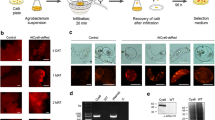
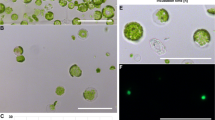
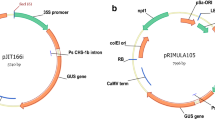
Abstract
Analysis of proteins often requires generation of transgenic plants to gain knowledge of their in planta function. The acquisition of such plants represents a significant investment in terms of workload, time, and space. Here we describe an easy-to-use alternative system by transforming Brassica napus cv. Jet Neuf suspension cells derived from microspores. The cells can be transformed with high efficiency, and even double transformations with different transgenes are possible. In addition, we describe a procedure that allows for long-term storage to maintain diverse transgenic cell lineages with low effort, to build up cell suspension libraries for research purposes. Overall, the work can be helpful for scientists that want to use B. napus as a research organism in a reduced time-frame without the need for generating whole plants and the requirement of larger greenhouse spaces.
Key message
This study establishes a protocol for generation of stable Brassica napus transgenic cell cultures, their long-term storage, and contains examples how these cell cultures can be used in studying gene function in planta without generating whole plants.




Similar content being viewed by others
Data availability
The datasets of the current study are presented in this article or are available upon a reasonable request.
References
Acquaviva C, Brockly F, Ferrara P, Bossis G, Salvat C, Jariel-Encontre I, Piechaczyk M (2001) Identification of a C-terminal tripeptide motif involved in the control of rapid proteasomal degradation of c-Fos proto-oncoprotein during the G(0)-to-S phase transition. Oncogene 20(51):7563–7572. https://doi.org/10.1038/sj.onc.1204880
Ahmad M, Hirz M, Pichler H, Schwab H (2014) Protein expression in Pichia pastoris: recent achievements and perspectives for heterologous protein production. Appl Microbiol Biot 98(12):5301–5317. https://doi.org/10.1007/s00253-014-5732-5
Alahakoon AY, Tongson E, Meng W, Ye ZW, Russell DA, Chye ML, Golz JF, Taylor PWJ (2022) Overexpressing Arabidopsis thaliana ACBP6 in transgenic rapid-cycling Brassica napus confers cold tolerance. Plant Methods 18(1) ARTN 62. https://doi.org/10.1186/s13007-022-00886-y
Aparicio-Fernandez X, Reynoso-Camacho R, Castano-Tostado E, Garcia-Gasca T, de Mejia EG, Guzman-Maldonado SH, Elizondo G, Yousef GG, Lila MA, Loarca-Pina G (2008) Antiradical capacity and induction of apoptosis on HeLa cells by a Phaseolus vulgaris extract. Plant Food Hum Nutr 63(1):35–40. https://doi.org/10.1007/s11130-007-0066-4
Arif B, Pavlik L (2013) Insect cell culture: virus replication and applications in biotechnology. J Invert Pathology 112:S138–S141. https://doi.org/10.1016/j.jip.2012.07.011
Banno H, Ikeda Y, Niu QW, Chua NH (2001) Overexpression of Arabidopsis ESR1 induces initiation of shoot regeneration. Plant Cell 13(12):2609–2618. https://doi.org/10.1105/tpc.13.12.2609
Bhalla PL, Singh MB (2008) Agrobacterium-mediated transformation of Brassica napus and Brassica oleracea. Nat Protoc 3(2):181–189. https://doi.org/10.1038/nprot.2007.527
Bihmidine S, Lin J, Stone JM, Awada T, Specht JE, Clemente TE (2013) Activity of the Arabidopsis RD29A and RD29B promoter elements in soybean under water stress. Planta 237(1):55–64. https://doi.org/10.1007/s00425-012-1740-9
Boo SH, Kim YK (2020) The emerging role of RNA modifications in the regulation of mRNA stability. Exp Mol Medicine 52(3):400–408. https://doi.org/10.1038/s12276-020-0407-z
Brandizzi F, Irons S, Kearns A, Hawes C (2003) BY-2 cells: culture and transformation for live cell imaging. Curr Protoc Cell Biol Chap 1: Unit 1 7. https://doi.org/10.1002/0471143030.cb0107s19
Cardoza V, Stewart CN (2003) Increased Agrobacterium-mediated transformation and rooting efficiencies in canola (Brassica napus L.) from hypocotyl segment explants. Plant Cell Rep 21(6):599–604. https://doi.org/10.1007/s00299-002-0560-y
Carswell MC, Grant BR, Plaxton WC (1997) Disruption of the phosphate-starvation response of oilseed rape suspension cells by the fungicide phosphonate. Planta 203(1):67–74. https://doi.org/10.1007/s004250050166
Curtis MD, Grossniklaus U (2003) A gateway cloning vector set for high-throughput functional analysis of genes in planta. Plant Phys 133(2):462–469. https://doi.org/10.1104/pp.103.027979
Efferth T (2019) Biotechnology applications of plant callus cultures. Engineering-Prc 5(1):50–59. https://doi.org/10.1016/j.eng.2018.11.006
Fischer R, Vaquero-Martin C, Sack M, Drossard J, Emans N, Commandeur U (1999) Towards molecular farming in the future: transient protein expression in plants. Biotechnol Appl Bioc 30:113–116
Fiserova J, Kiseleva E, Goldberg MW (2009) Nuclear envelope and nuclear pore complex structure and organization in tobacco BY-2 cells. Plant J 59(2):243–255. https://doi.org/10.1111/j.1365-313X.2009.03865.x
Hakkinen ST, Reuter L, Nuorti N, Joensuu JJ, Rischer H, Ritala A (2018) Tobacco BY-2 Media Component Optimization for a Cost-Efficient Recombinant Protein Production. Front Plant Sci 9. https://doi.org/10.3389/fpls.2018.00045
Hellwig S, Drossard J, Twyman RM, Fischer R (2004) Plant cell cultures for the production of recombinant proteins. Nat Biotechn 22(11):1415–1422. https://doi.org/10.1038/nbt1027
Hosseini R, Mulligan BJ (2002) Application of rice (Oryza sativa L.) suspension culture in studying senescence in vitro (I). Single strand preferring nuclease activity. Electron J Biotechn 5(1):15–16
Iwase A, Harashima H, Ikeuchi M, Rymen B, Ohnuma M, Komaki S, Morohashi K, Kurata T, Nakata M, Ohme-Takagi M, Grotewold E, Sugimoto K (2017) WIND1 promotes shoot regeneration through Transcriptional activation of ENHANCER OF SHOOT REGENERATION1 in Arabidopsis. Plant Cell 29(1):54–69. https://doi.org/10.1105/tpc.16.00623
Iwase A, Mita K, Favero DS, Mitsuda N, Sasaki R, Kobayshi M, Takebayashi Y, Kojima M, Kusano M, Oikawa A, Sakakibara H, Saito K, Imamura J, Sugimoto K (2018) WIND1 induces dynamic metabolomic reprogramming during regeneration in Brassica napus. Develop Biol 442(1):40–52. https://doi.org/10.1016/j.ydbio.2018.07.006
Jefferson RA (1989) The Gus Reporter Gene System. Nature 342(6251):837–838. https://doi.org/10.1038/342837a0
Kalengamaliro NE, Gana JA, Cunningham SM, Volenec JJ (2000) Mechanisms regulating differential freezing tolerance of suspension cell cultures derived from contrasting alfalfa genotypes. Plant Cell Tiss Org 61(2):143–151. https://doi.org/10.1023/A:1006408829105
Karimi M, Inze D, Depicker A (2002) GATEWAY vectors for Agrobacterium-mediated plant transformation. Trends Plant Sci 7(5):193–195. https://doi.org/10.1016/s1360-1385(02)02251-3
Kumagai-Sano F, Hayashi T, Sano T, Hasezawa S (2006) Cell cycle synchronization of tobacco BY-2 cells. Nat Protoc 1(6):2621–2627. https://doi.org/10.1038/nprot.2006.381
Kutik J, Kuthanova A, Smertenko A, Fischer L, Opatrny Z (2014) Cadmium-induced cell death in BY-2 cell culture starts with vacuolization of cytoplasm and terminates with necrosis. Physiol Plant 151(4):423–433. https://doi.org/10.1111/ppl.12124
Lichter R (1982) Induction of haploid plants from isolated pollen of Brassica-napus. Z Pflanzenphysiol 105(5):427–434. https://doi.org/10.1016/S0044-328x(82)80040-8
Linh TM, Mai NC, Hoe PT, Ngoc NT, Thao PTH, Ban NK, Van NT (2021) Development of a cell suspension culture system for promoting alkaloid and vinca alkaloid biosynthesis using endophytic fungi isolated from local Catharanthus roseus. Plants 10(4). https://doi.org/10.3390/plants10040672
Liu XX, Lang SR, Su LQ, Liu X, Wang XF (2015) Improved Agrobacterium-mediated transformation and high efficiency of root formation from hypocotyl meristem of spring Brassica napus ‘precocity’ cultivar. Genet Mol Res 14(4):16840–16855. https://doi.org/10.4238/2015.December.14.11
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Loh CS, Shu W, Koh WL (2004) Regeneration of plants via somatic embryogenesis. In: Pua E, Douglas CJ (eds) Brassica. Biotechnology in Agriculture and Forestry, vol 54. Springer, Berlin, Heidelberg
Ma WW, Xu WZ, Xu H, Chen YS, He ZY, Ma M (2010) Nitric oxide modulates cadmium influx during cadmium-induced programmed cell death in tobacco BY-2 cells. Planta 232(2):325–335. https://doi.org/10.1007/s00425-010-1177-y
Madhusudhan R, Ishikawa T, Sawa Y, Shigeoka S, Shibata H (2003) Characterization of an ascorbate peroxidase in plastids of tobacco BY-2 cells. Physiol Plant 117(4):550–557. https://doi.org/10.1034/j.1399-3054.2003.00066.x
Maiorella B, Inlow D, Shauger A, Harano D (1988) Large-scale insect cell-culture for recombinant protein-production. Bio-Technol 6(12):1406–1410. https://doi.org/10.1038/nbt1288-1406
Mooney S, Al-Saharin R, Choi CM, Tucker K, Beathard C, Hellmann HA (2019) Characterization of Brassica rapa RAP2.4-Related proteins in stress response and as CUL3-Dependent E3 ligase substrates. Cells 8(4). https://doi.org/10.3390/cells8040336
Mustafa NR, de Winter W, van Iren F, Verpoorte R (2011) Initiation, growth and cryopreservation of plant cell suspension cultures. Nat Protoc 6(6):715–742. https://doi.org/10.1038/nprot.2010.144
Nagata T, Nemoto Y, Hasezawa S (1992) Tobacco by-2 cell-line as the hela-cell in the cell biology of higher-plants. Int Rev Cytol - a Survey of Cell Biology 132:1–30. https://doi.org/10.1016/S0074-7696(08)62452-3
Naill MC, Kolewe ME, Roberts SC (2012) Paclitaxel uptake and transport in Taxus cell suspension cultures. Biochem Eng J 63:50–56. https://doi.org/10.1016/j.bej.2012.01.006
Nitsch JP, Nitsch C (1969) Haploid plants from pollen grains. Science 163(3862):85–87. https://doi.org/10.1126/science.163.3862.85
Niu Y, Wu GZ, Ye R, Lin WH, Shi QM, Xue LJ, Xu XD, Li Y, Du YG, Xue HW (2009) Global analysis of gene expression profiles in Brassica napus developing seeds reveals a conserved lipid metabolism regulation with Arabidopsis thaliana. Mol Plant 2(5):1107–1122. https://doi.org/10.1093/mp/ssp042
Nykiforuk CL, Furukawa-Stoffer TL, Huff PW, Sarna M, Laroche A, Moloney MM, Weselake RJ (2002) Characterization of cDNAs encoding diacylglycerol acyltransferase from cultures of Brassica napus and sucrose-mediated induction of enzyme biosynthesis. Bba-Mol Cell Biol L 1580(2–3):95–109. Pii S1388-1981(01)00200-1. https://doi.org/10.1016/S1388-1981(01)00200-1
Orr W, Keller WA, Singh J (1986) Induction of freezing tolerance in an embryogenic cell-suspension culture of Brassica napus by abscisic-acid at room-temperature. J Plant Phys 126(1):23–32. https://doi.org/10.1016/S0176-1617(86)80212-7
Orr W, Johnsonflanagan AM, Keller WA, Singh J (1990) Induction of freezing tolerance in microspore-derived embryos of winter Brassica napus. Plant Cell Rep 8(10):579–581. https://doi.org/10.1016/S0176-1617(86)80212-7
Ozyigit II, Kurtoglu KY (2020) Particle bombardment technology and its applications in plants. Mol Biol Rep 47(12):9831–9847. https://doi.org/10.1007/s11033-020-06001-5
Parker JL, Newstead S (2014) Method to increase the yield of eukaryotic membrane protein expression in Saccharomyces cerevisiae for structural and functional studies. Protein Sci 23(9):1309–1314. https://doi.org/10.1002/pro.2507
Potter H, Heller R (2018) Transfection by electroporation. Curr Protoc Mol Biol 121(3 1–9 3 13):9. https://doi.org/10.1002/cpmb.48
Pratt AI, Knoblauch J, Kunz HH (2020) An updated pGREEN-based vector suite for cost-effective cloning in plant molecular biology. MicroPubl Biol 2020. https://doi.org/10.17912/micropub.biology.000317
Rajabi F, Jessat J, Garimella JN, Bok F, Steudtner R, Stumpf T, Sachs S (2021) Uranium(VI) toxicity in tobacco BY-2 cell suspension culture - A physiological study. Ecotox Environ Safe 211. https://doi.org/10.1016/j.ecoenv.2020.111883
Reece-Hoyes JS, Walhout AJM (2012) Yeast one-hybrid assays: a historical and technical perspective. Methods 57(4):441–447. https://doi.org/10.1016/j.ymeth.2012.07.027
Reed KM, Bargmann BOR (2021) Protoplast regeneration and its use in new plant breeding technologies. Front Genome Ed 3:734951. https://doi.org/10.3389/fgeed.2021.734951
Rosano GL, Ceccarelli EA (2014) Recombinant protein expression in Escherichia coli: advances and challenges. Front Microbiol 5. https://doi.org/10.3389/fmicb.2014.00172
Shang HS, Chang CH, Chou YR, Yeh MY, Au MK, Lu HF, Chu YL, Chou HM, Chou HC, Shih YL, Chung JG (2016) Curcumin causes DNA damage and affects associated protein expression in HeLa human cervical cancer cells. Oncol Rep 36(4):2207–2215. https://doi.org/10.3892/or.2016.5002
Shi YZ, Xu GB, Warrington TB, Murdoch GK, Kazala EC, Snyder CL, Weselake RJ (2008) Microspore-derived cell suspension cultures of oilseed rape as a system for studying gene expression. Plant Cell Tiss Org 92(2):131–139. https://doi.org/10.1007/s11240-007-9313-5
Simmonds DH, Long NE, Keller WA (1991) High plating efficiency and plant-regeneration frequency in low-density protoplast cultures derived from an embryogenic Brassica napus cell-suspension. Plant Cell Tiss Org 27(3):231–241. https://doi.org/10.1007/Bf00157586
Sultana S, Frazier TP, Millwood RJ, Lenaghan SC, Stewart CN (2019) Development and validation of a novel and robust cell culture system in soybean (Glycine max (L.) Merr.) For promoter screening. Plant Cell Rep 38(10):1329–1345. https://doi.org/10.1007/s00299-019-02455-5
Winter D, Vinegar B, Nahal H, Ammar R, Wilson GV, Provart NJ (2007) An “Electronic Fluorescent Pictograph” Browser for Exploring and Analyzing Large-Scale Biological Data Sets. PloS one 2(8). https://doi.org/10.1371/journal.pone.0000718
Yamaguchi-Shinozaki K, Shinozaki K (1994) A novel cis-acting element in an Arabidopsis gene is involved in responsiveness to drought, low-temperature, or high-salt stress. Plant Cell 6(2):251–264. https://doi.org/10.1105/tpc.6.2.251
Zhai Z, Keereetaweep J, Liu H, Feil R, Lunn JE, Shanklin J (2018) Trehalose 6-Phosphate positively regulates fatty acid synthesis by stabilizing WRINKLED1. Plant Cell 30(10):2616–2627. https://doi.org/10.1105/tpc.18.00521
Zhang LN, Smertenko T, Fahy D, Koteyeva N, Moroz N, Kucharova A, Novak D, Manoilov E, Smertenko P, Galva C, Samaj J, Kostyukova AS, Sedbrook JC, Smertenko A (2021) Analysis of formin functions during cytokinesis using specific inhibitor SMIFH2. Plant Phys 186(2):945–963. https://doi.org/10.1093/plphys/kiab085
Acknowledgements
We would like to thank Prof. Thomas Clemente, Center for Plant Science Innovation, Department of Agronomy and Horticulture, University of Nebraska-Lincoln, for providing the RD29a promoter. We also would like to thank Dr. John Shanklin, Brookhaven National Laboratory, NY, for providing the stock of B. napus cv. Jet Neuf cell cultures used in this work, and WSU’s Franceschi Microscopy and Imaging Center for providing access to a confocal microscope and supporting undergraduate research.
Funding
This project was supported by an Agriculture and Food Research Initiative competitive grant 2019-67013-29160 of the USDA National Institute of Food and Agriculture (NIFA) to H.H. WSU financially supported J.L. in his undergraduate research studies through a CAS Summer Fellowship.
Author information
Authors and Affiliations
Contributions
J.L. is an undergraduate student of WSU’s Honors College who established the transformation and long storage procedures, and also performed the GUS stainings shown in this work. S.M. did the RT-qPCR analysis, S.S. did salt and low-sucrose with salt assays, M.K. did the confocal analysis, and R.A-S. generated the proESR1:GUS and proRD29a:BrRAP2.4-1 expression constructs. H.H. is the principal investigator, designed the figures, and developed the manuscript together with J.L. All authors contributed with editing and agreed on the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Heidi Halbwirth.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.


Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Landers, J., Mooney, S., Smalley, S. et al. Generation of stable transgenic Brassica napus cv. Jet Neuf cell cultures as a tool to investigate in planta protein function. Plant Cell Tiss Organ Cult 154, 633–643 (2023). https://doi.org/10.1007/s11240-023-02538-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-023-02538-y