Abstract
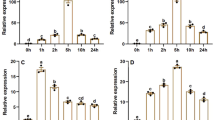
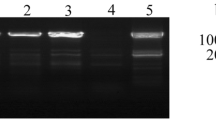
Tea plant (Camellia sinensis (L.) O. Kuntze) is an important woody economic crop that has been cultivated worldwide. Adversity stress acts as a significant factor affecting the growth and development of tea plants. Brassinazole-resistant (BZR) family protein is an essential factor in regulating plant stress resistance and plays a key role in cold stress response, heat, drought and salt resistances. In this study, the promoter region upstream of CsBZR1-2 was cloned. The promoter sequence and its 5′-end truncated segment were inserted upstream of β-glucuronidase (GUS) gene in the pCambia1391Z plasmid. Stably expressed transgenic tobacco lines were obtained by Agrobacterium tumefaciens-mediated genetic transformation. The results showed that except for promoter lacking the core element, both full-length and truncated promoters could initiate the expression of GUS, but the level of expression was affected by the degree of deletion at the 5′-end. The core region of the promoter was in a 311 bp region including the transcription start site (TSS). In addition, the regulatory mechanism of the promoter was explored. It was found that the genes regulated by the promoter were responsive to stress such as cis-acting elements (MeJA, ABRE and LTR), BR and light. This paper provides a theoretical basis to further explore the regulatory mode of the promoter and to achieve efficient gene expression.
Key Message
The CsBZR1-2 promoter was successfully cloned and studied its function in regulating the expression of the BZR1-2 gene.








Similar content being viewed by others
Data availability
All authors have ensured that all data, materials, software, and custom codes support the published claims and comply with field standards.
References
Birnboim HC (1983) A rapid alkaline extraction method for the isolation of plasmid DNA. Methods Enzymol 100:243–255. https://doi.org/10.1016/0076-6879(83)00059-2
Chang X, Yang M, Li H, Wu J, Zhang J, Yin C, Ma W, Chen H, Zhou F, Lin Y (2023) Cloning of the promoter of rice brown planthopper feeding-inducible gene OsTPS31 and identification of related cis-regulatory elements. Pest Manag Sci 202379:1809–1819. https://doi.org/10.1002/ps.7356
Chen J (2022) Low temperature regulation and biological function of transcription factor BZR1. Lanzhou University. https://doi.org/10.27204/d.cnki.glzhu.2018.000019
Cheng H, Gu L (2014) Isolation, molecular cloning and characterization of a cold-responsive gene, AmDUF1517, from Ammopiptanthus mongolicus. Plant Cell Tissue Organ Cult 117:201–211. https://doi.org/10.1007/s11240-014-0433-4
Guo JH (2017) Cloning and functional study of transcription factor genes CsWRKY57 and CsWRKY40 in tea plant. Northwest a&f Univ 02:123–126
Guo L, Guo JC, Zhu WC, Jiang XR (2016) Optimized synchronous extraction process of tea polyphenols and polysaccharides from Huaguoshan Yunwu tea and their antioxidant activities. Food Bioprod 100:303–310. https://doi.org/10.1016/j.fbp.2016.08.001
Han T, Yu J, Zhuang J, Wang Z, Sun X, Zhang Y (2022) The characterization of columnar apple gene MdCoL promoter and its response to abscisic acid, brassinosteroid and gibberel-lic acid. Int J Mol Sci 23:10781. https://doi.org/10.3390/ijms231810781
He JX, Gendron JM, Sun Y, Gampala SS, Gendron N, Sun CQ, Wang ZY (2005) BZR1 is a trans-criptional repressor with dual roles in brassinosteroid homeostasis and growth responses. Science 307:1634–1638. https://doi.org/10.1126/science.1107580
Hellemans J, Mortier G, De Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8:R19. https://doi.org/10.1186/gb-2007-8-2-r19
Hu X (2021) Cloning and Functional Analysis of Cpcor413pm1 Gene Promoter in Chimonanthus praecox. Dissertation, Southwest University
Jin Q, Wang Z, Chen Y, Luo Y, Tian N, Liu Z, Huang J, Liu S (2022) Transcriptomics analysis reveals the signal transduction mechanism of brassinolides in tea leaves and its regulation on the growth and development of Camellia sinensis. BMC Genomics 23(1):29. https://doi.org/10.1186/s12864-021-08179-9
Jing H, Dan W, Zhang J, Wang Y (2016) Functional analysis of the fruit-specific promoter of VqSTS6 from the Chinese wild grape, Vitis quinquangularis. Agric Gene 1:38–45. https://doi.org/10.1016/j.aggene.2016.06.002
Li Y, Wang C, Huai Q, Guo F, Liu L, Feng R, Sun C (2016) Effects of tea or tea extract on meta-bolic profiles in patients with type 2 diabetes mellitus: a meta-analysis of ten randomized controlled trials. Diabetes Metab Res 32:2–10. https://doi.org/10.1002/dmrr.2641
Li Z, Li B, Zhao Y, Zhao D (2021) Cloning and characterization of the DIR1 promoter from Eucommia ulmoides Oliv and its response to hormonal and abiotic stress. Plant Cell Tiss Organ Cult 146:313–322. https://doi.org/10.1007/s11240-021-02070-x
Li C, Jiang W, Jiang X, Gai Y (2023) Cloning and functional analysis of the PLkF3H2 promoter in Larix kaempferi. Plant Cell Tiss Organ Cult 154:481–491. https://doi.org/10.1007/s11240-023-02467-w
Lin M, Yan J, Ali MM, Wang S, Tian S, Chen F, Lin Z (2022) Isolation and functional characterization of a green-tissue promoter in Japonica Rice (Oryza sativa subsp. japonica). Biology (basel) 11:1092. https://doi.org/10.3390/biology11081092
Lv D, Zhang Y (2017) Isolation and functional analysis of apple MdHMGR1 and MdHMGR4 gene promoters in transgenic Arabidopsis thaliana. Plant Cell Tissue Organ Cult 129:133–143. https://doi.org/10.1007/s11240-016-1162-7
Ma Q, Ma B, Mu B, Ma H (2018) Research progress in cloning and analysis of plant gene promoters. China Agric Abstr Agric Eng 30:23–29. https://doi.org/10.16420/j.issn.0513-353x.2020-0717
Pan H, Wang F, Rankin GO, Rojanasakul Y, Tu Y, Chen YC (2017) Inhibitory effect of black tea pigments, theaflavin-3/3’-gallate against cisplatin-resistant ovarian cancer cells by inducing apoptosis and G1 cell cycle arrest. Int J Oncol 51:1508–1520. https://doi.org/10.3892/ijo.2017.4145
Qin Y, Quan X (2017) Cloning and identification of wheat stress-induced expression and tissue specific expression Promoter. In: Abstracts of the eighth national wheat genomics and molecular breeding conference
Samanta S (2020) Potential bioactive components and health promotional benefits of tea (Camellia sinensis). J Am Nutr Assoc 41:65–93. https://doi.org/10.1080/07315724.2020.1827082
Shi J, Wang JT, Peng QH, Lu HP, Baldermann S, Lin Z (2021) Changes of seven fat-soluble pigments in fresh tea leaves and postharvest oolong tea processing induced by exogenous methyl jasmonate. Agric Sci China 54(18):3984–3997
Stålberg K, Ellerström M, Josefsson LG, Rask L (1993) Deletion analysis of a 2S seed storage protein promoter of Brassica napus in transgenic tobacco. Plant Mol Biol 23:671–683. https://doi.org/10.1007/BF00021523
Tian S, Yin X, Fu P, Wu W, Lu J (2019) Ectopic expression of grapevine gene VaRGA1 in Arabidopsis improves resistance to downy mildew and Pseudomonas syringae pv. tomato DC3000 but increases susceptibility to Botrytis cinerea. Int J Mol Sci 21:193. https://doi.org/10.3390/ijms21010193
Wang ZY, Nakano T, Gendron J, He J, Chen M, Vafeados D, Yang Y, Fujioka S, Yoshida S, Asami T, Chory J (2002) Nuclear-localized BZR1 mediates brassinosteroid induced growth and feedback suppression of brassinosteroid biosynthesis. Dev Cell 2:505–513. https://doi.org/10.1016/S1534-5807(02)00153-3
Wang Y, Gao YH, Zhu YQ, Si JP (2019) Cloning and functional analysis of DcCDPK8 gene promoter from Dendrobium candidum. Chin J Tradit Chin Med 44:293–297. https://doi.org/10.19540/j.cnki.cjcmm.20181106.005
Wang Y, Xiong F, Nong S, Liao J, Xing A, Shen Q, Ma Y, Fang W, Zhu X (2020) Effects of nitricoxide on the GABA, polyamines, and proline in tea (Camellia sinensis) roots under cold stress. Sci Rep 10:12240. https://doi.org/10.1038/s41598-020-69253-y
Wei QC, Chang LP (2009) Study on preparation and preservation of competent cells of Agrobacterium tumefaciens. Anhui Agric Sci 37:6342–6343. https://doi.org/10.13989/j.cnki.0517-6611.2009.14.029
Xiong JQ, Yan M, Wang JX, Sun K, Zeng L, Wu ZJ (2023) Genome-wide identification and light response expression analysis of phototropin PHOT family genes in tea plant. Mole Plant Breed 1–29. http://kns.cnki.net/kcms/detail/46.1068.S.20211222.0920.006.html
Yin Y, Wang ZY, Mora-Garcia S, Li J, Yoshida S, Asami T, Chory J (2002) BES1 accumulates in the nucleus in response to brassinosteroids to regulate gene expression and promote stem elongation. Cell 109:181–191. https://doi.org/10.1016/S0092-8674(02)00721-3
Yue Z, Liu H, Ma F (2015) The Malus carotenoid cleavage dioxygenase 7 is involved in stress response and regulated by basic pentacysteine 1. Scientia Horticulturae 192:264–270. https://doi.org/10.1016/j.scienta.2015.06.027
Zhang CX, Wang WQ, Jiang XN, Chen XM (2004) Research progress on plant gene promoters. Acta Genet Sin 12:1455–1464
Zhou L, Hao Y, Lu G, Wang P, Guo H, Cheng H (2020) Cloning and functional analysis of AmDUF1517 promoter from Ammopiptanthus mongolicus. J Biosci Bioeng 130:233–238. https://doi.org/10.1016/j.jbiosc.2020.04.014
Zhu SW, Shi ZW, Xu SF, Zhu XC (1998) Methods for rapid tobacco DNA extraction. J Northeast Agric Univ 29:4. https://doi.org/10.19720/j.cnki.issn.1005-9369.1998.03.009
Zhu J, Quan J, He Y, Chen Y, Guo D, Li C, Cao T, Cao X (2006) Preparation of competent cells of Agrobacterium tumefaciens and study on the transformation of plasmid Prok. J Northwest a&f Univ. https://doi.org/10.13207/j.cnki.jnwafu.2006.07.020
Zhuang Y, Sharif Y, Zeng X, Chen S, Chen H, Zhuang C, Deng Y, Ruan M, Chen S, Weijian Z (2023) Molecular cloning and functional characterization of the promoter of a novel Aspergillus flavus inducible gene (AhOMT1) from peanut. Front Plant Sci 14:1102181. https://doi.org/10.3389/fpls.2023.1102181
Zou ZW, Fang WP, Zhang D, Duan YS, Li XH (2008) Differential analysis of gene expression in tea plants under low temperature stress. Tea Sci 04:249–254
Acknowledgements
This study is supported by the National Natural Science Foundation of China "Function Analysis of Brassinosteroid Transcription Factor CsBZRs in Cold stress Resistance of Tea Plant" (Project Number: 32160077).
Funding
This work was supported by the National Natural Science Foundation, under Grant number 32160077.
Author information
Authors and Affiliations
Contributions
ZYC contributed to the study conception and design. Material preparation, data collection and analysis were performed by ZT. The first draft of the manuscript was written by ZT, and ZYC commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Communicated by Wenwu Guo.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, T., Zhao, Y. Cloning and characterization of the BZR1-2 promoter from Camellia sinensis and its responses to hormonal and abiotic stresses. Plant Cell Tiss Organ Cult 155, 719–729 (2023). https://doi.org/10.1007/s11240-023-02592-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02592-6