and N.A., R.G drafted the manuscript. All authors have perused and consented to the submitted version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No possible conflict of interest was announced by the authors.
Additional information
Communicated by Jose M. Segui-Simarro.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Reisi, A., Askari, N., Sadat‑Hosseini, M. et al. Far-red spectrum leads to enhanced in vitro microtuberzation in potato (Solanum tuberosum cv. Sante). Plant Cell Tiss Organ Cult 156, 45 (2024). https://doi.org/10.1007/s11240-023-02673-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11240-023-02673-6