Abstract
Identifying a suscessul plant propagation method is very important to preserve wild plants as a genetic source. Hence, plant cells are driven by types and concentrations of plant growth regulators to produce in vitro callus mass to synthesize bioactive compounds. The aim of the present study is to induce callus from red ginger and to evaluate its suitability as a biofactory in terms of histology, phytochemical content and antioxidant potential. This is the first study to provide concrete evidence for the use of picloram for red ginger, Zingiber officinale var. rubrum. In this work, effect of different concentrations of auxinic picloram herbicide (0, 0.5, 1, 2, 4, and 8 mg/L) was evaluated on the callus induction of Zingiber officinale var. rubrum on various explants (leaf sheath, leaf, root). The induced callus was studied for growth kinetics, anatomical features, antioxidant capacity, and phytochemical content. The highest callogenesis frequency (93.75%) and biomass accumulation (3.68 g) were observed on leaf sheath explant cultured on ½ strength Murashige and Skoog medium supplemented with 8 mg/L which also requires earlier subculture duration (45 days post-inoculation) in comparison to lower concentrations. Morphological investigation through histological procedure demonstrated friable and non-embryogenic characteristics of the primary and subcultured callus which are used for secondary metabolite production. Cultivated leaf sheath (CLS) methanolic extract showed the highest total phenolic (191.26 mg GAE/g dry extract) and flavonoid (4.54 mg QE/g dry extract) contents contributing to antioxidant activity with an estimated EC50 of 0.208 mg/mL. Although comparatively lower than CLS extract, callus extracts showed higher antioxidant activity and significantly lower EC50 values than in vitro leaf sheath extract (IVLS extract). 4 H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl-, phenol, and phenolic glucoside were only present in callus cultures while methyl esters, fatty acids, and phytosterols could be obtained from leaf sheath and callus extracts. Notably beta-sitosterol was the highest in callus extracts while stigmasterol was only available in IVLS extracts. These phytosterols also responsible for synergistic antioxidant activity exhibited by callus extracts and IVLS, respectively. In conclusion, the callus culture of Z. officinale var. rubrum has been proven by the histological and phytochemical studies as a potential renewable source of bioactive phytochemical compounds. In the future, it can be employed for biotechnological practices such as elicitor-induced accumulation of secondary metabolites and genetic modification.
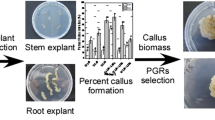
Graphical abstract

Key message
Picloram enhanced the callus induction, growth kinetics, antioxidant potentials and secondary metabolites production of Zingiber officinale var. rubrum callus cultures.





Similar content being viewed by others
References
Abd El-Hameid AR, Abo El-kheir ZA, Abdel-Hady MS, Helmy WA (2020) Identification of DNA variation in callus derived from Zingiber officinale and anticoagulation activities of ginger rhizome and callus. Bull Natl Res Cent 44:1–8. https://doi.org/10.1186/s42269-020-0281-9
Abdollahnezhad H, Bahadori MB, Pourjafar H, Movahhedin N (2021) Purification, characterization, and antioxidant activity of daucosterol and stigmasterol from Prangos ferulacea. Lett Appl NanoBioScience 10(2):2174–2180. https://doi.org/10.33263/LIANBS102.21742180
Aggarwal V, Joshi N, Varghese J (2018) Study of bioactive compounds from methanolic whole fruit extracts of Annona reticulata and Annona squamosa by GC–MS and evaluation of antioxidant activity. In: Dasgupta D (ed) Innovations in food, environment and healthcare national conference 2018. Life Science Informatics, Pune, pp 85–101
Al-Mansoub MA, Asmawi MZ, Murugaiyah V (2014) Effect of extraction solvents and plant parts used on the antihyperlipidemic and antioxidant effects of Garcinia atroviridis: a comparative study. J Sci Food Agric 94(8):1552–1558. https://doi.org/10.1002/jsfa.6456
Ali AMA, El-Nour MEM, Yagi SM (2018) Total phenolic and flavonoid contents and antioxidant activity of ginger (Zingiber officinale Rosc.) Rhizome, callus and callus treated with some elicitors. J Genet Eng Biotechnol 16(2):677–682. https://doi.org/10.1016/j.jgeb.2018.03.003
Amiri S, Shakeri A, Sohrabi MR, Khalajzadeh S, Ghasemi E (2019) Optimization of ultrasonic assisted extraction of fatty acids from Aesculus hippocastanum fruit by response surface methodology. Food Chem 271:762–766. https://doi.org/10.1016/j.foodchem.2018.07.144
An K, Zhao D, Wang Z, Wu J, Xu Y, Xiao G (2016) Comparison of different drying methods on Chinese ginger (Roscoe): changes in volatiles, chemical profile, antioxidant properties, and microstructure. Food Chem 197:1292–1300. https://doi.org/10.1016/j.foodchem.2015.11.033
Ashraf MA, Iqbal M, Rasheed R, Hussain I, Riaz M, Arif MS (2018) Environmental stress and secondary metabolites in plants: an overview. In: Ahmad P, Ahanger MA, Singh VP, Tripathi DK, Alam P, Alyemani MN (eds) Plant metabolites and regulation under environmental stress. Academic Press, Cambridge, pp 153–167
Asrori MI, Sasamoto H, Ogita S (2020) In vitro bioassay of allelopathy in Robusta coffee callus using sandwich method. Proc Int Conf Food Agric Nat Resour Indonesia 194:147–151. https://doi.org/10.2991/aer.k.200325.030H
Awan MF, Iqbal MS, Sharif MN, Tabassum B, Tariq M, Murtaza S, Ali S, Raza A, Bukhari SAR, Nasir IA (2019) Evaluation of genotypic and hormone mediated callus induction and regeneration in sugarcane (Saccharum officinarum L.). Evaluation 4(6):70–76
Basma AA, Zakaria Z, Latha LY, Sasidharan S (2011) Antioxidant activity and phytochemical screening of the methanol extracts of Euphorbia hirta L. Asian Pac. J Trop Med 4(5):386–390. https://doi.org/10.1016/S1995-7645(11)60109-0
Bertrand RL (2019) Lag phase is a dynamic, organized, adaptive, and evolvable period that prepares bacteria for cell division. J Bacteriol Res 201(7):e00697–e00618
Bhatia S, Sharma K, Dahiya R, Bera T (2015) Plant tissue culture. Mod Appl Plant Biotechnol Pharm Sci. https://doi.org/10.1016/B978-0-12-802221-
Bhattacharya P, Sen SK (1980) Potentiality of leaf sheath cells for regeneration of rice (Oryza sativa L.) plants. Theor Appl Genet 57(4):87–90. https://doi.org/10.1007/BF00277771
Binzet R, Binzet G, Gumus I, Turunc E, Solmaz U, Keskin E, Dogen A, Arslan H (2019) Chemical composition and antimicrobial activity of essential oil and various extracts of Onosma sieheana Hayek roots. J Essent Oil-Bear Plants 22(1):94–104
Burgos-Zazueta M, Alonzo-Macías M, Ramos-López MA, Cardador-Martínez A, Martínez-Peniche RA, Soto-Muñoz L (2021) Osmotic tolerance response of biocontrol agent Yarrowia lipolytica m18/3b under pre-and postharvest conditions. Biocontrol Sci Technol 31(2):153–170. https://doi.org/10.1080/09583157.2020.1833303
Castro AHF, Lima MM, Paiva R, de Alvarenga AA, Soter MO (2008) Growth curve, phenylanine ammonia-lyase activity and total phenol levels in callus of Stryphnodendron adstringens (Mart.) Coville (Fabaceae-Mimosoideae). Plant Cell Cult e Micropropag (Brazil) 4(2):99–104
Chakkinga Thodi R, Ibrahim JM, Nair AS, Thacheril Sukumaran S (2023) Exploring the potent inhibitor βstigmasterol from Pittosporum dasycaulon Miq. leaves against snake venom phospholipase A2 protein through in vitro and molecular dynamics behavior approach. Toxin Rev 42(1):85–98. https://doi.org/10.1080/15569543.2021.2021946
Chear NJY, Fauzi AN, Khaw KY, Choi SB, Yaacob NS, Lai CS (2019) Free radical scavenging and cytotoxic properties of acylated and non-acylated kaempferol glycosides from Stenochlaena palustris: a perspective on their structure–activity relationships. Pharm Chem J 53(3):1–6. https://doi.org/10.1007/s11094-019-01977-2
Cheng SY, Xu F, Wang Y (2009) Advances in the study of flavonoids in Ginkgo biloba leaves. J Med Plant Res 3(13):1248–1252
Choudhary D, Shekhawat JK, Kataria V (2019) GC-MS analysis of bioactive phytochemicals in methanol extract of aerial part and callus of Dipterygium glaucum Decne. Pharmacogn J. https://doi.org/10.5530/pj.2019.11.165
Coimbra MC, Chagas RCR, Vilela MSP, Castro AHF (2019) Growth, morphology and bioactive phenolic compounds production in Pyrostegia venusta calli. Biocatal Agric Biotechnol 18:101036. https://doi.org/10.1016/j.bcab.2019.101036
Da Costa CT, De Almeida MR, Ruedell CM, Schwambach J, Maraschin FDS, Fett-Neto AG (2013) When stress and development go hand in hand: main hormonal controls of adventitious rooting in cuttings. Front Plant Sci 4:133. https://doi.org/10.3389/fpls.2013.00133
da Costa L, da Silva J, Bier ALL, Brondani MCJ, Gollo GE, Letti AL, Erasmo LAJ, Soccol EAL (2015) Callus growth kinetics of physic nut (Jatropha curcas L.) and content of fatty acids from crude oil obtained in vitro. Appl Biochem Biotechnol 176(3):892–902. https://doi.org/10.1007/s12010-015-1618-y
Daffalla HM, Elsheikh AM, Ali HA, Khalafalla MM (2019) Callus maintenance and cell line selection of Grewia Tenax. J Herbs Spices Med Plants 25(3):218–235. https://doi.org/10.1080/10496475.2019.1595256
Damayanti F, Indrianto A, Sasongko AB, Fajarina S, Prabowo BH, Iskandar A, Hidayati L, Tunjung WAS (2020) September variation of 2, 4-dichlorophenoxyacetic acid (2, 4-D) concentration on kaffir lime callus growth as raw material for cell suspension. AIP Conf Proc 2260(1):030012
de Oliveira LAR, de Oliviera ACA, Machado CDA, Cardoso MN, Santana FV, Miranda ICD, de Castro EM, Muniz ADS, Ledo ADS (2018) Induction, growth kinetics and morpho-histological characterization of neem callus. J Agric Sci 10(6):283–290. https://doi.org/10.5539/jas.v10n6p283
Efferth T (2019) Biotechnology applications of plant callus cultures. Engineering 5(1):50–59. https://doi.org/10.1016/j.eng.2018.11.006
El-Mageid IS (2019) Evaluation of genetic stability by using protein and ISSR markers during callus development stage of some date palm (Phoenix dactylifera L.) cultivars under effect of 2, 4-D and Picloram. Middle East J Appl Sci 9(02):483–493
El-Nabarawy MA, El-Kafafi SH, Hamza MA, Omar MA (2015) The effect of some factors on stimulating the growth and production of active substances in Zingiber officinale callus cultures. Ann Agric Sci 60:1–9. https://doi.org/10.1016/j.aoas.2014.11.020
Geraldi A, Wardana AP, Aminah NS, Kristanti AN, Sadila AY, Wijaya NH, Diningrum NID, Hajar VR, Manuhara YSW (2022) Tropical medicinal plant extracts from Indonesia as antifungal agents against Candida albicans. Front Biosci Landmark. 27(9):274. https://doi.org/10.31083/j.fbl2709274
Ghasemzadeh A, Jaafar HZ (2011) Antioxidant potential and anticancer activity of young ginger (Zingiber officinale Roscoe) grown under different CO2 concentration. J Med Plant Res 5(14):3247–3255
Ghasemzadeh A, Jaafar HZ, Rahmat A, Wahab PEM, Halim MRA (2010) Effect of different light intensities on total phenolics and flavonoids synthesis and anti-oxidant activities in young ginger varieties (Zingiber officinale Roscoe). Int J Mol Sci 11(10):3885–3897. https://doi.org/10.3390/ijms11103885
Gnasekaran P, Mahmood M, Subramaniam S (2016) Ultrastructure study of Vanda Kasem’s Delight orchid’s protocorm-like body. Hortic Bras 34(3):333–339. https://doi.org/10.1590/S0102-05362016003005
Godoy-Hernández G, Vázquez-Flota FA (2006) Growth measurements. In: Loyola-Vargas VM, Vázquez-Flota F (eds) Plant cell culture protocols: methods in molecular biology™, 2nd edn. Humana Press, Totowa, pp 51–58
Gomathi S, Velayutham P, Karthi C, Santhoshkumar S (2019) In vitro callus induction and phytochemical screening of Corbichonia decumbens (Forssk.) Exell through GC MS analysis. J Pharmacogn Phytochem 8(5):566–571
Goswami S, Das R, Ghosh P, Chakraborty T, Barman A, Ray S (2020) Comparative antioxidant and antimicrobial potentials of leaf successive extract fractions of poison bulb, Crinum asiaticum L. Ind. Crops Prod 154:112667. https://doi.org/10.1016/j.indcrop.2020.112667
Gul MT, Dheyab AS, Shaker EK, Muhammad N, Pauzi AN (2020) In vitro evaluation of anti-urolithiatic properties of Strobilanthes crispus extracted using different solvents. Res J Chem Environ 24:1
Gurav SS, Gurav NS, Patil AT, Duragkar NJ (2020) Effect of explant source, culture media, and growth regulators on callogenesis and expression of secondary metabolites of Curcuma Longa. J Herbs Spices Med Plants 26(2):172–190. https://doi.org/10.1080/10496475.2019.1689542
Habibah NA, Moeljopawiro S, Dewi K, Indrianto A (2018), March Callus induction and flavonoid production on the immature seed of Stelechocarpus burahol. International Conference on Mathematics, Science and Education 2017, Semarang, Indonesia
Habibah NA, Widiatningrum T, Anggraito YU, Rahayu ES, Mukhtar K, Wijayanti N, Mustafa F (2019) Growth of Elaeocarpus grandiflorus callus cultures in MS medium with various concentrations of growth regulators. J. Phys. Conf. Ser. 1321(3):032037
Handayani D, Sari HC, Julianti E, Artasasta MA (2023) Endophytic fungus isolated from Zingiber officinale Linn. var. rubrum as a source of antimicrobial compounds. J Appl Pharm Sci. https://doi.org/10.7324/JAPS.2023.134154
Herawati H, Anisa AK, Widiatmoko KD, Alam SSP, Diari IA, Naprila ZH, Kisya RLA, Puspabela A, Permata FS (2022) Effect of red ginger powder (Zingiber officinale var. rubrum) as a feed additive for starter and finisher broiler chicken to increase immunoglobulin A and immunoglobulin Y expression and to prevent intestinal injury due to Salmonella enteritidis infection. Vet. World 15(6):1506. https://doi.org/10.14202/vetworld.2022.1506-1514
Hu W, Yu A, Wang S, Bai Q, Tang H, Yang B, Wang M, Kuang H (2023) Extraction, purification, structural characteristics, biological activities, and applications of the polysaccharides from Zingiber officinale Roscoe. (Ginger): a review. Molecules 28(9):3855. https://doi.org/10.3390/molecules28093855
Huang B, Han L, Li S, Yan C (2015) Optimization of induction, subculture conditions, and growth kinetics of Angelica sinensis (Oliv.) Diels callus. Pharmacogn Mag 11(43):574. https://doi.org/10.4103/0973-1296.160443
Hundare A, Dhaytadak B, Kudale S, Joshi N (2018) Growth kinetics and diosgenin estimation from callus cultures of Costus speciosus (Koen. Ex. Retz). Nat Prod Res 32(15):1809–1816. https://doi.org/10.1080/14786419.2017.1405402
Hussein S, Halmi MIE, Kiong ALP (2016) Modelling the growth kinetics of callus cultures from the seedling of Jatropha curcas L. according to the modified gompertz model. J Microbiol Biotechnol 4(1):20–23. https://doi.org/10.54987/jobimb.v4i1.284
Ibrahim DA, Danial GH, Mosa VM, Khalil BM (2015) Plant regeneration from shoot tips-derived callus of ginger (Zingiber officinale Rosc). J Exp Agric Int 7(1):55–61
Ijaz B, Sudiro C, Hyder MZ, Malik SI, Farrakh S, Schiavo FL, Yasmin T (2019) Histo-morphological analysis of rice callus cultures reveals differential regeneration response with varying media combinations. In Vitro Cell Dev Biol Plant 55(5):569–580. https://doi.org/10.1007/s11627-019-09974-6
Ikeuchi M, Sugimoto K, Iwase A (2013) Plant callus: mechanisms of induction and repression. Plant Cell 25(9):3159–3173. https://doi.org/10.1105/tpc.113.116053
Ishimaru Y, Hayashi K, Suzuki T, Fukaki H, Prusinska J, Meester C, Quareshy M, Egoshi S, Matsuura H, Takahashi K, Kato N (2018) Jasmonic acid inhibits auxin-induced lateral rooting independently of the coronatine insensitive1 receptor. Plant Physiol 177(4):1704–1716
Kaewthip W, Dheeranupattana S, Junta P, Shank L (2021) Sterile tissue preparation and callus induction of Curcuma longa Linn. CMUJ J Nat Sci 20(3):e2021062
Karuppusamy S (2009) A review on trends in production of secondary metabolites from higher plants by in vitro tissue, organ and cell cultures. J Med Plants Res 3(13):1222–1239
Khan N, Chaudhary MF, Abbasi AM, Khan SA, Nazir A (2017) Development of an efficient callus derived regeneration system for garlic (Allium safivum L.) from root explant. J Plant Breed Agric 1(1):3
Kirdat PN, Dandge PB, Hagwane RM, Nikam AS, Mahadik SP, Jirange ST (2021) Synthesis and characterization of ginger (Z. officinale) extract mediated iron oxide nanoparticles and its antibacterial activity. Mater Today Proc 43:2826–2831. https://doi.org/10.1016/j.matpr.2020.11.422
Kowalczyk T, Merecz-Sadowska A, Rijo P, Mori M, Hatziantoniou S, Górski K, Szemraj J, Piekarski J, Śliwiński T, Bijak M, Sitarek P (2022) Hidden in plants—a review of the anticancer potential of the Solanaceae family in in vitro and in vivo studies. Cancers 14(6):1455. https://doi.org/10.3390/cancers14061455
Laelago Ersedo T, Teka TA, Fikreyesus Forsido S, Dessalegn E, Adebo JA, Tamiru M, Astatkie T (2023) Food flavor enhancement, preservation, and bio-functionality of ginger (Zingiber officinale): a review. Int J Food Prop 26(1):928–951. https://doi.org/10.1080/10942912.2023.2194576
Lee JH, Pijut PM (2017) Adventitious shoot regeneration from in vitro leaf explants of Fraxinus nigra. Plant Cell Tissue Organ Cult 130(2):335–343. https://doi.org/10.1007/s11240-017-1228-1
Lemma T, Ruiz GCM, Oliveira ON Jr, Constantino CJ (2019) The pesticide picloram affects biomembrane models made with Langmuir monolayers. Colloids Surf B 181:953–958. https://doi.org/10.1016/j.colsurfb.2019.06.060
Li H, Tang XY, Wu CJ, Yu SJ (2019) Formation of 2, 3-dihydro-3, 5-dihydroxy-6-methyl-4 (h)-pyran-4-one (DDMP) in glucose-amino acids Maillard reaction by dry-heating in comparison to wet-heating. LWT 105:156–163. https://doi.org/10.1016/j.lwt.2019.02.015
Li Y, Kong D, Fu Y, Sussman MR, Wu H (2020) The effect of developmental and environmental factors on secondary metabolites in medicinal plants. Plant Physiol Biochem 148:80–89. https://doi.org/10.1016/j.plaphy.2020.01.006
Lukiati B, Sulisetijono S, Nugrahaningsih N, Masita R (2020) Determination of total phenol and flavonoid levels and antioxidant activity of methanolic and ethanolic extract Zingiber officinale Rosc var. Rubrum rhizome. AIP Conf Proc 2231(1):040003. https://doi.org/10.1063/5.0002657
Ma RH, Ni ZJ, Zhu YY, Thakur K, Zhang F, Zhang YY, Hu F, Zhang JG, Wei ZJ (2021) A recent update on the multifaceted health benefits associated with ginger and its bioactive components. Food Funct 12(2):519–542
Manisha P, Chandrashekhar P, Raghunath M (2018) Phytochemical investigation and validation of antioxidant potential of β-sitosterol from tubers of Eulophia herbacea and Eulophia ochreata. Int J Pharmacogn Phytochem Res 10(9):309–316. https://doi.org/10.25258/phyto.10.9.1
Manssouri M, Znini M, Majidi L (2020) Studies on the antioxidant activity of essential oil and various extracts of Ammodaucus leucotrichus Coss. & Dur. Fruits from Morocco. J Taibah Univ Sci 14(1):124–130. https://doi.org/10.1080/16583655.2019.1710394
Mavrikou S, Tsekouras V, Karageorgou MA, Moschopoulou G, Kintzios S (2020) Anticancer and biochemical effects of Viscum album L. protein extracts on HeLa cells. Plant Cell Tissue Organ Cult 140(2):369–378. https://doi.org/10.1007/s11240-019-01733-0
Mazumder K, Nabila A, Aktar A, Farahnaky A (2020) Bioactive variability and in vitro and in vivo antioxidant activity of unprocessed and processed flour of nine cultivars of Australian lupin species: a comprehensive substantiation. Antioxidants 9(4):282. https://doi.org/10.3390/antiox9040282
Mendoza MG, Kaeppler HF (2002) Auxin and sugar effects on callus induction and plant regeneration frequencies from mature embryos of wheat (Triticum aestivum L.). In Vitro Cell Dev Biol Plant 38(1):39–45. https://doi.org/10.1079/IVP2001250
Miri SM (2020) Micropropagation, callus induction and regeneration of ginger (Zingiber officinale Rosc). Open Agric 5(1):75–84. https://doi.org/10.1515/opag-2020-0008
Moriasi GA, Ireri AM, Ngugi MP (2020) In vivo cognitive-enhancing, ex vivo malondialdehyde-lowering activities and phytochemical profiles of aqueous and methanolic stem bark extracts of Piliostigma thonningii (schum.). Int J Alzheimers Dis. https://doi.org/10.1155/2020/1367075
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Musfir Mehaboob V, Faizal K, Thilip C, Raja P, Thiagu G, Aslam A, Shajahan A (2019) Indirect somatic embryogenesis and Agrobacterium-mediated transient transformation of ginger (Zingiber officinale Rosc.) Using leaf sheath explants. J Hortic Sci Biotechnol 94(6):753–760. https://doi.org/10.1080/14620316.2019.1624201
Mustafa I, Chin NL, Fakurazi S, Palanisamy A (2019) Comparison of phytochemicals, antioxidant and anti-inflammatory properties of sun-, oven-and freeze-dried ginger extracts. Foods 8(10):456. https://doi.org/10.3390/foods8100456
Nanok K, Sansenya S (2021) Combination effects of rice extract and five aromatic compounds against α-glucosidase, α-amylase and tyrosinase. J Biosci Bioeng 132(1):9–17. https://doi.org/10.1016/j.jbiosc.2021.02.003
Ningsih IY, Dianasari D, Hidayat MA (2023) Morphology and physicochemical properties of starch extracted from Indonesian ginger. Pharm Educ 23(2):47–52. https://doi.org/10.46542/pe.2023.232.4752
Ng ZX, Koick YTT, Yong PH (2020) Comparative analyses on radical scavenging and cytotoxic activity of phenolic and flavonoid content from selected medicinal plants. Nat Prod Res. https://doi.org/10.1080/14786419.2020.1749617
Nurwahyuni I, Situmorang M, Sinaga R (2020) Plant regeneration through callus cultures from leaf explant of Sumatra Benzoin (Styrax benzoin). Int J For Res. https://doi.org/10.1155/2020/8860178
Ozyigit II, Dogan I, Hocaoglu-Ozyigit A, Yalcin B, Erdogan A, Yalcin IE, Cabi E, Kaya Y (2023) Production of secondary metabolites using tissue culture-based biotechnological applications. Front Plant Sci 14:1132555. https://doi.org/10.3389/fpls.2023.1132555
Pawar NV, Pai SR, Nimbalkar MS, Dixit GB (2015) RP-HPLC analysis of phenolic antioxidant compound 6-gingerol from in vitro cultures of Zingiber officinale Roscoe. Plant Sci Today 2(1):24–28. https://doi.org/10.1016/j.foodchem.2010.11.090
Prakoeswa SA, Suryaningsih DR (2020) Vegetative propagation in vitro with content analysis of ginger oil from calluses of “Jahe Gajah” (Zingiber officinale) on treatments of types of mediums and carbohydrates. IOP Conf Ser Earth Environ. Sci. 484(1):012017. https://doi.org/10.1088/1755-1315/484/1/012017
Ramawat KG (2009) Herbal drugs: ethnomedicine to modern medicine. Springer-Verlag, Berlin
Rani M, Shylaja MR, Mathew D, Girija D, Shankar MA, Sureshkumar P, Beena C (2022) Potential of microrhizomes for in vitro gingerol and shogaol synthesis in ginger (Zingiber officinale Rosc.). Proc Natl Acad Sci India Sect B Biol Sci. 92(1):121–129. https://doi.org/10.1007/s40011-021-01314-2
Rawat V, Ghildiyal A, Singh L, Jugran AK, Bhatt ID, Nandi SK, Pande V (2020) Methyl jasmonate induced polyphenols and antioxidant production in callus suspension culture of Nardostachys jatamansi. Plant Biosyst 154(6):851–859. https://doi.org/10.1080/11263504.2019.1701124
Rubab M, Chelliah R, Saravanakumar K, Barathikannan K, Wei S, Kim JR, Yoo D, Wang MH, Oh DH (2020) Bioactive potential of 2-methoxy-4-vinylphenol and benzofuran from Brassica oleracea L. var. Capitate f, rubra (Red Cabbage) on oxidative and microbiological stability of beef meat. Foods 9(5):568. https://doi.org/10.3390/foods9050568
Sarmoko S, Solihati I, Setyono J, Ekowati H, Fadlan A (2020) Zingiber Officinale Var. Rubrum extract increases the cytotoxic activity of 5-fluorouracil in colon adenocarcinoma widr cells. Indones J Pharm. 31:266–272. https://doi.org/10.22146/ijp.859
Salma U, Kundu S, Ali MN, Mandal N (2019) Somatic embryogenesis-mediated plant regeneration of Eclipta alba (L.) Hassk. And its conservation through synthetic seed technology. Acta Physiol Plant 41(6):103. https://doi.org/10.1007/s11738-019-2898-6
Santos MRAD, Souza CAD, Paz ES (2017) Growth pattern of friable calluses from leaves of Capsicum annuum var. annuum cv. Iberaba Jalapeño. Ciênc Agron 48:523–530. https://doi.org/10.5935/1806-6690.20170061
Saw NMMT, Riedel H, Kütük O, Ravichandran K, Smetanska I (2010) Effect of elicitors and precursors on the synthesis of anthocyanin in grape Vitis vinifera cell cultures. Energy Res J 1(2):189–192. https://doi.org/10.3844/erjsp.2010.189.192
Sen MK, Nasrin S, Rahman S, Jamal AHM (2014) In vitro callus induction and plantlet regeneration of Achyranthes aspera L., a high value medicinal plant. Asian Pac J Trop Biomed 4(1):40–46. https://doi.org/10.1016/S2221-1691(14)60206-9
Sengar K, Sengar RS, Garg SK (2011) The effect of in-vitro environmental conditions on some sugarcane varieties for micropropagation. Afr J Biotechnol 10(75):17122–17126. https://doi.org/10.5897/AJB11.2195
Setiaji A, Annisa RR, Rumiyati R, Semiarti E (2020) Induction and growth kinetics callus of Tomato (Solanum lycopersicum). Biosaintifika: J Biol Biol Educ 12(1):35–41
Shah MD, Seelan JSS, Iqbal M (2020) Phytochemical investigation and antioxidant activities of methanol extract, methanol fractions and essential oil of Dillenia suffruticosa leaves. Arab J Chem 13(9):7170–7182. https://doi.org/10.1016/j.arabjc.2020.07.022
Sivakumar T (2019) Phytochemical screening and gas chromatography–mass spectroscopy analysis of bioactive compounds and biosynthesis of silver nanoparticles using sprout extracts of Vigna radiata L. and their antioxidant and antibacterial activity. Asian J Pharm Clin Res 12(2):180–184. https://doi.org/10.22159/ajpcr.2019.v12i2.29253
Somashekar PV, Rathore TS, Fatima T (2018) Vitro plant regeneration of Dendrocalamus stocksii (Munro) M. Kumar, Remesh & Unnikrisnan, through somatic embryogenesis. Am J Plant Sci 9(12):2429–2445. https://doi.org/10.4236/ajps.2018.912176
Stanly C, Bhatt A, Ali HMD, Keng CL, Lim BP (2011) Evaluation of free radical scavenging activity and total phenolic content in the petiole-derived callus cultures of Zingiber zerumbet Smith. J Med Plant Res 5(11):2210–2217
Sugimoto K, Gordon SP, Meyerowitz EM (2011) Regeneration in plants and animals: dedifferentiation, transdifferentiation, or just differentiation? Trends Cell Biol 21(4):212–218. https://doi.org/10.1016/j.tcb.2010.12.004
Suraiya M, Alina W (2018) Efficient callus induction and regeneration in selected Indica rice. Agronomy 8(5):77. https://doi.org/10.3390/agronomy8050077
Truong DH, Nguyen DH, Ta NTA, Bui AV, Do TH, Nguyen HC (2019) Evaluation of the use of different solvents for phytochemical constituents, antioxidants, and in vitro anti-inflammatory activities of Severinia buxifolia. J Food Qual. https://doi.org/10.1155/2019/8178294
Ullah MA, Tungmunnithum D, Garros L, Hano C, Abbasi BH (2019) Monochromatic lights-induced trends in antioxidant and antidiabetic polyphenol accumulation in in vitro callus cultures of Lepidium sativum L. J Photochem Photobiol B Biol 196:111505. https://doi.org/10.1016/j.jphotobiol.2019.05.002
Xu C, Cao H, Zhang Q, Wang H, Xin W, Xu E, Zhang S, Yu R, Yu D, Hu Y (2018) Control of auxin-induced callus formation by bZIP59–LBD complex in Arabidopsis regeneration. Nat Plants 4(2):108–115. https://doi.org/10.1038/s41477-017-0095-4
Zhang S, Kou X, Zhao H, Mak KK, Balijepalli MK, Pichika MR (2022) Zingiber officinale var. rubrum: red ginger’s medicinal uses. Molecules 27(3):775. https://doi.org/10.3390/molecules27030775
Zhang S, Pei Y, Wang J, Lin X, Tang Z, Zhou L, Chen T, Feng S, Ding C, Chen Y, Zhang Z, Yuan S, Yuan M (2023) Isolation and characterization of an endophytic fungi from Zingiber officinale Rosc. and its activity to promote the growth of tissue culture ginger plantlets in bottles. Plant Cell Tissue Organ Cult. https://doi.org/10.21203/rs.3.rs-2552252/v1
Acknowledgements
UMK Rising Star 2021 Grant (R/STA/A0800/01350A/007/2021/ 00937).
Funding
This study was funded by UMK Rising Star 2021 Grant Number (R/STA/A0800/01350A/007/2021/ 00937).
Author information
Authors and Affiliations
Contributions
PG, SA, ZAR, DKW, MS, NJYC and SS designed and conducted the experiments, and analysed the data. SS, VM, JU and CBL conceptualised and supervised the research. PG, SA and SS wrote the manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
The results in the Manuscript are the author’s original work. The authors confrm that this manuscript has not been previously published and is not currently under consideration by any other journal. Additionally, all of the authors have approved the contents of this paper and have agreed to the journal’s submission policies.
Additional information
Communicated by Ali R. Alan.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gnasekaran, P., Rahman, Z.A., Chew, B.L. et al. Picloram enhanced the callus induction, growth kinetics, antioxidant potentials, and secondary metabolites production of Zingiber officinale var. rubrum callus cultures. Plant Cell Tiss Organ Cult 155, 843–859 (2023). https://doi.org/10.1007/s11240-023-02603-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02603-6